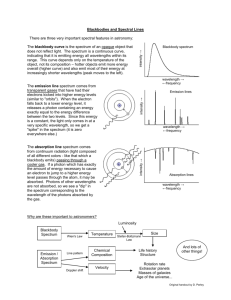
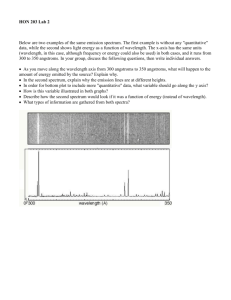
Spectroscopy - Chabot College
advertisement

WHAT DO YOU THINK? 1. 2. 3. Which is hotter, a “red-hot” or a “blue-hot” object? What color does the Sun emit most brightly? How can we determine the age of space debris found on Earth? In this chapter you will discover… properties of electromagnetic radiation related to TEMPERATURE, COMPOSITION, & MOTION the structure of atoms stars with different surface temperatures emit different intensities of electromagnetic radiation astronomers determine chemical compositions of stars and interstellar clouds by studying the wavelengths of electromagnetic radiation that they absorb or emit The importance of the DOPPLER shift to indicate motion Different COLOR indicates different temperature Properties of Thermal Radiation 1. Radiation is emitted in ALL wavelengths without breaks. “Continuous Spectrum” Properties of Thermal Radiation 2. Hotter objects emit much, much, much, MUCH more light at all frequencies per unit area. 3. Hotter objects emit bluer photons with a higher average energy. What are these? Absorption Spectrum of Sun Emission Line spectrum of Iron in laboratory on Earth Absorption Spectrum of Hydrogen Gas Emission Spectrum of Hydrogen Gas Spectral “Fingerprints” Absorption Emission Three TYPES of Spectra! Continuous Spectrum The spectrum of a common (incandescent) light bulb spans all visible wavelengths, without interruption. Emission Line Spectrum A thin or low-density cloud of gas emits light only at specific wavelengths that depend on its composition and temperature, producing a spectrum with bright emission lines. Absorption Line Spectrum A cloud of gas between us and a light bulb can absorb light of specific wavelengths, leaving dark absorption lines in the spectrum. What are the three basic types of spectra? Continuous Spectrum Emission Line Spectrum Absorption Line Spectrum Spectra of astrophysical objects are usually combinations of these three basic types. Chemical Fingerprints Energy levels of hydrogen Each type of atom has a unique set of energy levels. Each transition corresponds to a unique photon energy, frequency, and wavelength. Why Spectral Lines??? Atoms have nuclei (protons & neutrons) & electrons in specific energy level patterns Each element has different NUMBERS of electrons, in different energy level patterns Transitions of electrons absorbing or emitting energy produce different wavelengths of light we see. Simple “Model” of Atom Electron Energy Levels Nucleus Chemical Fingerprints Electron Transitions to LOWER energy states produce a unique pattern of emission lines. Chemical Fingerprints Atoms can also absorb photons with same energies. Transitions to higher energy states produce a pattern of absorption lines at the same wavelengths. Spectroscopy works at ALL wavelengths! Examples of Spectra! Emission Spectra from Hydrogen & Oxygen Thought Question Which letter(s) labels absorption lines? A B C D E Thought Question Which letter(s) labels absorption lines? A B C D E Thought Question Which letter(s) labels the peak (greatest intensity) of infrared light? A B C D E Thought Question Which letter(s) labels the peak (greatest intensity) of infrared light? A B C D E Thought Question Which letter(s) labels emission lines? A B C D E Thought Question Which letter(s) labels emission lines? A B C D E Interpreting an Actual Spectrum By carefully studying the features in a spectrum, we can learn a great deal about the object that created it. What is this object? Reflected Sunlight: Continuous spectrum of visible light is like the Sun’s except that some of the blue light has been absorbed—object must look red What is this object? Thermal Radiation: Infrared spectrum peaks at a wavelength corresponding to a temperature of 225 K What is this object? Carbon Dioxide: Absorption lines are the fingerprint of CO2 in the atmosphere What is this object? Ultraviolet Emission Lines: Indicate a hot upper atmosphere What is this object? Mars! How does light tell us the speed of a distant object? The Doppler Effect Doppler shift tells us ONLY about the part of an object’s motion toward or away from us. Measuring the Shift Stationary Moving Away Away Faster Moving Toward Toward Faster We generally measure the Doppler effect from shifts in the wavelengths of spectral lines. Thought Question I measure a line in the lab at 500.7 nm. The same line in a star has wavelength 502.8 nm. What can I say about this star? • • • It is moving away from me. It is moving toward me. It has unusually long spectral lines. Thought Question I measure a line in the lab at 500.7 nm. The same line in a star has wavelength 502.8 nm. What can I say about this star? • • • It is moving away from