Duel - Red Hook Central School District
advertisement

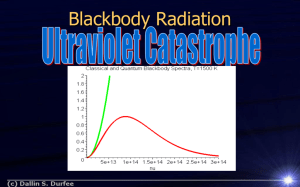
Quantum Nature of Matter & Energy • How is light produced? • Vibration of charged particles. • Is light a wave or a particle? Cite 2 pieces of evidence for light as a wave. • Diffraction • Interference What is the evidence for light as transverse. • Polarization • Max Planck studied how blackbodies emit radiation (EM waves). • A blackbody is an object that absorbs & emits all wavelengths of radiation. It does not reflect any light incident on it. • It was known that energy of a wave is related to its amplitude - Intensity. • As T goes up, total light emitted (I) goes up. • Frequency goes up too! Radiation & Temperature Hot Objects Emit Waves T vs. Intensity of a blackbody Spectrum of wavelengths emitted, but more in certain range. Classical Physics predicts E (I) becomes infinite as l approaches 0. As T , molc. vibration. Planck proposed that the E is emitted by vibrating molecules is quantized—E could only take on certain values. Energy come in little "chunks" of the frequency multiplied by a constant now called Planck's constant, h: Matter absorbs & emits energy in discrete units called quanta or photons. Plank’s formula gives the amount of energy based on the frequency of waves. E = hf. h is Plank’s constant 6.63 x 10-34 Js. E is energy in Joules f is frequency of radiation 1 eV = 1.6 x 10-19 J • Emitted E proportional to frequency. • Proportionality Constant = h. • Planck’s theory’s predictions agreed with experimental observations. Ex 1. Each photon of a certain color light has an energy of 2.5 eV. What is the frequency of and color of the light? Solution: E = hf f = E/h convert eV to Joules. (2.5 eV)(1.6 x 10-19J/eV) = 6 x 1014 s-1 or Hz 6.626 x 10-34 J s When you need to find # joules in a wave/photon use E = hf. Or E = hc/l. 2. What is the energy of light at 686 nm in Joules and eV? • • • • E = hf E = h c/l. 2.9 x 10-19 J 1.81 eV The fact that absorbed/radiated EM waves come in discrete packets indicated E is made of particles. • More evidence for the particle nature of light was later to come from work with the photoelectric effect. Photoelectric Effect It's been known (previously) that when EM waves shine on a metal surface, the surface can emit e-. You can start a current in a circuit just by shining a light on a metal plate. Materials that emit e- in this way are called photoemissive. The ejected e- are called photoelectrons. Predictions Classical Photon • EM waves absorbed by metal shake e- in metal until they overcome bond E and escape. • Longer exposure time needed to eject e-. • Higher A or f, will decrease needed exposure time. • Higher A will increase E of ejected e-. • 1 on 1 collisions btw photon & e- cause ejection. • Photon must have enough E to free e-. • No waiting time required. • Since E ~ f, there is a cut off f, below it, no e- ejected no matter how long the wait. • High A, I = more photons, more photoelectrons. Observations • • • • • • Cut off f, yes Minimum fo, need to eject e- (threshold f) Wo, F. No time delay Hi A, I (bright) = more e-. Higher f, faster e-. Easy Quantitative • • • • • • If F for a metal = 7eV. Red light has 6eV. What happens? No e- ejected. Blue light has 10 eV. What happens? e- ejected but 3 extra eV E. Could get lost in collisions in metal, but could be KE of e-. Called KE max. Example: 1. In the photoelectric effect, the following observations may be made. • I. The kinetic energy of the emitted electrons increases with increasing light frequency. • II. The electrons are emitted without time delay. Which of these observations, if any, can be explained in terms of the wave theory of light? • A. Neither I nor II • B. I and II • C. I only • D. II only 2. Photoelectron emission only occurs if the light incident on a metal surface is • • • • A. B. C. D. coherent. above a certain minimum intensity. below a certain minimum frequency. below a certain minimum wavelength. 3. Monochromatic light is incident on a metal surface and electrons are released. The intensity of the incident light is increased. What changes, if any, occur in the rate of emission of electrons and the KE of the emitted electrons? Rate of emission of e- KE of the emitted e- A. Increase increase B. decrease no change C. decrease increase D. increase no change Youtube can discharge • https://www.youtube.com/watch?v=WO38q VDGgqw Photoelectric Equation • Photon E in = electron E out. • hf = F + KE max. • hc/l = F + KE max. Ex. What minimum f, is needed to eject e- if F of metal is 4.3 x 10-19 J? • • • • hf = F + KE max. hf = F (6.63x10-34) f = (4.3 x 10-19 J) 6.5 x 1014 Hz. • Called threshold freq = fo. Ex: UV light l = 285 nm, falls on a metal surface. The maximum KE of photoelectrons is 1.40 eV. What is the work function of the metal? • • • • • • hc/l = F + KE max. (6.63x10-34)(3x108) = F + 1.40 eV. 285 x 10-9 m Change left side to eV (divide by e = 1.6 x 10-19) 4.36 eV = F + 1.40 eV. 3.06 eV = F . The Experiment and