Heat & Temperature
advertisement

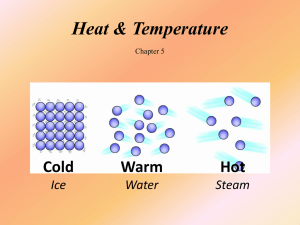
Heat & Temperature Chapter 5 Student Learning Objectives •Relate kinetic energy to temperature •Compare temperature scales •Analyze systems to determine methods & affects of heat transfer What does temperature measure? Temperature is a measure of the average kinetic energy of the atoms and molecules in a substance. We perceive the KE of atoms and molecules as being hot or cold. Thermal Equilibrium Materials at different temperatures come to the same temperature when placed in contact. Thermometer + Person Person + Metal Chair Thermal Expansion Most materials expand when the temperature is increased. Metal lids Bridge Joints Bimetallic strips Hot Air Balloons Temperature Fahrenheit & Celsius are based on boiling and freezing points of water. Fahrenheit Celsius Freezing (at sea level) Boiling (at sea level) 32o 212o 0o 100o Kelvin is based the average KE of atoms in a gas. F = (9/5)C + 32 C = (5/9)(F – 32) K = C + 273 0 Kelvin = ─273o C = ─460o F Examples Human Body Sun Blue Star Kelvin 310.15 K 5800 K 35000 K Fahrenheit 98.6 ºF 9,980 ºF 62,540 ºF Practice 1) Would you expect the surface temperature of a red star to be higher or lower than the surface temperature of our Sun? 2) What is a cold day on Earth in Fahrenheit? What is a hot day on Earth in Fahrenheit? Convert these temperatures to Celsius and Kelvin. Practice 3) Why do we rub our hands together to warm them? What is the physics behind this practice? 4) Do all substances expand when heated? What is heat? Heat is energy. Heat flows from one object to another if there is a temperature difference. The larger the temperature difference, the faster heat energy flows. Hot → Cold Practice 1)When ice and a warm liquid are combined, a. Ice cools the liquid b. Liquid warms the ice 2) Where would be the fastest place to cool a very hot pizza? a. On the counter b. In the refrigerator c. In the freezer The amount of heat it takes to increase the temperature of a material by 1o C is the specific heat capacity of the material. A substance with a high specific heat capacity will resist changes in temperature. Q = cmDT Practice 1) Does a swimming pool lose heat quickly or slowly during the night? 2) In a standard shower, 120 kg of hot water is used during a 10 minute shower. What is the heat energy required to raise the temperature of the 120 kg of water from 15°C to 61°C? More Practice 3) A solid copper penny is dropped from a height of 10 meters. If all energy is conserved within the penny, what is the temperature change of the penny when it lands on the ground? Thinking About The World Practice 4) Where would you expect a greater temperature change between day and night, in a humid location or a dry location? Why? 5) Are day to night temperature changes larger when the sky is clear or cloudy? What is the greenhouse effect? The greenhouse effect traps heat near the surface of a planet. Greenhouse gasses slow heat loss to space. Questions 1) What contributes most to the CO2 in our atmosphere? 2) Is the greenhouse effect “bad”? 3) What is global warming? What happens to temperature & energy during a phase change? A substance that has reached the temperature required for a phase change maintains its temperature during the phase change. DT = 0 Phase Change All energy lost or gained is used to change the phase of the substance. Evaporation (Liquid to gas) Sublimation (Solid to gas) Condensation (Gas to liquid) The amount of heat energy per kilogram that must be added or removed for a substance to change phase is called latent heat (L). Q = mL IceWaterSteam T (°C) 100 0 Joules Practice 1) Describe the physical processes which occur when –20ºC ice is added to +25º soda. 2) A 50 gram piece of steel is brought into full contact with a large block of 0o C ice, and the system of steel and ice is isolated. If the steel has an initial temperature of 95o C, how much of the ice will melt? More Practice In lab, a 64 gram piece of hot metal (97º C) is brought into contact with 64 grams of cold water (23ºC), and the system is isolated. Which do you expect to change temperature more, metal or water? Why? What are the three methods of heat transfer? Conduction Conduction is the transfer of heat energy from one place to another by direct collisions. then Insulators slow or impede the rate of heat flow. Styrofoam Fiberglass Thermal conductivity of a material depends on the movability of the electrons. Freely moving electrons = High thermal conductivity Pots & Pans Metal Spoons Convection Convection is the transfer of heat energy from one place to another by the bulk motion of groups of molecules in a fluid. Boiling Liquid Oven Sea Breeze Radiation Radiation is the transfer of heat energy from one place to another by electromagnetic waves. (light) Stars Fire People Practice 1) Solids tend to conduct heat better than liquids or gases. Why? 2) Does something that has a high thermal conductivity have a high or low specific heat capacity? More Practice 3) Why does hot fluid rise above cool fluid? 4) Ceiling fans change direction. Why? 5) Why do people tend to wear darker colored clothes in the winter and lighter colored clothes in the summer? Ideal Gas Law In a gas, pressure is determined by temperature, volume, and number of particles. PV = nkT Practice 1) A certain number of gas molecules are contained in a tire. What happens to the tire pressure if temperature increases? 2) A certain number of gas molecules are contained in a balloon. What happens to the balloon pressure if temperature decreases? More Practice 3) When you compress air in a bicycle tire pump, what happens? 4) On a hot day (101ºF), you measure the tire pressure to be 38 psi. The next day, the temperature only reaches 81 ºF. What is the tire pressure on the cooler day? What are the 3 laws of thermodynamics? 1st Law: The amount of heat energy transferred is equal to the change in internal energy plus work. Heat Energy DIE + Work 2nd Law: • Heat cannot flow spontaneously from a cold substance to a hot substance. Hot → Cold • Entropy (disorder) cannot spontaneously decrease. 3rd Law: If absolute 0 was attainable, all particle motion would stop. KE → 0 Entropy → 0 Practice 1) If a balloon is filled with air, sealed, and placed in the freezer, will the entropy increase, decrease, or remain constant? 2) What would happen to the motion, KE, and temperature of the air trapped in the balloon? More Practice Which is more disordered? The glass of ice chips or the glass of water?