Lab 4E, 4F & 2B
advertisement

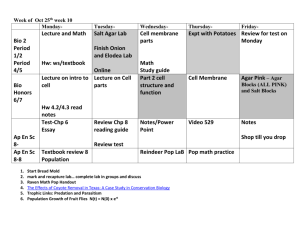
Lab 4E, 4F & 2B Media Prep. & Model Organisms Timeline/Overview Monday: Lecture Tuesday: Media Prep (4E & 4F) Pour Plates for Day 2 of Lab 2B Thursday: Media Prep (4E & 4F) Tuesday: Lab 2B—part I, II, III Wednesday: Lab 2B—part IV Thursday: Lab 2B—repeat part IV, compile data Preparation for 4E & 4F 4E & 4F Lb broth base and LB agar base Several 250ml jars Bring in extra hood 10% Bleach Prep. For Lab 2B Month Prior Check supplies: bleach, petri dishes, LB base, inoculating loops, 3 incubators, yeast, glucose, foil Order: E. coli, Aspergillus, Potato plates Week Prior prepare LB agar & broth during Lab 4E & 4F Pour plate (2 per group20) Set incubators at 30, 37 and 42 C Scalpels from other room 1 day prior make bacteria culture Media Prep. Luria Bertani (LB) Agar. Use to grow bacteria and any other microorganisms. An all purpose agar. Media Prep Equation: Mass1/Volume1 = Mass2/Volume2 Steps on Preparing Agar Plates Use a 250-mL beaker to use as a container for the agar. Pour LB Agar Base with Distilled water into the container and mix till the base is fully fused with the distilled water. Place the container on a Heat Source (i.e. microwave) for 15 seconds and every 15 seconds stir the container until the container is fully clear from debris. Repeat this step until the container is cleared. Finally, after the container is all clear from debris, place the cap of the container but, very loosely so, it can release pressure during the autoclave. Have the cap jiggle. Place the container in the autoclave from 15 to 20 minutes at 15 to 20 psi. Model Organisms Characteristics of Model Organisms: Easy to grow & maintain Easy to provide nutrients Short Generation time Well Understood Growth & Development Resembles Other Organisms Escherichia Coli (E. coli), Aspergillus & Yeast Levels of Biological Organization: Atoms Molecules Organisms Organelles Cells Tissues Organs 2B purpose To grow and study an organism in a laboratory, one should know the environmental preferences of the species. Maintaining an organism at less than optimal light intensity, temperature, or oxygen level, from example, may put the organism under stress, possibly affecting its growth or other processes. Pre-lab—Plate Pouring (4E & 4F) Pour LB agar base plates (Luria-Bertani ) E. Coli plate—use the LB agar with sterile technique, use a 10ml pipette to transfer 10ml into a sterile plate Yeast plate—use the LB* agar, sterile technique, transfer 10ml using a pippet *we are not using Malt extract agar because we do not have it, but LB agar should have enough nutrients to grow yeast Each group does 1 plate of each organism at their assigned Temperature—wrap in foil for dark 1. 2. 3. 4. 5. 6. 7. 8. 9. room room 30C 30C 37C 37C 42C 42C 8C (instead of the suggested 4C)—please do 2 sets Part I Tips Label the bottom Inoculating loop Triple Z streaking method Close lids with tape Make sure to use plate with LB agar Part II Tips Each group gets one plate instead of a tube of potato dextrose agar We are using a potato dextrose agar plate NOT tube Cut aspergillus from stock plate—will be in the hood Put the aspergillus (fungi) on the plate fungi side down to the agar and close the lid Part III Tips Label the bottom Inoculating loop Triple Z streaking method Close lids with tape Make sure to use plate with LB agar Part IV—Data Collection & Analysis Make sure to come in at FIRE on Wednesday to do a table like 2.3 (add space for 8C) We will repeat the table again on Friday in class This will provide us with more information of what happens over time as well as temperature The growth is exponential, but when nutrients run out, they go into the death phrase (smelly!), we want to try to catch them at their peak Use results from all the groups posted on the internet for you data Tutorials Media Prep. (13) LB Agar and Broth (14) Sterile technique (15) Pouring Plates (16) Starting a Broth (18) Streak Colonies (17)