ภาพนิ่ง 1
advertisement

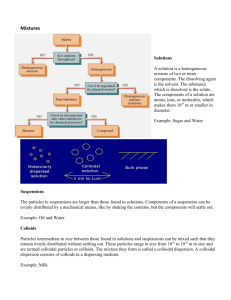
Introduction to Colloid & Surface Chemistry โดย รองศาสตราจารย์ เรืองศรี วัฒเนสก์ ruangsri@gmail.com 1 CHEM 324 : Physical Chemistry IV Part II Department of Chemistry, Faculty of Science, CMU. August 2009 Content ö The Colloidal State : Classification & preparation Surfactant & micellization ö The Properties of Colloidal Dispersion (Size & shape determination) : Colligative properties Kinetic properties Optical property ö Colloidal Stability and Its Applications The electrical double layer ö The gas-liquid and liquid-liquid interfaces ö The adsorption of gas on the solid surface ö The liquid-solid interface 2 References 1. Atkins, P. W., Physical Chemistry, 8th ed., Oxford University Press, Oxford, 2006. 2. Hiemenz, P. C. and Rajagopalan, R., Principles of Colloid Chemistry and Surface Chemistry : Revised and Expanded, 3rd ed., Marcel Dekker, New York, 1997. 3. Hiemenz, P. C., Principles of Colloid Chemistry and Surface Chemistry, Marcel Dekker, New York, 1977. 4. Hunter, R, J., Introduction to Modern Colloid Science, Oxford University Press, Oxford, 1998. 5. Shaw, D. J., Colloid &Surface Chemistry, 4th ed., Butterworth, Oxford, 1998. 6. http://wikipedia.com and other websites 3 Evaluation • Assignment/Homework/Quiz • Report/Presentation • Final Exam Total 20 points 20 points 60 points 100 points (50%) (Final exam : Sept 24, 2009 : 12-15 pm.) 4 1. The Colloidal State Definition : particle with linear dimension between 10-7 cm (10 AO) and 10-4 cm (1 ) 1 - 1000 nm Other definitions particle weight/ particle size etc. 5 Length Scales C-C bond Radius of Gyration Micelles Vesicles Emulsion Droplets Latex Particles “Colloidal Crystals” 0.1 nm 1 nm 10 nm 100 nm 1000 nm 6 Homogeneous Mixtures (True composition and properties) Property Particle type True solution Colloid Ions, small Large molecules, molecules particles Suspension Large particles Particle size 0.1-1 nm 1-100 nm 100 nm and larger Tyndall effect No Yes Yes 7 Colloidal Systems Dispersed Medium Gas Cont. Medium Gas Liquid Solid NONE Liquid Aerosol Fog, mist Solid Aerosol Smoke, air particulates Sol Paint, pigmented ink, blood Solid Sol Cranberry glass, ruby glass Liqui d Foam Whipped cream Emulsion Milk, mayonnaise, hand cream Solid Solid Foam Aerogel, styrofoam, pumice Gel Gelatin, jelly, cheese, opal 8 Macromolecular Colloids Colloidal Dispersion aerosols, emulsions, sol,foam, paste Silica gel Basalt Zeolite Associated Colloids Surfactants “surface active agents” Micelle formation 9 CMC determinations 10 Common Surfactant Classes _ + N Br Cationic cetyltrimethylammonium bromide (CTAB) _ O O O Na+ S O Anionic sodium dodecylsulphate (SDS) Non-ionic O O O OH polyethylene oxides (CiEj) 11 C12E6 adsorbed at the air-water interface in the presence of oil 12 13 14 Biocolloids myoglobin haemoglobin Nano “golf balls” Multiple Colloids Megaspore wall from a fossil of Salvinia cerebrata. 15 Preparation of Colloidal Systems Two basic methods of forming a colloid : reduction of larger particles to colloidal size condensation of smaller particles into colloidal particles i.e. • spontaneous dispersion in the proper solvent • metal evaporation in an electric arc to form a metal vapor cools. • solid reduction in a colloid mill. • emulsion preparation by homogenization with the addition of an emulsifying agent. • Condensation of smaller particles to form a colloid usually involves chemical reactions—typically displacement, hydrolysis, or oxidation and reduction • making colloidal gold, colloidal silver. 16 Some areas of Science and Technology in which particles in the colloidal size range are regularly encountered Analytical chemistry : adsorption indicators, ion exchange, nephelometry, precipitate filterability, chromatography, and decolorization Physical chemistry : nucleation; superheating, supercooling and supersaturation; and liquid crystals Biochemistry and molecular biology : electrophoresis; osmotic and Donnan equilibria and other membrane phenomena; viruses, nucleic acids, and proteins; and hematology 17 Chemical manufacturing : catalysis, soaps and detergent, paints, adhesives, ink; paper and paper coating; pigment; thickening agents; lubricants Environmental science : aerosols, fog and smog, foams, water purification and sewage treatment; cloud seeding; clean rooms Materials science : powder metallurgy, alloys, ceramics, cement, fibers, and particles of all sorts Petroleum science, geology and soil science : oil retrieval, emulsification, soil porosity, floatation, and ore enrichment Household and consumer products : milk and dairy products, beer, water proofing, cosmetics, and encapsulated products 18 Assignment -1 1. Search from Journals or websites to find an example, in any branches of Science, of which particles in the colloidal-size range are encountered. 2. Write up in your own words a concise report in A4 paper. 3. Report is due on August 21, 2009. (10 points) 19