presentation source
advertisement

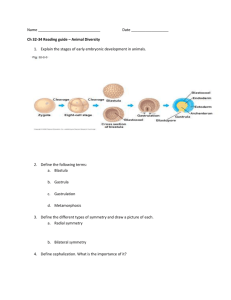
186-225A Properties of Minerals Week 3: cleavage One of the qualities generally required from abrasives used in sandblasting is: • no embedment in target material This will depend partly on the shape of crystals or of their broken fragments. corundum dolomite baking soda garnet Tendency of minerals to break along flat planes is called CLEAVAGE. You can only notice cleavage in specimens where single crystals are large enough (> a few mm). If there is no preferred plane of weakness in the structure, the pattern is described as FRACTURE. A plane of weakness due to defects arising from irregularities during crystal growth is called PARTING. Fe2O3, Al2O3 are based on XO6 polyhedra. Hematite and corundum share the same structure. Their cations are in 6-fold coordination with oxygen anions, and these coordination polyhedra share edges and faces with their neighbours. However, the Fe-O bonds in hematite are not as strong as the AlO bonds in corundum. Cleavage is well developed in solids showing a wide range of bond types. It is absent only in purely metallic solids. halite, NaCl fluorite CaF2 sylvite, KCl Metallic native elements (gold, silver, copper) do not cleave along flat planes. . The sea of mobile delocalized valence electrons acts as a “glue”. The solid deforms plastically and stretches before breaking. This gives an uneven jagged surface described as a “hackly” fracture. Structures with similar type of bonds (here, ionic bonds) but different bond orientationss give rise to different types of cleavage. In halite, the planes of weakness are perpendicular to 3 perpendicular axes. The cleavage is cubic, i.e. along three perpendicular planes. Orientation of Ca-F bonds towards body diagonals of a cube result in an octahedral cleavage. Galena, PbS, has the same structure as halite. It also displays the same cubit cleavage. AgCl, has the same structure as NaCl. It does not cleave as cleanly, but can be cut with a knife (sectile). Bonding among larger ions (compare radii: Ag+ > Na+) gradually takes a more metallic character. In structure with dominantly covalent bonds, the strength of the bonds is not as important as their orientation. In structures with strong bonds, cleavage may be present if some planes cut across a lower density of strong bonds. Sulfur breaks with a conchoidal to uneven fracture. The S8 rings are not aligned predominantly along certain planes within the structure. The weak Van der Waals forces are not restricted to any particular direction. Diamond, a crystalline polymorph of carbon Tetrahedral coordination gives diamond its octahedral cleavage. These planes cut across less C-C bonds per unit area. Cutting a rough diamond into a gemstone requires knowing where to cleave it. A sharp blow is all that’s needed to split the stone. Silicate minerals show combinations of bonds that are partly covalent and ionic. Si-O bonds are the strongest bonds in their structures. Garnet group A2B3(SiO4) 3 Large cations in AO8 polyhedra share edges with BO6 and SiO4 groups: • no good cleavage • dense • hard • rounded grains Quartz is a tectosilicate. All SiO44- units share every oxygen ion at their corners. Unlike diamond, tetrahedra do not point in same direction. Quartz shows no pronounced plane of weakness. A “wire model” of Si-O bonds shows the symmetry which accounts for quartz’ hexagonal crystal shape. A material without any pronounced plane of weakness may break with a conchoidal fracture (right), or an irregular fracture (below). Quartz and garnet are good examples of this type of fracture. olivine (Mg, Fe)SiO4 • XO6 share edges with each other and with SiO4 • low silica content • dense • moderate hardness • no good cleavage • grains equant, subrounded Feldspars: KAlSi3O8 NaAlSi3O8 CaAl2Si2O8 tectosilicates There are 2 quasi-perpendicular planes of weakness where more Ca-O, K-O or Na-O bonds and less Si-O bonds are broken. Single crystals of feldspars break with blocky outlines that are quasiperpendicular. Platy or micaceous cleavage refers to a plane of weakness in a single direction. graphite: C-C bond length is 1.42 Å within sheets, but much longer (3.35 Å) between sheets. metallic luster Molybdenite, MoS2, also shows micaceous cleavage and softness. Only VanderWaals bonding between adjacent layers of S ions. It is used as a lubricant, like graphite. phyllosilicates: sheets of SiO4 units are weakly linked by Van der Waals forces or K-O bonds Micas can be cleaved into thin sheets. Mica flakes are used to prepare “mica paper” which is an insulating material for electric equipment.