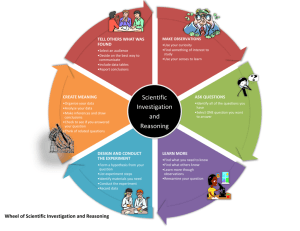
Toxins Unit Investigation IV
advertisement

Toxins Unit Investigation IV: Neutralizing Toxins Lesson 1: Heartburn Lesson 2: Watered Down Lesson 3: pHooey! Lesson 4: Proton Shuffle Lesson 5: Neutral Territory Lesson 6: Drip Drop Lesson 7: Call Poison Control Toxins Unit – Investigation IV Lesson 1: Heartburn ChemCatalyst Countless products are advertised on TV with the promise of reducing acid indigestion. • What is acid indigestion? What is acidity? • What does acid have to do with your stomach? • How do you think acid “reducers” work? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • What are some characteristics of solutions that are acidic, basic, or neutral? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Describe the differences among acidic, basic, and neutral solutions. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes • Indicators: A set of substances that respond to other substances with vivid color changes. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: This activity will introduce you to a special category of solution. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Safety note: Do not get acids and bases on your skin. In case of a spill, rinse with large amounts of water. Wear goggles. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Substance table salt (aq) Formula NaCl vinegar (aq) C2H4O2 rubbing alcohol C3H8O ammonium hydroxide (aq) distilled water NH4OH stomach acid (aq) washing soda (aq) lemon juice drain cleaner (aq) Cabbage Indicator Indicator Juice color number H2O HCl Na2CO3 C6H8O7 NaOH © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) 0 7 14 (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Observations when added to CaCO3 water stomach acid vinegar drain cleaner © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • Look for patterns in the names, chemical formulas, uses, and properties of the substances you tested. • List four characteristics of acids. • List four characteristics of bases. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes Indicators can be used to identify substances that are acidic, basic, and neutral when dissolved in water. • Acids turn cabbage juice pink and are between 0 and 7 on the universal indicator scale. • Bases turn cabbage juice green or blue and are between 7 and 14 on the universal indicator scale. • Neutral substances do not change color with cabbage juice and are at or very near 7 on the universal indicator scale. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) • pH scale: A number line from 0 to 14. The numbers are associated with indicator colors. • The numbers associated with indicator colors are also called pH numbers or simply pH. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • An unknown substance is purple with cabbage juice and does not react with calcium carbonate. Is it an acid, base or neutral substance? Explain. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • Acids and bases are different types of solutions and are classified according to their observable behavior. • Acids and bases respond differently to indicators. Depending on the indicator, different colors will form that show either an acidic or basic substance. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 2: Watered Down ChemCatalyst • Describe what you observe in the three vials. • What is one explanation for the differences you observe? • If you tested each solution with universal indicator before putting in the gummy bears, what colors would you expect to see? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • How does dilution affect the acidity or basicity of a solution? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Explain how dilution affects the concentration and pH of a solution. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: In this activity you will examine the effect of concentration on acidity and basicity of solutions. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Safety note – Do not get acids and bases on your skin. In case of a spill, rinse with large amounts of water. Wear goggles. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Data Table for dilution of 1.0 M HCl Well # Concentration 1.0 M HCl Color pH number 0.10 M HCl 0.010 M HCl 0.0010 M HCl 0.00010 M HCl 0.000010 M HCl 0.0000010 M HCl 0.00000010 M HCl 0.000000010 M HCl © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Data Table for dilution of 1.0 M NaOH Well # Concentration 1.0 M NaOH Color pH number 0.10 M NaOH 0.010 M NaOH 0.0010 M NaOH 0.00010 M NaOH 0.000010 M NaOH 0.0000010 M NaOH 0.00000010 M NaOH 0.000000010 M NaOH © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Data Table for dilution of 1.0 M NaCl Well # Concentration Color pH number 1.0 M NaCl 0.10 M NaCl 0.010 M NaCl 0.0010 M NaCl 0.00010 M NaCl 0.000010 M NaCl 0.0000010 M NaCl 0.00000010 M NaCl 0.000000010 M NaCl © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • What does concentration have to do with the acidity or basicity of a solution? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes 0 7 14 More OH– More H+ pH scale (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Arrhenius definition: • An acid is any substance that adds a hydrogen ion (H+) to the solution. • A base is any substance that adds a hydroxide ion (OH–) to the solution. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) HA H+ + A– XOH X+ + OH– © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • Lemon juice has a pH of about 2. What does this tell you about what is in the solution? • What would happen if you diluted this solution? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • The acidity and basicity of a solution are related to the concentration of the solution. Dilute solutions are less acidic and basic than more concentrated solutions. • Dilution of an acid or a base results in a solution that is increasingly neutral. