Optical Isomerism
advertisement

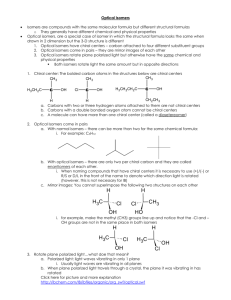
Title: Lesson 10 Optical Isomerism Learning Objectives: – Understand and identify optical isomerism – Understand the chemical significance of optical isomerism Refresh The molecular formula, C3H4Cl2 represents several isomeric compounds. Some isomers are cyclic and some are unsaturated. a) Draw the structures of two cyclic compounds that are structural isomers and state the names of both isomers. b) Two of the non-cyclic compounds have geometrical isomers. Draw the structures of these compounds and their geometrical isomers. Main Menu Modelling Build a model of 2-methylbutan-2-ol Compare your model with your neighbours Have you all made the same thing? Main Menu Stereoisomerism: true or false? 4 of 30 © Boardworks Ltd 2009 Optical isomerism Another form of stereoisomerism is optical isomerism, in which a molecule can exist as two isomers that are nonsuperimposable, mirror images of each other, just like a left hand and right hand. optical isomers of the amino acid alanine Optical isomers have the same physical properties, but they rotate polarized light in opposite directions. 5 of 30 © Boardworks Ltd 2009 Optical Isomerism Optical isomerism occurs when you have at four different groups all bonded to a central atom. For example: amino acids Build these for yourself to see that they are different… They are said to be Chiral. The four groups are arranged tetrahedrally around the carbon atom with bond angles of 109.5o. They can be arranged in two different 3D configurations with mirror images of each other… Main Menu Chiral molecules have no plane of symmetry… Mirror images are non-superimposable… If we put them on top of each other they do not line up… Optical isomers are non-superimposable forms known as enantiomers. Main Menu Some terminology The central carbon is referred to as ‘chiral’ You will see books refer to the ‘chiral carbon’ or ‘chiral centre’ You may see books talking about the ‘chirality’ of a molecule A molecule might be described as ‘chiral’ if it has a ‘chiral centre’ A carbon must have at least 4 different groups to be chiral The two optical isomers are referred to as enantiomers A racemic mixture is a one with a 50:50 mix of the two enantiomers Enantiomers can be referred to as right or left handed If you see ‘R-’ or ‘L-’ in a name, this is what they are referring to There is a standard way to work this out but we don’t need it! Main Menu Molecules that have more than one chiral centre… Enantiomers have opposite configurations at each chiral centre… So the whole molecule will be a mirror image. Diastereomers will have opposite configurations at some but not all chiral centres… So they will not be a mirror image of each other Main Menu Main Menu Properties of optical isomers Enantiomers share virtually identical chemical and physical properties However: Two enantiomers will rotate planepolarised light in opposite directions…hence the term ‘optical isomerism’ More on the next slide Chemical properties differ significantly in chemical systems where 3D-shape is important, particularly biochemistry The image above right shows an enzyme, these are sensitive to the exact shape of a molecule Main Menu Rotation of plane-polarised light Light normally vibrates in many different directions Plane-polarised light only vibrates in one direction The polarizer blocks out light waves in all other planes. Only plane polarised light passes through. A pure solutions of enantiomers rotate the plane-polarised light in opposite directions To compare different solutions, you need the same: • Concentrations of the solutions • Wavelength of light • Sample path A racemic mixture will not rotate light as the rotations from each enantiomer cancel each other out… OPTICALLY INACTIVE!!! This rotation can be detected using a polarimeter. The analyser is a second polarizer that can be detected until the rotated light passes through. Extent and direction of rotation can be deduced. Richard Thornley - Distinguishing Between Optical Isomers Video Main Menu Reactivity with other chiral molecules Left rotation of plane polarized light is (-) Right rotation of plane polarized light is (+) When a racemic mixture is reacted with a single isomer of another chiral compound, the (-) and (+) components react to form two different products. These products have distinct chemical and physical properties. These can be separated relatively easily, so can be used to separate the enantiomers from the racemic mixture, a process known as resolution. Main Menu Main Menu Solutions Main Menu Main Menu To do: Assess whether or not the following compounds display optical isomerism. If they do, draw each of the two enantiomers in 3D and place a * next to the chiral carbon. Build them from molecular models if it helps: 2-chloropropane 2-chlorobutane 2-chloropropanal chlorocyclopropane 1,2-dichlorocyclobutane Research the importance of optical isomerism in biochemistry. Give details of two important examples. Draw a Venn diagram summarising geometrical and optical isomerism Main Menu Key Points Optical isomers (enantiomers) rotate plane-polarised light in opposite directions To display optical activity, a compound must have at least four different groups attached to the same atom The central atom is referred to as chiral Main Menu