Slide
advertisement
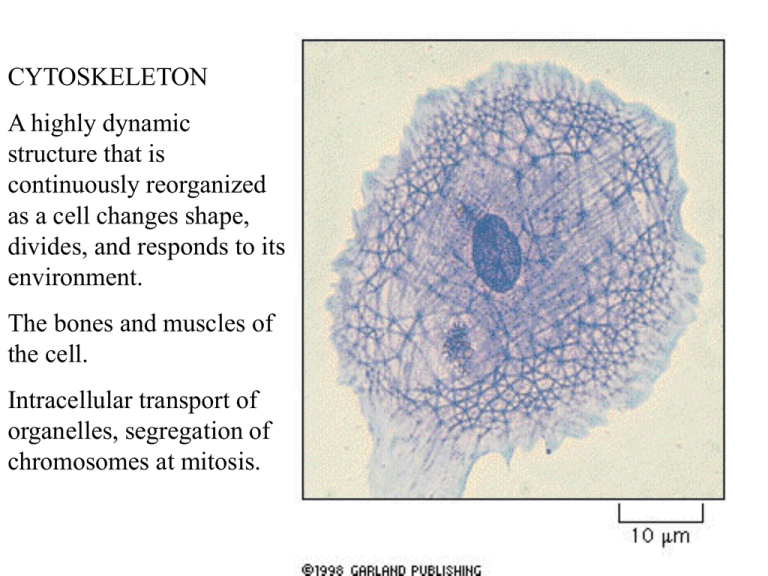
CYTOSKELETON A highly dynamic structure that is continuously reorganized as a cell changes shape, divides, and responds to its environment. The bones and muscles of the cell. Intracellular transport of organelles, segregation of chromosomes at mitosis. 10 nm static 25nm dynamic * * 7nm * dynamic INTERMEDIATE FILAMENTS enable the cell to withstand mechanical stress. These are the toughest, most durable and are found in the cytoplasm of most animal cells forming a network found in the nucleus (nuclear lamina) and throughout the cytoplasm. Epidermal cells - network indirectly connected to neighbors through desmosomes Amino terminal globular head Carboxyl-terminal globular tail tail Form stable dimers Globular head and tail are exposed on the surface, interact with other components of the cytoplasm, and vary greatly in size and amino acid sequence. The central rod domains are all similar in size and amino acid sequence and pack together to form filaments of similar diameter Diverse - gut, skin, hair, feathers, claws formed from a mixture of different keratin subunits directly connect through cell-cell junctions like desmosomes epidermolysis bullosa simplex mutation in keratine Intermediate filaments strengthen animal cells and are prominent in cells subject to mechanical stress, like nerve cell axons, muscle cells, epithelial cells. Nuclear lamina - lines the inner face of the nucleus and provides attachment sites for DNA-containing chromatin. Composed of lamins. MICROTUBULES DYNAMIC usually growing out of a Microtubule Organizing Center Extending out into the cell periphery, they create a system of tracks along which vesicles, organelles etc can be moved. STRUCTURE OF MICROTUBULE Tubulin is a dimer cylinder made of 13 parallel protofilaments has structural polarity with the alpha subunit exposed at one end the minus end and the beta subunit exposed at the other end, the plus end. Grows by adding individual tubulin dimers Microtubules are maintained by a balance of assembly and disassembly. Cell contains half microbubules (which are continually graowing and shrinking) and half free tubulin Colchicine binds tightly to free tubulin and prevents its polymerization. Taxol binds tightly to microtubules and prevents them from losing subunits. Same overall result - arrests dividing cells in mitosis and eventually kills dividing cells. Polymerization of tubulin on a centrosome, the major microtubule-organizing center in animal cells. Centrosomes contain hundreds of rin-shaped structures formed of gamma tubulin. This serves as the starting point - nucleation site. It adds alph/beta tubulin in a specific orientation, with the minus end embedded in the centrosome - growth occurs at the plus end. Not centrioles function unknown GTP hydrolysis is thought to control the growth of microtubules: leading to dynamic instability: each free tubulin dimer contains one tightly bound GTP molecule that is hydrolyzed to GDP shortly after it is added to the growing chain. When polymerization occurs more rapidly (lots of tubulin) it grows, when GTP is hydrolyzed more quickly, it shrinks Once started, it will continue GTP cap prevents depolymerization What is the function of this continual shooting out of new microbubules and retracting them? “Reaching” out. Once the microtubule establishes contact with another rmolecule or cell structure it will establish a relatively stable link - a road that links this structure to the centrosome. The result is a dynamic, but highly organized system of microtubules linking selected parts of the cell. This is used to position organelles and move vesicles. This allows cells to modify the dynamic instability for particular purposes - mitosis. And to fix a structure in a differentiated cell - as in the axons of neurons, in which proteins bind to the ends and along their length to stabilize them - maintaining the organization of the cell Differentiated cells are polorized - ex. dendrites,cell body, axon Movement along the microtubule is faster and directed. Intracellular organelle movement - taken with less than a minute between 1st and last frame Both actin filaments and microbubules are involved in saltatory movement - start/stop