2013-04-15
advertisement

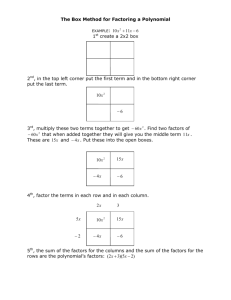
4/15/2013 (periods 1,3, & 5) 4/16/2013 (periods 2 & 6) Standards: 5 (Acids and Bases) Objectives: ● Complete CST 83-90 ● Students will be able to simplify ugly looking calculations DO NOW: 1. After turning in HW, Get responders and sign-in 2. Review how to calculate a log using my simple estimating log chart 3. CST Problems 83-90 with responders (practice) HOMEWORK: NONE =========================================================== TURNED IN: ● Journal 4/12 (what you learned about how to calculate the log of a number with a power of 10) 1. Ch 19.3 Ch 19.2 p. 611-613 19 (a,b,c); 20 (a,b,c) (5 pts) Std 4 2. CST Problems 77-82 (5 pts) Std MISC (KEEP FOR NOTEBOOK) For 4/16, see whiteboard for 4/15 4/17/2013 Standards: 5 (Acids and Bases) Objectives: ● Students will be able to calculate pH of solutions of STRONG acids or bases give the concentration of the STRONG acids or bases ● Students will be able to "file dump" for CST ● Students will be able to do neutralization problems DO NOW: 1. Copy the file dump page HOMEWORK: 1. Ch 19.3 p. 614 22 (a,b,c,d); p. 616 24-26; p. 621 30,31,32 (5 pts) Std 5 =========================================================== NO STAMPING: ● Journal 4/15 OR 4/16 (what you learned about simplifying calculations on CST) (KEEP FOR NOTEBOOK) # left of 1 2 3 4 5 6 7 8 9 10 10x log of# left 0.00 0.30 0.48 0.60 0.70 0.78 0.85 of 0.90 0.95 1.00 10x File dump for CST LeChat Conversions Diatomic elements ↓ ↑ stress LeChat does LeChat wants shift chart Avo g <----> mol <----> atoms/molecules NOH on 17 Trends vertica l& horian tal arrows loose electronegativity ionization energy atom size ion size mass metal non-met reactivity Review of calculating pH & pOH Multiply 2 x 2 = 4 Exponent Increases 1 x 10-1 = 0.1 1 x 10-2 = 0.01 1 x 10-3 = 0.001 1 x 10-4 = 0.0001 1 x 10-5 = 0.00001 1 x 10-6 = 0.000001 1 x 10-7 = 0.0000001 1 x 10-8 = 0.00000001 1 x 10-9 = 0.000000001 # left of 10x increases 1 x 10-9 = 0.000000001 2 x 10-9 = 0.000000002 3 x 10-9 = 0.000000003 4 x 10-9 = 0.000000004 5 x 10-9 = 0.000000005 6 x 10-9 = 0.000000006 7 x 10-9 = 0.000000007 8 x 10-9 = 0.000000008 9 x 10-9 = 0.000000009 10 x 10-9 = 0.000000010 = 1 x 10-8 1 x 10-8 = 0.00000001 2 x 10-8 = 0.00000002 3 x 10-8 = 0.00000003 4 x 10-8 = 0.00000004 5 x 10-8 = 0.00000005 6 x 10-8 = 0.00000006 7 x 10-8 = 0.00000007 8 x 10-8 = 0.00000008 9 x 10-8 = 0.00000009 10 x 10-8 = 0.00000010 = 1 x 10-7 1 x 10-7 = 0.0000001 2 x 10-7 = 0.0000002 PATTERN for # left of 10x and the the log of those numbers 1 x 10-9 ------------> 10 x 10-9 = 1.0 x 10-8 # left of 10x log of # left of 10x 1 2 3 4 5 6 7 8 9 10 0.00 0.30 0.48 0.60 0.70 0.78 0.85 0.90 0.95 1.00 1 x 10-7 ------------> 10 x 10-7 = 1.0 x 10-6 # left of 10x log of # left of 10x 1 2 3 4 5 6 7 8 9 10 0.00 0.30 0.48 0.60 0.70 0.78 0.85 0.90 0.95 1.00 CONTINUED 1 x 10-8 ------------> 10 x 10-8 = 1.0 x 10-7 # left of 10x log of # left of 10x 1 2 3 4 5 6 7 8 9 10 0.00 0.30 0.48 0.60 0.70 0.78 0.85 0.90 0.95 1.00 1 x 10-6 ------------> 10 x 10-6 = 1.0 x 10-5 # left of 10x log of # left of 10x 1 2 3 4 5 6 7 8 9 10 0.00 0.30 0.48 0.60 0.70 0.78 0.85 0.90 0.95 1.00 What is the take away of this exercise? 3 x 10-6 = 10-5.52 (6 - .48 = 5.52) = ph or pOH -log of 3 = .48 6 - .48 = 5.52 = -log of the number = ph or pOH STRONG ACIDS AND BASES I O N I Z E ionize 100% strong acids (memorized) IONIZE 100% HCl HNO3 HClO4 H2SO4 H3PO4 1 0 0 % (Gp 1 or Gp 2 with OH1-) HCl -----> H1+ + OH1- gas (one piece) H Cl strong bases ion ion M1V1a = M2V2b 4/18/2013 Standards: Biology - NCLB practice Objectives: ● Students will perform well for the State NCLB Bio Test DO NOW: 1. Read the NCLB background and answer as many of the questions as you can. Practice Quiz due end of period, you keep background notes. HOMEWORK: 1. =========================================================== TURN IN: ● Journal 4/17 (Calculating pH & pOH) (KEEP FOR NOTEBOOK)