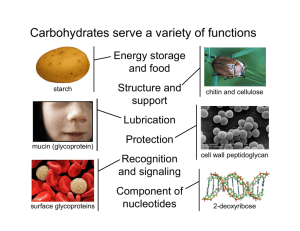
Carbohydrates
advertisement

Carbohydrates Polysaccharides Dr. Nikhat Siddiqi 1 Contents • Polysaccharides – Classification • Homoglycans • Heteroglycans Dr. Nikhat Siddiqi 2 Joining of monosaccharides • Monosaccharides can be joined to form disaccharides, oligosaccharides, and polysaccharides. • Important disaccharides include lactose (galactose + glucose), sucrose (glucose + fructose), and maltose (glucose + glucose). • Important polysaccharides include branched glycogen (from animal sources) and starch (plant sources) and unbranched cellulose (plant sources); each is a polymer of glucose. • The bonds that link sugars are called glycosidic bonds. These are formed by enzymes known as glycosyltransferases. Dr. Nikhat Siddiqi 3 Naming glycosidic bonds • Glycosidic bonds between sugars are named according to the numbers of the connected carbons, and also with regard to the position of the anomeric hydroxyl group of the sugar involved in the bond. Dr. Nikhat Siddiqi 4 Naming glycosidic bonds • If this anomeric hydroxyl is in the α configuration, the linkage is an α-bond. • If it is in the β configuration, the linkage is a β-bond. • Lactose, for example, is synthesized by forming a glycosidic bond between carbon 1 of β-galactose and carbon 4 of glucose. The linkage is, therefore, a β(1→4) glycosidic bond. Dr. Nikhat Siddiqi 5 Polysaccharides • Polysaccharides are also called as glycans. • Made up of large number of monosaccharides linked by glycosidic linkages. • Smaller glycans are called as oligosaccharides (10-15 monomers) found attached to popypeptides in glycoproteins and some glycolipids. • Found in membrane and secretary proteins. Dr. Nikhat Siddiqi 6 Polysaccharides - Classification Homoglycans Polysaccharides Heteroglycans Dr. Nikhat Siddiqi 7 Polysaccharides - Classification Structural Polysaccharides Polysaccharides Storage Polysaccharides Dr. Nikhat Siddiqi 8 Homoglycans • Found in starch, glycogen, cellulose and chitin. • Starch, glycogen, cellulose give D-glucose when hydrolysed. • Starch –energy storage molecule in plants. • Glycogen –energy storage molecule in animals. • Chitin – component of exoskeleton of insects, cell wall of fungi, yields glucose derivative Nacetylglucosamine when hydrolyzed. Dr. Nikhat Siddiqi 9 Starch (Storage Polysacchardes) • Significant source of carbohydrate in human diet. • Source- potatoes, rice, wheat, corn. Amylose Starch Amylopetin Dr. Nikhat Siddiqi 10 Amylose • Composed of long, unbranched chains of Sglucose residues that are linked by (1,4) glycosidic bonds. • Have one reducing end in which the ring can open to form free aldehyde group with reducing properties. Dr. Nikhat Siddiqi 11 Amylose • Contains several thousand glucose residues has molecular weight 150,000 to 600,000. • Linear amylose molecule forms tight helices. • Gives blue colour with iodine due to interaction between iodine molecules and the helically arranged glucose rsidues. Dr. Nikhat Siddiqi 12 Amylopectin • is branched polymer containing (1,4) and (1,6)glycosidic linkages. • The (1,6) points may occur every 20 to 35 residues and prevent helix formation. Dr. Nikhat Siddiqi 13 • Starch digestion begins in the mouth where the salivary enzyme - amylase initiates hydrolysis of glycosidic bonds. • Digestion continues in the small intestine where pancreatic -amylase hydrolyzes all the (1,4) glycosidic bonds except the branch points. • The products of - amylase maltose, trisaccharide maltotriose and limit dextrin. Dr. Nikhat Siddiqi 14 Starch - Structure Dr. Nikhat Siddiqi 15 Glycogen (Storage Polysaccharides) • Storage carbohydrate in vertebrates. • Found in liver and skeletal muscle. • Structure similar to amylopectin except that it has more branch point possibly at every fourth glucose residue. Dr. Nikhat Siddiqi 16 Glycogen-Structure Dr. Nikhat Siddiqi 17 Glycogen- Structure Dr. Nikhat Siddiqi 18 Cellulose (Structural Polysaccharides) • Polymer of D-glucopyranose residues linked by (1,4) glycosidic bonds. Unbranched • Structural polysaccharide in plants. • Pairs of unbranched cellulose molecules may contain as many as 12,000 glucose units each held together by hydrogen bonding to form sheet like strips called microfibrils. These structures are found in primary and secondary cell walls of plants. Dr. Nikhat Siddiqi 19 • The ability to digest cellulose is found in microorganism which contain the enzyme cellulase. • Cellulose can be hydrolyzed to its constituent glucose units by microorganisms that inhabit the digestive tract of termites and ruminants. • Cellulose makes up dietary fiber. • Paper, wood, textiles are some of cellulose containing products. Dr. Nikhat