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X • The pH scale is related to the concentration of H+ and OH– ions in solution. • According to Arrhenius, an acid is any substance that adds a hydrogen ion (H+) to the solution and a base is any substance that adds a hydroxide ion (OH–) to the solution. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 3: pHooey! ChemCatalyst Examine the following data: Substance 0.10 M HCl 0.010 M HCl 0.0010 M HCl 1.0 M NaOH 0.10 M NaOH 0.010 M NaOH Water pH 1.0 2.0 3.0 14.0 13.0 12.0 7.0 (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) • What do you think pH really represents? • How does the concentration of the solutions relate mathematically to the pH of the solutions? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • How does pH relate to the concentrations of acidic and basic solutions? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Find the pH of a solution if you know the concentrations of hydrogen or hydroxide ions that it contains. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: You will explore the relationship between pH, pOH, [H+], and [OH–]. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Substance Acidic pH or Basic? [H+] (in decimal) [H+] (in scientific notation) 1 M HCl Stomach acid (0.1 M HCl) Acidic Clear Soda Rain Water 3 1.0 10–6 M 7 0.0000001 M Basic 1 M NaOH 1.0 10–8 M 8 Ammonia Drain Cleaner (0.1 M NaOH) 0.0000001M Neutral Salt Water Washing Soda 0.001 M Acidic Distilled Water Alcohol 1.0 10–1 M 0.1 M 0.0000000001 M 1.0 10–10 M 13 1.0 10-14 M © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Well A HCl B C D E F G H I pH 1 2 3 4 5 6 7 7 7 [H+] 1.0 10–1 M [OH–] pOH 13 1.0 10–2 M 1.0 10–12 M 1.0 10–3 M 1.0 10-4 M 1.0 10–10 M 1.0 10–5 M 1.0 10–6 M 1.0 10–7 M 1.0 10–7 M 1.0 10–7 M 1.0 10–7 M 12 10 7 7 © 2004 Key Curriculum Press. Unit IV • Investigation IV-X NaOH Well R Q P O N M L K pH 7 7 7 8 9 10 11 12 [H+] [OH–] pOH 1.0 10–7 M 1.0 10–7 M 7 1.0 10–7 M 7 1.0 10–7 M 1.0 10–8 M 1.0 10–9 M 1.0 10–5 M 5 1.0 x10–10 M 1.0 10–11 M 3 1.0 10–12 M 1.0 10–2 M J 13 1.0 10–13 M 1.0 10–1 M 1 © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • How are pH and pOH related to each other mathematically? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes pH = 4 pH = 5 This box would show 1,000 H+ ions and 1,000 Cl– ions. Key: pH = 6 pH = 7 This box would show 100 H+ ions and 100 Cl– ions. H+ (cont.) Cl– © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) pH = –log[H+] pOH = –log[OH–] (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Sample Problem: • What is the pH of a solution with a hydrogen ion concentration of 3.4 10–4 moles/ L? (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Please be careful when entering scientific notation into your calculator— it is very easy to make a mistake. When entering 3.4 10–4, first enter “3.4”. Then press the button labeled “EXP” or “10X” or “EE”. Now enter –4, using the “+/–“ key, not the subtraction key. Never try to enter this number by pressing “X” and “10” and “–4”. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • What is the pH of a solution with [OH–] = 1.0 10–12 M? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • The relationship between pH and pOH of a solution is expressed by the following: pH + pOH = 14. • The relationship between [H+] and [OH–] in a solution is expressed by the following: – log [H+] + –log [OH–] = 14. • The pH of a solution is logarithmically related to the concentration of hydrogen ion and can be calculated mathematically using the following equation: pH = log [H+] © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 4: Proton Shuffle ChemCatalyst Besides HCl, the digestive system also produces a compound called bicarbonate, HCO3–. Bicarbonate plays a vital role in regulating the pH of the digestive system. • Do you think bicarbonate, HCO3–, is an acidic, basic or neutral substance? Explain your reasoning. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • Is there a better way to classify acids, bases, and neutral substances? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Explain the behavior of acids and bases on a molecular level. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: This activity will provide you with information to expand on your definition of acids and bases. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • The Arrhenius definition of acids and bases defines them as substances that release either H+ or OH–. How can we expand on this definition to include substances like methylamine, CH3NH2, and ammonia, NH3? (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Acids (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Bases (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Neutral Substances © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes Brønsted-Lowry definitions • An acid is a substance from which a proton can be removed. • A base is a substance that can remove a proton from another substance. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) H2O (l) H+(aq) + OH– (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) NH3 (aq) + H2O (l) NH4+ (aq) + OH– (aq) (base) (acid) HNO3 H+ + NO3– HNO3 + H2O H3O+ + NO3– (acid) (base) pH = –log[H3O+] © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • Do you predict a solution of CH4 to be acidic, basic or neutral? Explain. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • A Brønsted-Lowry acid is a substance from which a proton can be removed. • A Brønsted-Lowry base is a substance that removes a proton from an acid. • When an acid reacts with water, hydronium ion is produced. • Some substances can act as both acid and base. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 5: Neutral Territory ChemCatalyst Milk of magnesia, Mg(OH)2, can reduce excess stomach acid, HCl. • What products do you think are produced when Mg(OH)2 and HCl are mixed? • What do you think happens when you add an acid together with a base? