1.28.08 105K lecture
advertisement

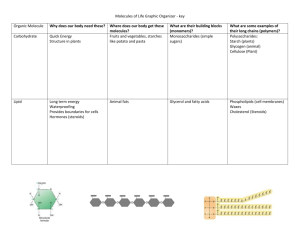
M W Day Date 1/14 1/16 Subject To be read prior to this class period: no class - Snow Day! Evolution: Unity and Diversity Chapter 1 M W 1/21 1/23 no class – Rev. Martin Luther King, Jr. Day Water and the Chemical Environment Chapter 2 (except pp30-35) M 1/28 Quiz #1 on Ch. 1 and parts of Ch. 2 and related lectures/handouts (no make ups for missed quiz) Cell Membranes Chapter 6 W M 1/30 2/4 Macromolecules Macromolecules W 2/6 Quiz #2 on Ch. 3 and 6 and related lectures/handouts (no make ups for missed quiz) The Cell Chapter 7 M W M W M 2/11 2/13 2/18 2/20 2/25 Bioenergetics Cell Respiration no class - Winter Break no class - Winter Break EXAM I Chapter 3 (except pp63-69) Chapter 9 Chapters 1, 6,7, parts of 2, 3 1) All living things are composed of cells* 2) All cells come from pre-existing cells* * The Cell Theory 3) All cells exist in a water environment 4) All cells are bounded by a membrane 5) All cells have similar chemistrys (proteins, proteins, proteins) 6) DNA is the primary heritable molecule (DNA’s BIG job is to code for proteins) 7) The concept of gene regulation and totipotency 1) All living things are composed of cells* 2) All cells come from pre-existing cells* * The Cell Theory 3) All cells exist in a water environment 4) All cells are bounded by a membrane 5) All cells have similar chemistrys (proteins, proteins, proteins) 6) DNA is the primary heritable molecule (DNA’s BIG job is to code for proteins) 7) The concept of gene regulation and totipotency methane, methanol, and amphipathic Dipole-dipole interactions: Because they have slight positive and negative charges, polar covalent bonds are attracted to other polar covalent bonds. A special example of these are HYDROGEN BONDS – These are sooo important! When H forms covalent bonds with O or N, the polar bond can form hydrogen bonds with other atoms. For example, water makes hydrogen bonds with other water molecules and other POLAR molecules or IONS. on your handout Water makes hydrogen bonds with other water molecules and other POLAR molecules or IONS and these are HYDROPHILIC (water loving) interactions. Things that dissolve in water are hydrophobic. Molecules that lack this capability (because they are composed of nonpolar covalent bonds) are HYDROPHOBIC (water fearing). Oil does not dissolve in water because the nonpolar bonds in the oil molecules are hydrophobic. on your handout NONCOVALENT INTERACTIONS – These occur between molecules (or between different parts of large molecules). The atoms do not share electrons. These are often called noncovalent “bonds” which is too bad because then they can get confused with covalent bonds. 1) Dipole-dipole interactions 2) London Dispersion forces 3) Ionic interactions on your handout MOLE – This a specific number similar to the concept that 12 is one “dozen”. You can have a dozen of anything (eggs, donuts, etc.) and it means you have twelve. Similarly, you can have a mole of anything (carbon, sucrose, etc.) and it means you have 6.02 x 1023. This funny number was established to make the following relationships convenient: MOLARITY - The concentration of a molecule in a solution. One mole per liter is called a 1 Molar ( 1 M) solution. on your handout Lowering the pH decreases the ability of water to dissolve salt. acetic acid 1) All living things are composed of cells* 2) All cells come from pre-existing cells* * The Cell Theory 3) All cells exist in a water environment 4) All cells are bounded by a membrane 5) All cells have similar chemistrys (proteins, proteins, proteins) 6) DNA is the primary heritable molecule (DNA’s BIG job is to code for proteins) 7) The concept of gene regulation and totipotency