physical properties cleavage
advertisement

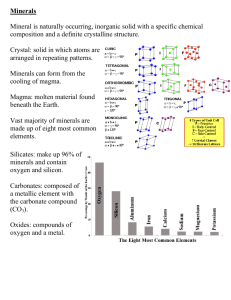
PHYSICAL PROPERTIES OF MINERALS Mineral Identification Basics What is a Mineral? There is a classic definition for mineral. Minerals must be: Inorganic Occurs Naturally on Earth Always a Solid Has Definite Chemical Composition Crystalline Structure Cubic Fluorite Crystal Mineral Identification Basics What is a Mineral? Naturally Occurring Minerals are not synthetic or man made - they are produced by the natural geological processes working on Earth. Tourmaline Crystal from Brazil Mineral Identification Basics What is a Mineral? Inorganic Barite Rose - A flower like growth of Barite crystals. Minerals are NOT produced by organic living processes. As a result things like pearls, coral, coal and amber are NOT considered minerals. Mineral Identification Basics What is a Mineral? Crystalline Structure Minerals are the result of atoms joining together through electrical bonds to produce a definite internal structure. Crystalline Pattern of Halite Halite (salt) from Searles Lake, CA Red = Sodium Green = Chlorine (*) Mineral Identification Basics What is a Mineral? Definite Chemical Composition Halite - NaCl For every atom of Sodium there is an atom of Chlorine. Minerals can be expressed by a chemical formula. The internal order of minerals means that there is a definite relationship in the number of atoms that makes up the mineral. Mineral Identification Basics PHYSICAL PROPERTIES HARDNESS HARDNESS is defined as the resistance a mineral has to being scratched - its “scratchability”. Hardness tests are done by scratching one mineral against another. The mineral that is scratched is softer than the other. Pyrite Crystals Hardness of 6.5 Mineral Identification Basics PHYSICAL PROPERTIES HARDNESS In this photo, a quartz crystal will be rubbed across a glass plate. The result is that the glass plate will be scratched. The quartz is therefore harder than the glass. Quartz is harder than glass. Mineral Identification Basics PHYSICAL PROPERTIES HARDNESS Care must be taken on some minerals that crumble easily. Remember that hardness is the resistance a mineral has to being scratched - NOT how easily it breaks apart. Mineral Identification Basics PHYSICAL PROPERTIES HARDNESS MOH’S SCALE OF MINERAL HARDNESS 1. TALC 6. FELDSPAR 2. GYPSUM 3. CALCITE 4. FLUORITE 5. APATITE 7. QUARTZ 8. TOPAZ 9. CORUNDUM 10. DIAMOND OTHER MATERIALS COMMONLY USED: Moh’s scale is a list of minerals with increasing hardness.(*) 2.5 - FINGERNAIL 3 - COPPER PENNY 5.5 - GLASS 6-6.5 - STEEL FILE Mineral Identification Basics PHYSICAL PROPERTIES CLEAVAGE CLEAVAGE is the property of a mineral that allows it to break repeatedly along smooth, flat surfaces. These GALENA cleavage fragments were produced when the crystal was hit with a hammer. Note the consistency of the 90o angles along the edges. These are FLUORITE cleavage fragments. Mineral Identification Basics PHYSICAL PROPERTIES CLEAVAGE Within this crystalline pattern it is easy to see how atoms will separate to produce cleavage with cubic (90o) angles. (*) It is similar to tearing a piece of paper that has perforations in it. The paper has a tendency to tear along the perforations. They are zones of weakness. (*) In this example the lines represent breaks between the atoms that make up the mineral. Cleavage is guided by the atomic structure. (*) Mineral Identification Basics PHYSICAL PROPERTIES CLEAVAGE These pictures show different cleavage angles and the quality of cleavage. Fluorite has cleavage in four directions. (*) Mica A thin has sheet perfect of Muscovite cleavage inseen ONE on direction. edge. (*) Mineral Identification Basics PHYSICAL PROPERTIES CLEAVAGE Common salt (the mineral HALITE) has very good cleavage in 3 directions. (*) These 3 directions of cleavage are mutually perpendicular resulting in cubic cleavage. (*) Mineral Identification Basics PHYSICAL PROPERTIES CLEAVAGE Rhombohedral Cleavage - 3 directions CALCITE Even these tiny fragments have rhombohedral cleavage. (*) (*) Mineral Identification Basics PHYSICAL PROPERTIES FRACTURE FRACTURE is defined as the way a mineral breaks other than cleavage. (*) This is a piece of volcanic glass called OBSIDIAN. Even though it is NOT a mineral, it is shown here because it has excellent conchoidal fracture. (*) If you try this yourself, use caution. Conchoidal fracture in obsidian can produce extremely sharp edges. (*) Mineral Identification Basics PHYSICAL PROPERTIES FRACTURE This Quartz crystal will be struck with a hammer to show how that the external form of the crystal does not repeat when broken. (The flat crystal faces are not cleavage faces.) This is a good example of conchoidal fracture. (*) Note the smooth curved surfaces. (*) Mineral Identification Basics PHYSICAL PROPERTIES STREAK STREAK is defined as