me. It is moving toward me. It has unusually long spectral lines. Summary of Key Ideas By studying the wavelengths of electromagnetic radiation emitted and absorbed by an astronomical object, astronomers can learn about its temperature, chemical composition, rotation rate, companion objects, and movement through space. Blackbody Radiation A blackbody is a hypothetical object that perfectly absorbs electromagnetic radiation at all wavelengths. The relative intensities of radiation that it emits at different wavelengths depend only on its temperature. Stars closely approximate blackbodies. Wien’s law states that the peak wavelength of radiation emitted by a blackbody is inversely proportional to its temperature—the higher its temperature, the shorter the peak wavelength. The intensities of radiation emitted at various wavelengths by a blackbody at a given temperature are shown as a blackbody curve. Blackbody Radiation The Stefan-Boltzmann law shows that a hotter blackbody emits more radiation at every wavelength than does a cooler blackbody. The motion of an object toward or away from an observer causes the observer to see all of the colors from the object to blueshift or redshift, respectively. This is generically called a Doppler shift. Discovering Spectra Spectroscopy—the study of electromagnetic spectra— provides important information about the chemical composition of remote astronomical objects. Kirchhoff’s three laws of spectral analysis describe the conditions under which absorption lines, emission lines, and a continuous spectrum can be observed. Spectral lines serve as distinctive “fingerprints” that identify the chemical elements and compounds comprising a light source. Atoms and Spectra An atom consists of a small, dense nucleus (composed of protons and neutrons) surrounded by electrons. Atoms of different elements have different numbers of protons, while different isotopes have different numbers of neutrons. Quantum mechanics describes the behavior of particles and shows that electrons can only be in certain allowed orbits around the nucleus. The nuclei of some atoms are stable, while others (radioactive ones) spontaneously split into pieces. The spectral lines of a particular element correspond to the various electron transitions between allowed orbits of that element with different energy levels. When an electron shifts from one energy level to another, a photon of the appropriate energy (and hence a specific wavelength) is absorbed or emitted by the atom. Atoms and Spectra The spectrum of hydrogen at visible wavelengths consists of part of the Balmer series, which arises from electron transitions between the second energy level of the hydrogen atom and higher levels. Every different element, isotope, and molecule has a different set of spectral lines. When an atom loses or gains one or more electrons it is said to be charged. The atom loses an electron when the electron absorbs a sufficiently energetic photon, which rips it away from the nucleus. The equation that describes the Doppler effect states that the size of a wavelength shift is proportional to the radial velocity between the light source and the observer. Key Terms absorption line absorption line spectrum atom atomic number blackbody blackbody curve blueshift continuous spectrum diffraction grating Doppler shift electromagnetic force electron element emission line emission line spectrum energy flux excited state ground state ion ionization isotope Kirchhoff’s laws luminosity molecule neutron nucleus (of an atom) periodic table Planck’s law proper motion proton quantum mechanics radial velocity radioactive redshift spectral analysis spectrograph spectroscope Stefan-Boltzmann law strong nuclear force transition (of an electron) transverse velocity weak nuclear force Wien’s law WHAT DID YOU THINK? Which is hotter, a “red-hot” or a “blue-hot” object? Of all objects that glow visibly from heat generated inside them, those that glow red are the coolest. WHAT DID YOU THINK? What color does the Sun emit most brightly? The Sun emits all wavelengths of electromagnetic radiation. The colors it emits most intensely are in the blue-green part of the spectrum. Because the human eye is less sensitive to blue-green than to yellow, and Earth’s atmosphere scatters blue-green wavelengths more readily than longer wavelengths, we normally see the Sun as yellow. WHAT DID YOU THINK? How can we determine the age of space debris found on Earth? We measure how much the long-lived radioactive elements, such as 238U, have decayed in the object. Carbon dating is only reliable for things that formed within the past 100,000 years. It cannot be used for determining the age of debris found on Earth, which was all formed more than 4.5 billion years ago.