Metabolic Processes
advertisement

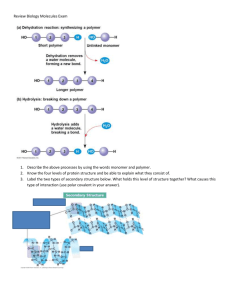
Carbohydrates Carbohydrate Linguistics ‘Carbo’ for carbon, ‘hydrate’ for water. Empirical Formula is (CH2O)n where n is the number of carbon atoms and is ≥ 3. Also called ‘saccharides’ after the Greek word for sugar. Carbohydrate Uses by the Body Used for: Cell to cell signaling when attached to cell membranes Building blocks for larger molecules Energy source Monosaccharide Chemistry AKA the ‘Simple Sugars’, mono- for 1 and saccharide for sugar. Carbohydrates are either aldehydes or ketones depending on which carbonyl group they possess. They can be classified based on the number of carbon atoms they have and the suffix ‘-ose’ Hexoses have 6 Carbons… The carbon atoms are numbered. * Common Monosaccharides Structural Formulae Monosaccharides exist as straight chain molecules, however when there are 5 or more carbons, the monosaccharide can also form a ring structure, when dissolved in water. Ring structures are usually more conducive to chemical reactions/interactions. Hexoses Glucose, Galactose and Fructose are all isomers of one another, meaning they have the same chemical formula, but a different structural formula (pages 29-30 in your textbook!). As with all isomers, these three sugars possess different chemical and physical properties. Ring Structure Sometimes there is more than one possible ring structure for a monosaccharide. The picture on the right is of the two possible ring structures for Glucose. α-Glucose is the isomer of glucose our body is able to use. β-Glucose is an isomer of glucose our body does not recognize. Notice the difference between the two is the placement of the hydroxyl group on carbon 1. Monosaccharides and Diet The body quickly breaks down simple carbohydrates to use as energy. Simple carbohydrates are found naturally in foods such as fruits, milk, and milk products. They also occur in processed and refined sugars such as candy, table sugar, syrups, and soft drinks. The majority of carbohydrate intake should come from complex carbohydrates (starches) and naturally occurring sugars rather than processed or refined sugars. Oligosaccharides These are what we call sugars with 2 carbons (‘disaccharides’) or 3 carbons (‘trisaccharides’). They are formed in an anabolic chemical reaction known as a dehydration synthesis reaction. Common Oligosaccharides Maltose is two α-glucose molecules together… found in beer. Sucrose which is an αglucose and a fructose together… table sugar. Lactose which is an αglucose and a galactose together… milk sugar. Maltose formation The glycosidic linkage, the bond between the sugars, in maltose is called an 1-4 glycosidic linkage because it occurs between carbon 1 of the first glucose and carbon 4 of the second glucose. Lactose Formation It is an 1-4 glycosidic linkage in lactose as well as the linkage is between carbon 1 of the glucose and carbon 4 of the galactose. Sucrose Formation The glycosidic linkage in sucrose is called an 1-2 glycosidic linkage because it occurs between carbon 1 of the glucose and carbon two of the fructose. Polysaccharide Information AKA ‘Complex Carbohydrates’, poly- many and saccharide for sugar. These are hundreds to thousands of monosaccharides held together by glycosidic linkages. Some are straight chained, others are branched, while some have side groups attached. Primarily used as carbohydrate storage, long-term energy source. Some are also used for structural support of the cell/organism. Starch It is the primary carbohydrate storage of plants. It is composed of two types of polysaccharides: Amylose which is a straight chain of α-glucose molecules in α 1-4 glycosidic linkages and Amylopectin which has a main chain of α-glucose in α 1-4 glycosidic linkages and branches of α-glucose off the main chain in α 1-6 linkages at the branch points. AMYLOSE AMYLOPECTIN Starch Starch is insoluble in water due to the angles of the glycosidic linkages. It is stored primarily in chloroplasts and some fruit of plants. Humans are able to break these glycosidic linkages to get at the energy in Starch. AMYLOSE AMYLOPECTIN Glycogen It is the long term energy storage molecule in animals. Any glucose we do not use as energy gets stored as glycogen. It is very similar to Amylopectin, but with more branching. Glycogen is stored in the liver and our muscles. Cellulose Cellulose is found in plant cell walls. It is used for structural support. It is a straight chain of β-glucose held together by β 1-4 glycosidic linkages. Every second glucose molecule is inverted (upsidedown). Cellulose Cellulose does not branch or coil. Cellulose chains pack close together because hydrogen bonds form between the hydroxyl groups of an inverted glucose and the hydroxyl group of a normal glucose. Cellulose We cannot digest cellulose, as the enzymes in our body only are able to hydrolyze 1-4 glycosidic linkages, but cellulose is an important part of our diet because it clears out our digestive tract (fibre!). Chitin Chitin is the material that makes up the exoskeletons of many arthropods. It is similar to cellulose however its monomers are glucose with a nitrogen group attached to the second carbon. Again due to the β-glucose, we cannot break it down for energy. Chitin is used in contact lenses and biodegradable stitches. Polysaccharides and Diet Complex carbohydrate foods provide vitamins, minerals, and fiber that are important to the health of an individual. The majority of carbohydrates should come from complex carbohydrates (starches) and naturally occurring sugars, rather than processed or refined sugars, which do not have the vitamins, minerals, and fiber found in complex and natural carbohydrates. Refined sugars are often called "empty calories" because they have little to no nutritional value.