POB Photosynthesis Lab
advertisement

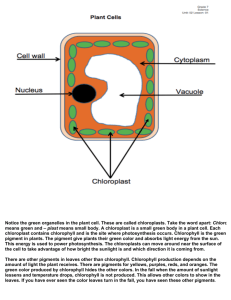
POB PHOTOSYNTHESIS LAB Amanda Matsumura Nathan Marcy Alisa Craig Tim Waldrip Dr. Ellen Turner, Instructor Introduction The purpose of experiment one is to demonstrate the reactants and products of photosynthesis. The purpose of experiment two is to demonstrate that energy is stored in plants in the form of starch. Experiment three demonstrates which colors of light are absorbed by plant pigments. Experiment four clearly demonstrates visually the different primary pigments and accessory pigments stored in the chloroplasts of spinach. Experiment I • Pour 50ml of bromothymol blue solution in a beaker • Gently blow into the solution until it turns yellowish-green • Place a sprig of Elodea into a vial with the yellowish-green bromothymol solution • Create a control vial with the yellowish-green solution alone • Place under a bright light (overhead light i.e.) • Observe changes you see in colors and also the presence of bubbles Experiment II • • • • • • • Pour 350 ml of water into a 600ml and bring to a boil on a hot plate Get one of the coleus leaves that have been kept away from light and one that has been photosynthesizing. Observe the differences between the two Boil the leaves for two minutes Place the leaves in a 250ml beaker and pour 50ml of ethanol over them Place the 250ml beaker with the leaves in the 600ml on the hot plate Boil the leaves in the alcohol in the water until the leaf turns pale, due to a loss of chlorophyll Carefully remove the chlorophyll-containing alcohol from the water bath and allow it to cool under the ventilation hood Experiment III • Place a sample of dried spinach leaves in a mortar, add 5ml of ethanol and grind into a fine, watery pulp • Put some of the ethanol-ground extract into a centrifuge tube and place it in one of the slots in the centrifuge head. Balance with a tube directly across from the extract • Centrifuge the sample for 5 minutes • Decant the chlorophyll extract into a Spectrophotometer curette. Read directions for use of the spectrophotometer (which is at the end of the lab) Experiment IV • Use a lead pencil to lightly mark a straight line across a strip • • • • • of chromatographic paper 1cm from the end Make cuts from the 1cm mark to the center of the end of the strip (diagram in lab explains) Use a Pasteur pipette to make a line of chlorophyll extract across the paper strip about 2cm above the point Place the chromatography paper strip into a bottle containing a small amount of petroleum ether solvent so only the tip is touching the liquid Bend the top of the paper over the lip of the bottle and lightly cap the bottle to slow the evaporation Measure the distance from the origin at the chlorophyll extract line to the center of each band and record Results Experiment 1: Acidic solution converts to basic solution as CO2 is consumed and oxygen is produced. Experiment 2: The leaf that has been starved contains no starch. The leaf that has been nourished has abundant starch. Experiment 3: Spectrophotometer readings confirm the absorption spectrums of pigments in chloroplasts. Plants absorb all wavelengths of light except green. Experiment 4: The pigments found in spinach clearly form different bands of color. EXPERIMENT 1 These are the materials needed for experiment one. On the left is the bromothymol blue solution (basic) and on the right is the elodea leaves. EXPERIMENT 1 Addition of CO2 to Bromothymol solution makes the solution acidic. A pH indicator in the solution causes the color to change from blue green to yellow. EXPERIMENT 1 The bromothymol blue solution in the presence of carbon dioxide becomes acidic. The indicator in the solution turns yellow to indicate a low pH. EXPERIMENT 1 Place the acidic bromothymol solution and the elodea in a sealed container. It should look similar to the above. EXPERIMENT 1 Place the sealed containers in a source of heat and light. For the purposes of this experiment, an overhead projector can be used. The class set up one control group and four experimental groups. EXPERIMENT 1 The results here clearly show that the acidic bromothymol solution turns back to a basic solution. Here we see the target experimental group next to the control group. EXPERIMENT 2 Plant one versus plant two. Differences in appearance are apparent. EXPERIMENT 2 Boiling the two leaves to remove waxy coating from the exterior. EXPERIMENT 2 Boiling the leaves again in alcohol to remove the chlorophyll. EXPERIMENT 2 The leaves, after boiling. Remove the leaves and dye them with iodine. Shine a light on the remaining solution. EXPERIMENT 2 The leaves after iodine staining. On the left, the malnourished leaf contains very little starch. On the right, the nourished leaf has abundant starch. EXPERIMENT 2 The alcohol containing the pigments from the leaves fluoresces red when a light is shone on it. EXPERIMENT 2 The pigments emit a red light when light is shone on them and no electron acceptor is present. EXPERIMENT 3 On the left, ethyl alcohol. On the right, spinach placed in a mortar. EXPERIMENT 3 Use the mortar and pestle to grind the alcohol soluble pigments put of the spinach leaves. EXPERIMENT 3 Decanting the primary and secondary pigments using alcohol in combination with a mortar and pestle. EXPERIMENT 3 Place two test tubes, one with the chlorophyll containing solution and the other with a ballast solution of water or alcohol on opposing sides of the centrifuge to properly balance it. EXPERIMENT 3 A centrifuge uses gravitational and inertial forces to separate components of a solution by weight. The pigments settle to the bottom while anything else rises to the top. EXPERIMENT 3 Using curettes and alcohol, zero the spectrophotometer. Then, place the pigment solution in the machine to obtain a reading. Record the data as instructed, and be sure to zero the machine between each reading. EXPERIMENT 4 Using the pigment solution from experiment 3, use a capillary tube or pipette to place a line on chromatography paper shaped as shown on the right. EXPERIMENT 4 Place the paper so that it is able to soak in petroleum ether. Be very careful, as petroleum ether is quite flammable. EXPERIMENT 4 Placing the chromatography paper inside the bottle of ether. Place just the tip inside the ether and allow the rest to sit above. Please note: put the cap back on the bottle of ether after the paper is in place or the ether will evaporate. EXPERIMENT 4 The results of the experiment. Several lines each indicative of a separate pigment can clearly be seen.