File
advertisement

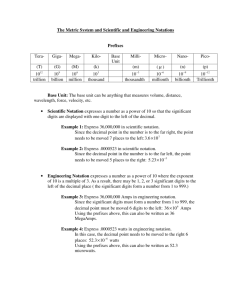
Teacher Resources Needed for Lesson • Copies of the following worksheets: – Rounding with Addition and Subtraction – Rounding with Multiplication and Division – Scientific Notation – Significant Digit Worksheet Rounding Answers and Scientific Notation Calculating with Significant Digits • In this course you will often take measurements and use them to calculate other quantities • Must be careful to keep track of which digits in your calculations and results are significant • We do this because your results of your calculations should not imply more certainty than your measured quantities justify Example • You are required to calculate the volume of a box. The dimensions of the box are determined to be 12.2cm by 10.1 cm by 9.40 cm. • When you do your calculation you get: 𝑉 = 𝑙𝑤ℎ 𝑉 = 12.2𝑐𝑚 10.1𝑐𝑚 9.40𝑐𝑚 𝑉 = 1158.268𝑐𝑚3 • The measurement all have three significant digits but the answer is reported to seven significant digits. This is saying that you are more certain of the value of the volume than you are of the initial measurements. This is not possible. Rules for Reporting Significant Digits in Calculations • Rule 1: Multiplying and Dividing The value with the fewest number of significant digits, going into the calculation, determines the number of significant digits that you should report in your answer. • Rule 2: Adding and Subtracting: The value with the fewest number of decimal places, going into the calculation, determines the number of decimal places that you should report in your answer. Rules for Reporting Significant Digits in Calculations • Rule 3: Rounding To get the appropriate number of significant digits (rule 1) or decimal places (rule 2), you may need to round your answer. If the first digit to be dropped is 4 or less, the preceding digit is not changed. (example: 8.674 is rounded to 8.67) If the first digit to be dropped is 5, the preceding digit is increased by 1. (example: 8.675 123 is rounded up to 8.68) If the first digit to be dropped is a lone 5 or a 5 followed by zeros, the preceding digit is not changed if it is even, but is increased if it is odd. (example: 8.675 is rounded up to 8.68, but 8.665 is rounded down to 8.66) When solving multistep problems record all the digits until the final answer to avoid rounding error Practice Problem (as a class) • A student measured a rectangular shaped sample of iron and found it to be 6.78 cm long, 3.906 cm wide, and 11 cm tall. Determine its volume to the correct number of significant digits. Practice Problem (as a class) • Suppose that you measure the masses of four objects as 12.5 g, 145.67 g, 79.0 g, and 38.438 g. What is the total mass of the objects? Homework!!! • Complete the following worksheets: – Rounding with addition and subtraction – Rounding with multiplication and division Scientific Notation Scientific Notation • Scientific notation expresses a number by writing it in the form 𝑎 × 10𝑛 , where 1 ≤ 𝑎 < 10 • Example: 2.997 924 58 × 108 𝑚/𝑠 • Digits in the coefficient 𝑎 are all significant • This makes it easier to write extremely large and extremely small numbers Converting Numbers into Scientific Notation • To convert a number into scientific notation you must first move the decimal place until the number is 1 ≤ 𝑎 < 10 • Count the number of times you move the decimal place and whether it has been moved to the right or the left • If the decimal is moved to left 𝑥 times then the power is 𝑎 × 10𝑥 Example 1: Convert 20 580 into scientific notation. 1) 2) 3) Move decimal place to the left four times 2.0580 Place the number of times you moved the decimal place as your power of ten 2.0580 × 104 If you are unsure if you did it correctly move the decimal place the number of times indicated in the power and if you end up with the number you started with originally you have done it correctly Converting Numbers into Scientific Notation • To convert a number into scientific notation you must first move the decimal place until the number is 1 ≤ 𝑎 < 10 • Count the number of times you move the decimal place and whether it has been moved to the right or the left • If the decimal is moved to right 𝑥 times then the power is 𝑎 × 10𝑥 Example 2: Convert 0.00000546 into scientific notation. 1) Move decimal place to the right six times 5.46 2) Place the number of times you moved the decimal place as your power of ten 5.46 × 10−6 (notice the negative sign when the decimal is moved to the right) Practice Problems • Copy the following into your notes and convert them into scientific notation: a) b) c) d) e) f) 9 523 458 0.000 001 952 756 300 000 000 0.000 000 000 000 123 15 0.23 Practice Problems • Copy the following examples into your notes and convert them into standard notation: a) b) c) d) e) 1.235 × 107 5.68 × 10−5 7.896 54 × 100 4.52 × 1012 8.889 × 10−8 Calculations Involving Scientific Notation • Can enter these values into a calculator and calculate an answer • If you are not using a calculator there are some rules you can follow to make calculations easier Addition and Subtraction: • Change all the numbers to the same power of 10 and then add or subtract the numbers Example: 1.234 × 105 + 4.2 × 104 = 1.234 × 105 + 0.42 × 105 = 1.234 + 0.42 × 105 = 1.654 × 105 = 1.65 × 105 Calculations Involving Scientific Notation Multiplication and Division: • Multiply or divide the coefficients, add or subtract the exponents, and express the result in scientific notation General Form for Exponents: 𝑥 𝑎 𝑥 𝑏 = 𝑥 𝑎+𝑏 𝑥𝑎 𝑎−𝑏 = 𝑥 𝑥𝑏 Example 1: 1.36 × 104 3.76 × 103 = 5.11 × 107 Example 2: 4.51 × 105 9 = 0.572 × 10 7.89 × 10−4 = 5.72 × 108 Homework!!! • Complete the following worksheets: – Scientific Notation – Significant Digit Worksheet