jerky • Accessory proteins bind to microtubules and serve various functions – stabilize microtubules against disassembly – coordinate and interact with other cytoskeleton components – distribute membranes • Motor proteins use energy of ATP hydrolysis to transport organelles, vesicles, etc. along tracks provided by actin filaments and microtubules Motor protein Microtubule motor proteins Kinesins and dyneins generally move in opposite directions. Heads are ATPhydrolyzing enzymes Organelles move along microtubules in a directed fashion. Kinesins move toward the plus end and dyneins move toward the minus end. Both exist in many forms, each thought to transport different “cargo”. The tail of the motor protein determines the cargo. Both the endoplasmic reticulum and the golgi apparatus depend on microtubules for their alignment and positioning. As the cell develops and the ER grows, kinesins attached to the outside of the ER membrane pull it outward along microtubules. Dyneins pull the golgi the other way. When cells are treated with colchicine, microtubules disassemble and ER collapses to the center of the cell, while the golgi fragments into small vesicles. Golgi yellow ER - blue Cilia of the human respiratory tract sweep layers of mucus with trapped dust etc Cilia contain a core of stable microtubules arranged in a bundle which grow from a basal body. Are covered in the plasma membrane Figure 8C. Move in a whiplike motion Flagella move in a repetitive wavelike motion as in the flagella of sperm or this green alga “9 + 2” array of cilia and flagella in all eucaryotic cells. Microtubule-associated proteins serve as cross-links too hold the bundle together and others generate the force to bend. Ciliary dynein resembles cytoplasmic dynein. Attached by its tail to one microtubule, its heads interact with adjacent microtubules, sliding between the two - generating the force needed to bend. Because the two microtubules are linked, they bend rather than slide. ACTIN FILAMENTS: found in all eucaryotic cells. Depending on their association with different proteins, actin filaments various permanent structures such as (A) microvilli on brush-border cells lining the intestine, (B) small contractile bundles in the cytoplasm that act like the “muscles” of the cell, (C) temporary structures like the filopodia (pseudopodia) of a crawling fibroblast, or (D) the contractile ring that pinches the cytoplasm in two when an animal cell divides. Which structures are formed is determined by the actin-binding proteins present in the cell at this time. Found as crosslinked bundles and networks of actin filaments Actin filaments are twisted chains of identical globular actin molecules, all pointing the same way - polar again. They are thinner, more flexible, and usually shorter than microtubules. There are many more actin filaments in a cell than microtubules Actin and tubulin polymerize by similar mechanisms. Actin filaments grow from both ends, but plus end is faster. Actin filament is also inherently unstable, disassembly at both ends. ATP is hydrolyzed instead of GTP, and this promotes depolymerization. The ability to assemble and disassemble is also required for actin filaments - toxins that prevent polymerization or depolymerization freeze cell movements like crawling. Regulate actin polymerization Major classes o f actin-bindng proteins control behavior of filaments Convert an actin gel into a more fluid state Hold actin filaments together in a gel-like meshwork within the cell cortex - just beneath the plasma membrane And microvilla Form contractile bundles as in muscle cells, serve as tracks along which motor proteins transport organells. The plasma membrane must be strengthened by the cell cortex. This is a framework of proteins attached to the membrane via transmembrane proteins.The shape and mechanical properties of the plasma membrane is determined by a meshwork of fibrous proteins - the cell cortex. Red blood cells are very simple, allowing study of the cell cortex in simple form. Genetic abnormalities in spectrin structure result in anemia, spherical, fragile rbcs The spectrin-based cell cortex of human red blood cells. Much simpler than other cells. Actin filaments in most cells is more concentrated, linked by