Siddiqi 20 Cellulose - Structure Dr. Nikhat Siddiqi 21 Chitin (Structural Polysaccharide) • Chitin is an unbranched polymer of N-Acetyl-Dglucosamine. • It is found in fungi and is the principal component of arthropod and lower animal exoskeletons, e.g., insect, crab, and shrimp shells. • It may be regarded as a derivative of cellulose, in which the hydroxyl groups of the second carbon of each glucose unit have been replaced with acetamido (-NH(C=O)CH3) groups. Dr. Nikhat Siddiqi 22 Chitin - Structure Dr. Nikhat Siddiqi 23 Polysaccharides Heteroglycans Dr. Nikhat Siddiqi 24 Heteroglycans • High molecular weight carbohydrate polymers that contain more than one kind of monosaccharides. • Major classes found in animals are N and Olinked glycans attached to proteins. Dr. Nikhat Siddiqi 25 Glycosaminoglycans (GAGs) • Glycosaminoglycans (GAGs) are large complexes of negatively charged heteropolysaccharide chains. • They are generally associated with a small amount of protein, forming proteoglycans, which typically consist of over 95% carbohydrate. Dr. Nikhat Siddiqi 26 • Glycosaminoglycans have the special ability to bind large amounts of water, thereby producing the gel-like matrix that forms the basis of the body's ground substance, which, along with fibrous components such as collagen, make up the extracellular matrix. • The viscous, lubricating properties of mucous secretions also result from the presence of glycosaminoglycans, which led to the original naming of these compounds as mucopolysaccharides. Dr. Nikhat Siddiqi 27 Structure of Glycosaminoglycans • Glycosaminoglycans are long, unbranched, heteropolysaccharide chains generally composed of a repeating disaccharide unit [acidic sugar– amino sugar]n. Dr. Nikhat Siddiqi 28 Structure of Glycosaminoglycans • The amino sugar is either Dglucosamine or Dgalactosamine, in which the amino group is usually acetylated, thus eliminating its positive charge. • The amino sugar may also be sulfated on carbon 4 or 6 or on a nonacetylated nitrogen. • The acidic sugar is either Dglucuronic acid or its carbon-5 epimer, L-iduronic acid. Dr. Nikhat Siddiqi 29 Structure of Glycosaminoglycans • A single exception is keratan sulfate, in which galactose rather than an acidic sugar is present. • These acidic sugars contain carboxyl groups that are negatively charged at physiologic pH and, together with the sulfate groups, give glycosaminoglycans their strongly negative nature. Dr. Nikhat Siddiqi 30 Some monosaccharide units found in glycosaminoglycans. Dr. Nikhat Siddiqi 31 Functions of GAGs • Because of their large number of negative charges, these heteropolysaccharide chains tend to be extended in solution. • They repel each other, and are surrounded by a shell of water molecules. • When brought together, they “slip” past each other, much as two magnets with the same polarity seem to slip past each other. • This produces the “slippery” consistency of mucous secretions and synovial fluid. Dr. Nikhat Siddiqi 32 Functions of GAGs---cont • When a solution of glycosaminoglycans is compressed, the water is “squeezed out” and the glycosaminoglycans are forced to occupy a smaller volume. • When the compression is released, the glycosaminoglycans spring back to their original, hydrated volume because of the repulsion of their negative charges. • This property contributes to the resilience of synovial fluid and the vitreous humor of the eye. Dr. Nikhat Siddiqi 33 Classification of the glycosaminoglycans • The six major classes of glycosaminoglycans are divided according to monomeric composition, type of glycosidic linkages, and degree and location of sulfate units. Dr. Nikhat Siddiqi 34 Structure and distribution of glycosaminoglycans (GAGs). Dr. Nikhat Siddiqi 35 Hyaluronic Acid • Hyaluronate molecules may consist of as many as 25,000 disaccharide units, with molecular weights of up to 107. • Hyaluronates are important components of the vitreous humor in the eye and of synovial fluid, the lubricant fluid of joints in the body. Dr. Nikhat Siddiqi 36 • The chondroitins and keratan sulfate are found in tendons, cartilage, and other connective tissue, whereas dermatan sulfate, as its name implies, is a component of the extracellular matrix of skin. • Heparin, with the highest net negative charge of the disaccharides shown, is a natural anticoagulant substance. Dr. Nikhat Siddiqi 37 Mucopolysaccharidoses • The mucopolysaccharidoses are hereditary disorders (1:25,000 births) that are clinically progressive. • They are characterized by accumulation of glycosaminoglycans in various tissues, causing varied symptoms, such as skeletal and extracellular matrix deformities, and mental