Lesson-4-Halogenoalkanes-
advertisement

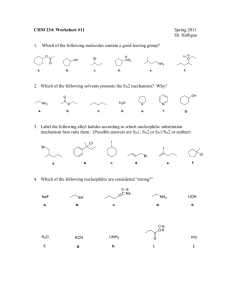
Halogenoalkanes Halogenoalkanes substitution reactions of halogenoalkanes with hydroxide https://www.youtube.com/watch?v=hqJ1ciZDXH8&index=25&list=PL73D2B CC23E83D555 Explain why the hydroxide ion is a better nucleophile than water https://www.youtube.com/watch?v=Do8ugMmvMs&index=29&list=PL73D2BCC23E83D555 Describe and explain rates of nucleophilic substitution https://www.youtube.com/watch?v=SBxsoWUxQbg&index=30&list=PL73D 2BCC23E83D555 Explain how the rate of Sn1/Sn2 of halogenoalkanes by OH- changes https://www.youtube.com/watch?v=SBxsoWUxQbg&index=30&list=PL73D 2BCC23E83D555 Animation: https://www.youtube.com/watch?v=TnY1S5IdVqI Nucleophilic Substitution RX on primary halogenoalkane: SN2 Mechanisms C2H5Cl + OH- chloroethane C2H5OH + Clethanol Bimolecular Reaction : rate=k[OH-][CH3CH2Cl] proportional to both reactants= order 2 - Inversion of the arrangement of the atoms around C = Stereospecific. - Faster reaction favored by aprotic and polar solvent: OH- is not solvated by solvent and reacts faster. Reaction profile for an SN2 reaction ( ONE STEP- one Ea ) Nucleophilic Substitution RX on tertiary halogenoalkane: SN1 Mechanisms (CH3)3CCl + OH- (CH3)3COH + Cl- 2-chloro-2-methylpropane Steric hindrance: Difficult for incoming group to react due to bulky 3 CH3 groups Unimolecular Rx 2-methyl-propan-2-ol Positive induction: Stabilization effect on the carbocation rate =k[(CH3)3CCl] determined by unimolecular slow step = order 1 OHOH- Y Cl OH-Cl- During both steps, there is some arrangement of the atoms which causes an energy maximum. This is called a transition state ( ts1 and ts2 ) The stability of the carbocation intermediate is shown by the fact that there are small activation barriers to its conversion either into product or back into reactants. Reaction favored by protic and polar solvent (with OH or NH) able to stabilize the intermediate carbocation by ion-dipole interaction and the leaving group ( ex: Cl-) by proton. Comparison of the rates of nucleophilic substitutions SN1 SN2 Mechanism: Tertiary C > SN1 Secondary > Primary SN1- SN2 SN2 Leaving halogen: C-I > longer- weaker C-Br > C-Cl > C-F shorter-stronger Solvent: - SN1 favored by polar, protic solvent (with OH or NH). Stabilization of carbocation. - SN2 favored by polar, aprotic solvent. OH- non solvated (bare) The faster reactions (SN1) occur with tertiary iodoalkanes in polar, protic solvents. lower Rate of reaction ( C-I> C-Br > C-Cl > C-F) rate = k [OH-] [CH3Cl] rate = k [CH3Cl] higher One more tutorial on : -SN2 https://www.youtube.com/watch?v=h5xvaP6bIZI -SN1 https://www.youtube.com/watch?v=JmcVgE2WKBE -Solvent effect https://www.khanacademy.org/science/organic-chemistry/substitutionelimination-reactions/sn1-sn2-sal/v/sn1-sn2-solvent-effects One last animation : http://www.colby.edu/chemistry/OChem/DEMOS/Substitution.html We SN1 and SN2