the color of the mineral in powder form. (*) Streak is normally obtained by rubbing a mineral across a “streak plate”. This is a piece of unglazed porcelain. The streak plate has a hardness of around 7 and rough texture that allows the minerals to be abraded to a powder. This powder is the streak. (*) Hematite on Streak Plate Hematite has a reddish brown streak. (*) Mineral Identification Basics PHYSICAL PROPERTIES STREAK Sphalerite is a dark mineral, however, it has a light colored streak. Next to the reddish brown streak of hematite is a light yellow streak. This is the streak of the sphalerite. (*) Light colored streaks are often difficult to see against the white streak plate. It is often useful to rub your finger across the powder to see the streak color. (*) Sphalerite has a light yellow streak. (*) Mineral Identification Basics PHYSICAL PROPERTIES LUSTER LUSTER is defined as the quality of reflected light. Minerals have been grossly separated into either METALLIC or NON-METALLIC lusters. Following are some examples: Native Silver has a Metallic Luster. (*) Mineral Identification Basics PHYSICAL PROPERTIES LUSTER METALLIC Stibnite Galena The basic idea for Metallic Luster is that the minerals look like metals. (*) Pyrite (*) Marcasite Mineral Identification Basics NON-METALLIC LUSTER VITREOUS Vitreous Luster means that the mineral has a “glassy” look. Normally we think of glass as being clear, but there are many different colors of glass and they are all very “glassy” looking. Even china plates and glazed porcelain are vitreous. Here are some examples: (*) Olivine - Peridot Spinel Wulfenite Quartz (*) Mineral Identification Basics NON METALLIC LUSTER Miscellaneous Lusters Asbestos - Silky Apophyllite – Pearly (*) Graphite has a greasy or submetallic luster and easily marks paper. (*) Limonite - Dull or Earthy Sphalerite - Resinous Mineral Identification Basics PHYSICAL PROPERTIES COLOR The COLOR of a mineral is usually the first thing that a person notices when observing a mineral. However, it is normally NOT the best physical property to begin the mineral identification process. (*) Following are some examples of color variation within mineral species followed by minerals that have a distinctive color: (*) Various colors of CALCITE. (*) Mineral Identification Basics PHYSICAL PROPERTIES COLOR Amethyst Quartz comes in a wide range of colors. It is very easily colored by even trace amounts of impurities. (*) Ionic Iron Clear - Without Impurities Hematite Inclusions Various colors of Quartz. Chlorite inclusions Mineral Identification Basics INDICATIVE COLOR Some minerals do have a certain color associated with them. Here are some examples: (*) Turquoise Malachite Rhodochrosite Azurite (*) Sulfur Mineral Identification Basics PHYSICAL PROPERTIES SPECIFIC GRAVITY The SPECIFIC GRAVITY of a mineral is a measure of the mineral’s density. It is related to the types of elements that make up the mineral and how they are packed into the mineral’s atomic structure. Gold in Quartz Mineral Identification Basics PHYSICAL PROPERTIES TASTE IT IS NOT RECOMMENDED THAT A TASTE TEST BE PERFORMED ON MINERALS AS A STANDARD PROCESS. SOME MINERALS ARE TOXIC. However, the mineral HALITE is common salt and has a unique taste. (*) Halite cubes from Trona, CA (*) Mineral Identification Basics PHYSICAL PROPERTIES MAGNETISM MAGNETISM is the ability of a mineral to be attracted by a magnet. This most commonly is associated with minerals rich in iron, usually magnetite. This is a piece of MAGNETITE with a magnet adhering to it. Magnetite is a mineral that is strongly magnetic in that a magnet will easily be attracted to it. (*) Mineral Identification Basics PHYSICAL PROPERTIES MAGNETISM This is a sample of “black sand” from Lynx Creek, Arizona. Its dark color is due to its high concentration of magnetite. See what happens when a magnet is place beneath the bottom right portion of the paper. (*) This technique is used to separate out much of the unwanted material in the search for gold in placer deposits. (*) Mineral Identification Basics PHYSICAL PROPERTIES MAGNETISM LODESTONE is a variety of Magnetite that is naturally a magnet. (*) Mineral Identification Basics DOUBLE REFRACTION DOUBLE REFRACTION: Is a property shared by many minerals ( but not those in the isometric crystal system). It is produced by the separating of a beam of light as it passes through the crystal structure. It is best displayed in the mineral CALCITE. This image clearly shows the double image below the calcite. Mineral Identification Basics CHEMICAL PROPERTIES REACTION TO ACID Some minerals, notably the carbonates, react to cold dilute HCl. In this illustration a piece of CALCITE is shown to react (fizz) after HCl is applied. Calcite Reacts to HCl (*) This ends the basic introduction to the Physical Properties of Minerals.