Forces in a Nucleus - albert

Fundamentals of Radiation
Partial Periodic Table
Alkali
Metals
1A
1
H
1.01
Hydrogen
Alkaline
Earth
Metals
IIA
3
Li
6.94
Lithium
4
Be
9.01
Beryllium
IIIA
5
B
10.81
Boron
6
C
12.01
Carbon
Atomic
Chemical
Atomic
Name of
Number
Symbol
Mass
Element
Noble Gases
0
IVA
6
C
12.01
Carbon
VA
7
N
14.01
Nitrogen
VIA
8
O
16.00
Oxygen
VIIA
9
F
19.00
Flourine
2
He
4.00
Helium
10
Ne
20.18
Neon
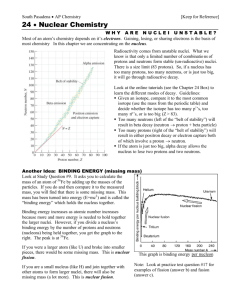
The Periodic Table provides the atomic number (Z), the chemical symbol, atomic mass, and element name. It also groups the elements based on their electron structure (I.e., how they react chemically).
Structure of the Atom
Nucleus
The nucleus contains neutrons and protons, also referred to as nucleons. The electrons orbit the nucleus.
The electrons are responsible for chemical reactions (e.g., formation of molecules).
The protons have a positive charge, the electrons a negative charge, and the neutrons are not charged.
Orbiting Electrons
The nucleons are responsible for nuclear reactions (e.g., radioactive decay).
Nomenclature
Z = Number of Protons
(determines the chemical element)
N = Number of Neutrons
(determines the isotope of the element)
A = Neutrons plus Protons
(atomic mass of the isotope)
A = Z + N
A
Z
X
N
X = Chemical Symbol
The chemical symbol and the atomic mass define the individual nuclide . (e.g., 3 H has 1 proton and 2 neutrons).
Isotopes of an element have the same number of protons, but a different number of neutrons in the nucleus.
Forces in a Nucleus
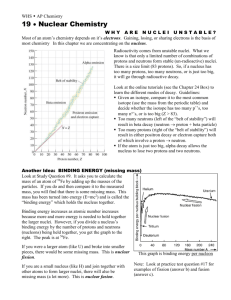
Nuclear force is an attractive force between each of the nucleons (i.e., neutrons and protons) over relatively short distances.
p
Nuclear force of neutron on proton n n Nuclear force of proton on neutron
Hydrogen-3 (Tritium)
Electrostatic force is a repulsive force between the like charged protons over a greater distance than nuclear forces.
Electrostatic force of proton on proton p
Nuclear force of proton on proton n p Nuclear force of proton on proton
Electrostatic force of proton on proton
Helium - 3
Radioactive Decay
The nuclides, as with most things in nature, want to be at their lowest energy state which is a stable nucleus.
Radioactive decay occurs in nuclides where the nucleus is unstable.
For stable nuclides with low atomic masses, the number of neutrons is equal to, or approximately equal to the number of protons (except for 1 H which only has one nucleon).
As the atomic mass of the nuclide increases, the ratio of neutrons to protons must be greater than one for it to be stable, suggesting that more neutrons are required to provide nuclear forces to offset the electrostatic repulsive force between the increased number of protons.
The nucleus may also become unstable when energy is added to it, placing it in an excited state. An example of this would be a free moving neutron inside of a reactor being captured by the nucleus of a 238 U nucleus.
The nuclide reaches its stable state by undergoing radioactive decay.
Types of Radiation
There are four types of radiation of interest:
1) Alpha (
) which is a positively charged helium nucleus (2 protons and 2 neutrons).
2) Beta (
) which is a negatively charged electron.
3) Gamma (
) which is a packet of energy with zero rest mass.
4) Neutron (n) which is a released neutron. Mainly a concern during nuclear reactor operation.
Alpha Particle
Helium-4 Nucleus
(2 neutrons, 2 protons)
Slow moving, but high energy
Cannot penetrate material easily
Stopped by one piece of paper
Stopped by dead layer of skin
Example of Alpha Decay
Radium-226
(88 protons, 138 neutrons)
Alpha particle
Radon-222
(86 protons, 136 neutrons)
Alpha decay occurs when the nuclides of high atomic mass have a lower neutron to proton ratio than stable nuclides and ejects an alpha particle.
Alpha decay is rare for nuclides with low or intermediate mass numbers.
Beta Particle
Electron
Fast moving, Medium energy
Can penetrate material well
Stopped by 100 to 150 pieces of paper
Stopped by 0.5 -1 centimeter of water
Example of Beta Decay
Carbon - 14
(6 protons, 8 neutrons)
Beta particle
Nitrogen - 14
(7 protons, 7 neutrons)
Beta decay occurs when the nuclides have a higher neutron to proton ratio than stable nuclides.