retardation. • Mucopolysaccharidoses are caused by a deficiency of any one of the lysosomal hydrolases normally involved in the degradation of heparan sulfate and/or dermatan sulfate . Dr. Nikhat Siddiqi 38 Mucopolysaccharidoses MPS Type I Hurler’s syndrome results from a deficiency of alpha-L-iduronidase. Heparan sulfate and dermatan sulfate accumulate. There is growth and mental retardation with characteristic facial changes. MPS Type II Hunters syndrome is similar to Hurler’s syndrome but the enzyme deficiency is for iduronate sulfatase and the inheritance is X-linked. Dr. Nikhat Siddiqi 39 Glycoconjugates • Compounds which result from the covalent linkages of carbohydrate molecules to both proteins and lipids. • Two classes • A) proteoglycans • B) glycoproteins • Glycolipids are oligosaccharides containing lipid molecules found mainly on the outer surface of plasma membranes. Dr. Nikhat Siddiqi 40 Polysaccharides • Two broad classed viz., N-linked and O-linked. • N-linked are attached to polypeptides by an Nglycosidic bond with the side chain amide group of amino acid asparagine. • O-linked are attached to polypeptides by side chain hydroxyl group of the amino acid serine or threonine in a polypeptide chain or the hydroxyl group of membrane lipids. Dr. Nikhat Siddiqi 41 Polysaccharides linked to polypeptides Dr. Nikhat Siddiqi 42 Proteoglycans • High carbohydrate content (about 95%). • Occur on cell surfaces or are secreted into extracellular matrix. Dr. Nikhat Siddiqi 43 Proteoglycans • Proteoglycans consist of a protein core (brown) and one or more covalently attached glycosaminoglycan chains ([blue]. Dr. Nikhat Siddiqi 44 • Examples include syndecans, glycipcans and affrecans • The syndecans are a class of heparan sulfate and chondroitin sulfate containing proteoglycans in which the core protein is a transmembrane protein. • Aggregans is found in cartilage. Dr. Nikhat Siddiqi 45 Proteoglycan • Proteoglycan monomers typically are bound non-covalently to a hyaluronic acid molecule in association with linker proteins in a “bottle brush” arrangement. Dr. Nikhat Siddiqi 46 Dr. Nikhat Siddiqi 47 Functions of Proteoglcans • Organizing extracellular matrix. • Membrane bound syndecans, glycipcans bind to specific signal molecules like growth factors involved in cell cycle regulation. • Because of their vast number of polyionic GAG chains, the aggrecans trap large volume of water. • Give strength, flexibility to cartilage and tensile strength to collagen fibers. Dr. Nikhat Siddiqi 48 Glycoproteins • Proteins that are linked covalantly to carbohydrates through N- or 0- linkages. • Carbohydrate content varies from 1% to more than 85% of total weight. • Carbohydrates include monosaccharides or disaccharides such as those attached to collagen or branched oligosaccharides found on plasma glycoproteins. Dr. Nikhat Siddiqi 49 Polysaccharides linked to polypeptides Dr. Nikhat Siddiqi 50 Differences between Glycoproteins and Protoglycans • They differ from the proteoglycans (which might be considered a special case of glycoproteins) in that the length of the glycoprotein's carbohydrate chain is relatively short (usually 2–10 sugar residues in length, although they can be longer). Dr. Nikhat Siddiqi 51 Differences between Glycoproteins and Protoglycans ---cont • In addition, whereas glycosaminoglycans have diglucosyl repeat units, the carbohydrates of glycoproteins do not have serial repeats. • The glycoprotein carbohydrate chains are often branched instead of linear, and may or may not be negatively charged. Dr. Nikhat Siddiqi 52 • Glycoproteins contain highly variable amounts of carbohydrate. • For example, immunoglobulin IgG, contains less than 4% of its mass as carbohydrate, whereas human gastric glycoprotein (mucin) contains more than 80% carbohydrate. Dr. Nikhat Siddiqi 53 Functions of glycoproteins • Membrane-bound glycoproteins participate in a broad range of cellular phenomena, including cell surface recognition (by other cells, hormones (insulin receptors), and viruses), • cell surface antigenicity (such as the blood group antigens), • as components of the extracellular matrix and • of the mucins of the gastrointestinal and urogenital tracts, where they act as protective biologic lubricants. Dr. Nikhat Siddiqi 54 Functions of Glycoproteins Dr. Nikhat Siddiqi 55 • Sialic acid residues are responsible for high viscosity and luricating properties of saliva. • Cellular adhesion eg selectins (transient cellcell interaction), intergins (cell attachment to components of extracellular matrix) and cadherins (calcium dependent binding of cells to each other within tissue. Dr. Nikhat Siddiqi 56 Dr. Nikhat Siddiqi 57