Associated Graph Stopping Potential Milliken’s experiment measured the KE of photo e- by applying a stopping potential – a voltage that brought the velocity of e- to 0. How to measure the values? Use stopping/retarding voltage until no photocurrent. Phet • • • PE gained = KE lost by electron. qV = KE max. eV = KE max. • http://www.walter-fendt.de/ph6en/ • http://phet.colorado.edu/en/simulation/photoelectric IB Data Booklet Work function. Stopping Potential. Graphing E vs. f. • hf = F + KE max. • Rearrange to solve for KE max. • KE max. = hf • eV (st pot) = hf - F. hfo . • Linear Eq. KE Y axis, h = slope, F = Y intercept. • Metals have work functions a few eV. • Energy in J/e = energy in eV. Photoelectric Experiment Applet create graph • http://www.walter-fendt.de/ph6en/ • eV = hf hfo . • Slope is Planck’s constant h/ e (1.6 x 10-19). Ex 1: Ultra-violet light is shone on a zinc surface and photoelectrons are emitted. The sketch graph shows how the stopping potential Vs varies with frequency f. Planck’s constant may be determined from the charge of an electron e multiplied by • A. the x-intercept. • B. the y-intercept. • C. the gradient. • D. the area under the graph. KE max = hf - F. As f of EM wave increases, KE increases, slope = h. F min E J, needed to eject e. Ex 2: Light of frequency f is incident on a metal surface. The work function of the metal is φ. Which of the following is the maximum kinetic energy of the electrons emitted from the surface? A. hf – B. h ( f ) e C. – hf D. h ( f ) e 3. Which of the following is a correct statement associated with the photoelectric effect? • A. Electron emission is instantaneous. • B. Electrons are only emitted if the incident light is above a certain minimum wavelength. • C. The energy of the emitted electrons depends on the light intensity. • D. The energy of the emitted electrons does not depend on the frequency of the incident light. Summery • Can measure KEmax using stopping V needed to bring fastest e- to stop. • Energy of e- is PEelc qV, for the retarding V. • Graph of V vs. f, the Y intercept is the Wo, X intercept = fo, and slope is h/e. • Graph of KE vs. f, slope = h. Hist of Quantum pt 1 British 15 min Max Planck and E= hf. http://www.youtube.com/watch?v=zB TbqOgdfEY IB Set 3 Photoelectric Questions. Hwk. Hamper pg 261 #2,3 Write out all. http://www.youtube.com/watch? v=B7pACq_xWyw&feature=rela ted Watch this. Good summery of light. 9:45 min. Hwk Holt Photo Elect and E = hf. 23 – 1 and 23 – 2 Do page 833 all and page 856 # 2, 4, 7, 10, 11 Models of The Atom Rutherford Equivalence of Mass & Energy Einstein realized that matter contains energy. There is an equivalence of mass & energy. Energy is stored in the nucleus of atoms. The energy stored any mass obeys Einstein’s equation: E = energy in J. E = mc2. m = mass kg c = vel of light Ex 2: How much energy is produced when 2.5 kg of matter are completely converted to energy? How much energy is that in eV? E = mc2. =(2.5 kg )(3x108 m/s)2. = 2.25 x 1017 J in eV (2.25 x 1017 J)(1 eV / 1.6 x 10 –19 J) = 1.4 x 1036 eV. Atomic Mass Units: amu or u Mass of atoms very small so they are measured in amu or u. Since mass is equivalent to energy, 1 amu = 931 MeV or 931 x 106 eV. Ex 3: One universal atomic mass unit is equivalent to an energy of 931 MeV. Calculate the mass in kg of one universal mass unit. Hint: Use E = mc2 where energy is known in eV. Don’t forget to convert MeV to eV. (1 u) x (931 MeV/u) x (106eV/MeV) x (1.6 x 10 –19 J / eV) = 1.49 x 1010 J E = mc2 so m = E/c2. (1.49 x 1010 J) / (3x108 m/s)2 = 1.66 x 10 –27 kg The mass units are based on 1 the mass of a proton or H. (A hydrogen nucleus) Particle Properties of Waves extend to conservation of energy and momentum. Photons may give up all or part of their energy in collisions, but the sum of the momentums and energy before must equal the sum after. Compton Effect If light behaves like a particle, then a collision btw photon & e- should be similar to billiard balls colliding. Photons must have momentum (p), & energy. In collision of photons with particles (like e-), conservation of energy & conservation of momentum apply. If the photon gives only part of its energy & momentum to an e-, its momentum decreases after the collision by the same amount as absorbed by the electron. Therefore, the frequency or energy of the photon decreases. The wavelength increases. pbefore = pafter. E photon before = KEelc after. + E photon after hfi = KEelc after + hff photon after pphoton = hf/c = h/l. The wavelength of the photon increases after collision. Matter has wave-like properties. 1924 Louis DeBroglie suggested that since waves had particle properties, matter might have wave properties. It turns out that matter does have wave properties which are inversely related to the momentum of the particle. For matter: l =h/p or l = h/mv. Since the mass of most objects is so large, the wavelengths would be very small & not measurable. Electrons, however, do show diffraction & other wave characteristics.