actin- binding proteins into a meshwork gel that gives the membrane shape and mechanical strength. The cortex provides the molecular basis for shape changes and locomotion • Cells must be able to move toward food, along a tract (axon of a new neuron), toward an infection (neutrophiles). This requires actin, and entails coordinated changes of many molecules in different regions of the cell. Three interelated processes (all which require actin) known to be essential include. – 1. The cell pushes out protrusions at its leading edge – 2. these protrusions adhere to the surface the cell is crawling over – 3. the rest of the cell drags itself forward on molecules which anchor it. First step, pushing the cytoplasm forward, is driven by actin polymerization. A human fibroblast Both filopodia and the larger lamellipodia are exploratory, motile structures that form and retract with great speed. Both are generated by rapid local growth of actin filaments which are nucleated at the plasm membrane and push it out without tearing it Growth of filopodia: A nucleation complex at the plasma membrane (like the centrosome) organizes growth of actin filaments. Difference between actin and microtubules = nucleation complex is at the + or growing end. Step 2. Transmembrane proteins including integrins which bind to the extracellular matrix or other cells capture these areas of nucleation. Dark staining shows areas of close contact between the glass and the fibroblast. Here, by staining actin, it is shown that actin filaments terminate at or close to sites of contact. Therefore, external adhesion molecules and internal actin filaments are working together in close contact. Step 3. Cell moves forward. Exact mechanism is unknown. But release of adhesion molecules (integrins) from the substratum (surface the cell is moving over) and contraction of the cell cortex may Step 3 do the trick. Step 1 Step 2 Adhesion molecules associated with actin filaments. MYOSINS: All actin-dependent motor proteins belong to the mysoin family, provide energy by hydrolyzing ATP, and move along actin from the minus end to the plus end. Studied in skeletal muscle but found in other cells contractile bundles, contractile ring of the dividing cell. The heads hydrolyze ATP and change shape to bind, detach and rebind. Moving along the actin filament. The tails of Myosin II can associate with each other to form mysoin II filaments. The tail varies and determines which cell components will be dragged along The head interacts with actin filaments and contains the ATP hydrolyzing activity dimer Coiled-coil tail ACTIN FILAMENTS: found in all eucaryotic cells. Depending on their association with different proteins, actin filaments various permanent structures such as (A) microvilli on brush-border cells lining the intestine, (B) small contractile bundles in the cytoplasm that act like the “muscles” of the cell, (C) temporary structures like the filopodia (pseudopodia) of a crawling fibroblast, or (D) the contractile ring that pinches the cytoplasm in two when an animal cell divides. Which structures are formed is determined by the actin-binding proteins present in the cell at this time. Found as crosslinked bundles and networks of actin filaments Myosin II has two heads pointing in the opposite direction. One binds to actin filaments in one orientation and moves them that way. The other binds an actin filament in the opposite direction. SOME ROLES OF MYOSIN-1 AND MYOSIN-2 Skeletal muscle cell: huge multinucleated cells also called muscle fibers contain numerous myofibrils. During muscle contraction actin filaments slide against myosin filaments. Cardiac muscle and smooth muscle (gut etc) have a different structure, but use actin and myosin in a similar way to contract. A) What properties of intermediate filament monomers distinguish them from the monomers that make up actin filaments or microtubules? A. They bind to each other covalently. B. They are fibrous rather than globular proteins. C. They do not bind and hydrolyze nucleotides. D. There are numerous different types in different cell types. E. They are glycosylated. (B) Which of the above differences makes intermediate filaments less dynamic structures than actin filaments or microtubules? (