A neutron converts to a proton, electron (
), and a neutrino.
A neutrino is a high energy particle with zero rest mass with high penetrating capability.
Gamma Decay
Electromagnetic radiation
Similar to light, x-rays, radio waves
Emitted only by certain nuclei
Speed of light; low to high energy
Highly penetrating
Stop half of the
s with about 1 cm of lead
or 5 to 15 cm of water
Example of Gamma Decay
238 U + neutron
239 U +
Gamma decay occurs as a means of removing energy from the nucleus of an excited nuclide.
The gamma may be ejected alone or in conjunction with the emission of another radioactive particle (e.g.,
) to reduce the nucleus energy.
Examples of Neutron
Emission
There are Neutron (n) emissions associated with the following reactions.
2 H +
1 H + neutron
9 Be +
2 ( 4 He) + neutron
9 Be +
12 C + neutron
Neutron emissions of interest in a nuclear reactor occur when the excited nucleus of a specific high atomic mass nuclide splits into two or more smaller nuclides during the fission process.
235 U + n
135 I + 97 Y + 3n
Neutrons with high kinetic energy are released in the process.
Half-life
Each radioactive nucleus has a certain probability of decay per time
Some decay quickly (fractions of a second), some later (thousands of years)
Rate of decay depends on the number of nuclei available
As number decreases, rate of decay decreases
Half-life
In theory, all the radioactive material will never totally decay
Define Half-life
Time for half of the sample to decay
Half-life
1
1
16
1
8
1
4
1
2
1 2 3
Time (Number of half-lives)
4
Example Half-lives -
Natural
Uranium-238 (In soil)
4.5 Billion years
Potassium-40 (in soil and body)
1.3 Billion years
Carbon-14 (In all living tissue)
5730 years
Hydrogen-3 (in all water)
12 years
Example Half-lives -
Natural
Radium-226 (In soil - produces radon)
1600 years
Radon-222 (in soil and air)
3.8 days
Polonium-214 (radon progeny that decays in lungs)
164 microseconds (0.000164 s)
Example Half-lives -
Medical Uses
Iodine - 131 (Thyroid treatment)
8 days
Technetium-99m (Nuclear medicine)
6 hours
Gold-198 (Tumor therapy)
2.7 days
Activity
Activity = Decays per time
Units:
1 Becquerel = 1 decay per second (dps)
1 Curie = 37 Billion dps
1 microCurie ( m
Ci) = 37,000 dps
1 picoCurie (pCi) = 0.037 dps
Example Activities -
Regulations
Typical maximum alpha emitting radionuclide allowed without a license
(some exceptions)
0.1 m
Ci
Typical maximum beta emitting radionuclide allowed without a license
(some exceptions)
10 m
Ci
Example Activities - Natural
Radioactivity
Uranium-238 in cup of soil (typical)
0.003 m
Ci = 3000 pCi
Radon-222 in air
0.5 pCi per liter (outdoor air)
1 to hundreds of pCi per liter (houses)
Potassium-40 in human body
0.1 m
Ci
Radiation Dose
Dose = Energy absorbed per mass
Units:
Rad
Gray (Gy) [1 Gy = 100 rad)
Radiation Dose Equivalent
Different radiations do different amounts of biological damage
Dose Equivalent = Dose X QF
QF = Quality factor
Betas, Gamma: QF = 1; Alpha: QF = 20
Units
Rem (1 mrem = 0.001 rem)
Sievert (Sv) [1 Sv = 100 rem)]
Radiation Exposure
Old unit of exposure
Amount of radiation present in air
Only applicable for x-rays and gamma radiation
Units:
Roentgen (R)
1 R exposure in air will produce about 1 rad dose in human tissue
Example Doses
Natural annual background radiation
cosmic: 27 mrem (0.27 mSv)
Terrestrial: 28 mrem (0.28 mSv)
Internal: 39 mrem (.039 mSv)
[total natural (excl. Radon): ~100 mrem]
Radon-lungs: 2400 mrem (24 mSv)
[effective whole body: 200 mrem]
Source: NCRP Report #93
Example Doses
Medical Radiation (Effective Whole
Body Dose Equivalent)
Chest X-ray: 8 mrem (0.08 mSv)
Head CT scan: 111 mrem (1.11 mSv)
Barium Enema: 406 mrem (4.06 mSv)
Extremity X-ray: 1 mrem (0.01 mSv)
Source: NCRP Report 100
Radiation Safety
Radioactive materials produce a dose rate per time
To reduce total dose:
Minimum time
Half the time - half the dose
Shielding
Maximum distance
Twice the distance - one-fourth the dose