Stereoisomers
advertisement

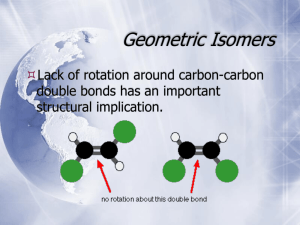
STEREOISOMERS Nabila Al-Jaber Associate Professor of Chemistry Isomers isomers are different compounds that have the same molecular formula. ISOMERS Different compounds with the same molecular formula Each isomer could have stereoisomers STEREOISOMERS Isomers with the same order of attachment, but a different configuration (3D arrangement) of groups on one or more of the atoms double bond or ring (geometric) cis/trans ISOMERS with a ring both can apply CONSTITUTIONAL ISOMERS Isomers with a different order of attachment of the atoms in their molecules ENANTIOMERS Stereoisomers whose molecules are nonsuperimposible mirror images of each other DIASTEREOMERS Stereoisomers whose molecules are not mirror images of each other TYPES OF ISOMERISM Isomerism Structural isomerism Chain Ex. N. and isobutane Steroisomerism Position Functionl Ex. Ortho, meta, para Ex.ethanol, diethylether Optical Isomerism Geometrical Isomerism(cis , trans isomers) Relationship of Constitutional and Stereoisomers Relationships of Stereoisomers constitutional isomers C4H8 hydrocarbons Structural isomers. * A fifth possible isomer of formula C4H8 is CH3CH=CHCH3 . * This would be named 2-butene according to the IUPAC rules. * These isomers may be isolated as distinct compounds, having characteristic and different properties . stereoisomers. * The only difference being the relative • orientation or configuration of the two methyl groups . * They are shown here with the designations • cis and trans. Configurational Stereoisomers of Alkenes Configurational Stereoisomers of Alkenes The Sequence Rule for Assignment of Alkene Configurations • Assign priorities to double bond substituents by looking at the atoms attached directly to the double bond carbons. 1. The higher the atomic number of the • immediate substituent atom, the higher the priority. For example,H– < C– < N– < O– < Cl–. • (priority increases left to right) (Different isotopes of the same element are • assigned a priority according to their atomic mass.) 2. If two substituents have the same – immediate substituent atom, move to the next atom (away from the double bond) until a difference is found. For example, CH3– < C2H5– – < ClCH2– < BrCH2– < CH3O–. Cycloalkane Stereoisomers Configurational Stereoisomers of Cycloalkanes Rings Conformations Some Conformations of Cyclohexane Rings Conformational Energy Profile of Cyclohexane TC = twist chair B = boat TB = twist boat C = chair Conformational Structures of Disubstituted Cyclohexanes 1,1-dimethylcyclohexane 1-t-butyl-1-methylcyclohexane cis-1,2-dimethylcyclohexane trans-1,2-dimethylcyclohexane cis-1,3-dimethylcyclohexane trans-1,3-dimethylcyclohexane cis-1,4-dimethylcyclohexane trans-1,4-dimethylcyclohexane Ethane Conformations Name of Conformer Wedge-Hatched Bond Structure Sawhorse Structure Newman Projection Conformational Energy Profile of Ethane Potential Energy Profile for Ethane Conformers Dihedral Angle Bond Repulsions in Ethane Conformational Isomerism Extended Chain Coiled Chain – Four Conformers of Butane Potential Energy Profile for Butane Conformers Potential Energy Profile for Butane Conformers summarize some important aspects of conformational stereoisomerism at this time. summarize some important aspects of conformational stereoisomerism at this time. (i) Most conformational interconversions in simple molecules occur rapidly at room temperature. Consequently, isolation of pure conformers is usually not possible. summarize some important aspects of conformational stereoisomerism at this time. (ii) Specific conformers require special nomenclature terms such as staggered, eclipsed, gauche and anti when they are designated. summarize some important aspects of conformational stereoisomerism at this time. • (iii) Specific conformers may also be designated by dihedral angles. In the butane conformers shown above, the dihedral angles formed by the two methyl groups about the central double bond are: A 180º, B 120º, C 60º & D 0º. summarize some important aspects of conformational stereoisomerism at this time. (iv) Staggered conformations about carbon-carbon single bonds are more stable (have a lower potential energy) than the corresponding eclipsed conformations. The higher energy of eclipsed bonds is known as eclipsing strain. summarize some important aspects of conformational stereoisomerism at this time. (v) In butane the gauche-conformer is less stable than the anticonformer by about 0.9 kcal/mol. This is due to a crowding of the two methyl groups in the gauche structure, and is called steric strain or steric hindrance. summarize some important aspects of conformational stereoisomerism at this time. • (vi) Butane conformers B and C have non-identical mirror image structures in which the clockwise dihedral angles are 300º & 240º respectively. These pairs are energetically the same, and have not been distinguished in the potential energy diagram shown here.