Cyclic Hydrocarbons
advertisement

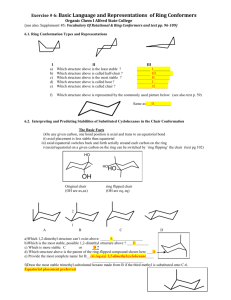
- alkanes which contain one or more carbon rings - general formula is CnH2n Ex.; Ex.; Name by IUPAC rules for hydrocarbons, EXCEPT add the prefix cyclo- to the name that indicates the number of carbons in the ring Ex.; Halogenation ◦ 1. Chlorination ◦ 2. Brominaaton – cyclopropane has highly strained bond angles and tends to open the ring, whereas larger rings do not 1. Reduction of Dihalides - brings about coupling between two carbons of the same molecule using zinc as the catalyst 2. Wurtz Reaction - uses sodium as the catalyst Because of the restricted rotation of carbon atoms in ring structures, disubstituted compounds will exist in two different isomeric forms 1. cis isomer - has the two smallest groups on the same side of the ring 2. trans isomer – has the two smallest groups on opposite sides of the ring conformation – different forms of the same compound, caused by rotation about single carbon-carbon bonds 1. conformation of ethane ◦ A. eclipsed formation - hydrogens are aligned with each other ◦ B. staggered formation - hydrogens on one carbon are located “between” the hydrogens on the other carbon ◦ - this is more stable because of the greater separation of atoms because of the 120° bond angles required for a six-membered planar ring, the cyclohexane ring is not planar, but “puckered” into two main conformations in order to maintain the 109° Equitorial bond – in the plane of the carbons Axial bond – perpendicular to the plane of the carbons all the hydrogens are staggered, therefore this is the more stable conformation There are 2 different chair forms that exist: the axial bonds of one chair form are the equitorial bonds of the other chair form and vice versa. The chair forms can be converted to each other by “ring flipping”. here, there is torsional strain; the hydrogens are too close together Substituted cycloalkanes prefer conformations where substituents occupy equitorial positions rather than axial positions. Trans isomer is more stable than the cis isomer because only in the trans isomer is a conformation possible in which both groups are equitorial Cis and trans isomers do not interconvert – that would require bond breaking