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • How do acids and bases interact with each other? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Write balanced chemical equations describing the interactions between acids and bases. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: In this activity, you will examine the products of reactions between acids and bases. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X starting solution pH at start solution added pH after mixing acidic, basic, or neutral Did a reaction occur? Reactions with NaOH 0.10 M HCl 0.10 M NaOH 0.10 M HNO3 0.10 M NaOH solid Ca(OH)2 0.10 M NaOH Reactions with HCl 0.10 M NaOH 0.10 M HCl 0.10 M HNO3 0.10 M HCl solid Ca(OH)2 0.10 M HCl © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense Complete the following set of general equations for the reaction of HCl and NaOH with the “generic” acid HA and the “generic” base MOH. HA + HCl HA + NaOH MOH + HCl MOH + NaOH © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes • A neutralization reaction is a reaction in which an acid and a base react in aqueous solution to produce a salt and water. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) HA + MOH MA + H2O (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) 2HCl (aq) + Mg(OH)2 MgCl2 (aq) + 2 H2O (l) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • Sulfuric acid, H2SO4, reacts with magnesium hydroxide, Mg(OH)2. Write a balanced equation for the reaction that occurs. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • A neutralization reaction is a reaction between an acid and a base to produce a salt and water. • When acids are mixed with acids, the pH remains less than 7. When bases are mixed with bases, the pH remains greater than 7. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 6: Drip Drop ChemCatalyst A student mixes 100 mL of 0.10 M HCl with different volumes of 0.10 M NaOH. A. 100 mL of 0.1 M HCl + 50 mL of 0.1 M NaOH B. 100 mL of 0.1 M HCl + 100 mL of 0.1 M NaOH C. 100 mL of 0.1 M HCl + 150 mL of 0.1 M NaOH (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) • Which solution is the least toxic? • Are the final solutions acid, basic, or neutral? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • How can the interactions of acids and bases be used to determine the concentration of a particular acidic or basic solution? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Use the experimental method known as “titration” to determine the unknown concentration of an acid or base. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: This activity will introduce you to a laboratory procedure used to determine the concentration of an acid or a base. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) HCl solution Drops of 0.10 M Calculated NaOH added to concentration neutralize of HCl 20 drops HCl Solution A 20 drops HCl Solution B 20 drops HCl Solution C (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Initial volume of HCl 1.0 L Volume of 0.10 M NaOH added 1.0 L 100 mL 200 mL 50 mL 200 mL Total moles of NaOH 0.10 moles 0.020 moles 0.0025 moles 100 mL Total Initial HCl moles of concentrat HCl ion 0.10 moles 0.020 moles 0.10 M 0.050 M 73 mL © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • Describe how you determined the concentration of the NaOH solution in Problem 6. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Notes • A titration is a method for determining the concentration of an acid or base solution by reacting a known volume of the solution with a solution of known concentration. When the moles of H+ are equal to the moles of OH– in the solution, the reaction has reached the equivalence point of the titration, sometimes referred to as the endpoint. (cont.) © 2004 Key Curriculum Press. Unit IV • Investigation IV-X (cont.) Example: • If 25.00 mL of 0.50 M nitric acid, HNO3, solution are required to titrate 62.00 mL of sodium hydroxide, NaOH, what is the molarity of the NaOH? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In You place 50 drops of HCl in a beaker along with a drop of phenolphthalein indicator. After you have added 100 drops of 0.10 M NaOH, the color is a very faint pink. • What is the concentration of the HCl solution? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • A titration is a procedure that allows you to calculate the concentration of an unknown acid or base using a neutralization reaction. • During a titration, when the moles of H+ are equal to the moles of OH– in the solution, the reaction has reached the equivalence point of the titration, sometimes referred to as the endpoint. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Toxins Unit – Investigation IV Lesson 7: Call Poison Control ChemCatalyst Imagine that your little brother spilled a toxic substance on his skin. You decide to call Poison Control: 1 (800) 222–1222. • What are some things that you might want to know about the substance? • What kinds of questions do you think Poison Control will ask you? What might they tell you to do? Explain your thinking. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X The Big Question • How do solubility and pH contribute to the toxicity of a substance? © 2004 Key Curriculum Press. Unit IV • Investigation IV-X You will be able to: Use the mole concept and balanced chemical equations to calculate the mass relationships between products and reactants and determine the concentrations or pH of solutions in which the reactions are a part. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Activity Purpose: The purpose of this worksheet is to review some of the key ideas in the Toxins Unit. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Making Sense • No Making Sense exercises. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Check-In • No Check-In exercise. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X Wrap-Up • No Wrap-Up points. © 2004 Key Curriculum Press. Unit IV • Investigation IV-X