Covalent Bonding
advertisement

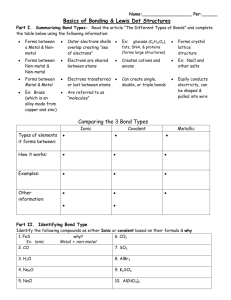
Chemistry Matter and Change Chapter 8 COVALENT BONDING CHAPTER 8 MAIN IDEA Covalent bonds form when atoms share electrons. 8.1 Main Idea Atoms gain stability when they share electrons and form covalent bonds. REVIEW VOCABULARY & CONCEPTS Chemical bond Valence electrons Electronegativity Lewis structure NEW VOCABULARY & CONCEPTS Covalent bond Molecule Sigma (σ)bond Pi (π)bond Endothermic reaction Exothermic reaction Single bond Double bond Triple bond WHY DO ATOMS BOND? Sharing electrons takes less energy than being “alone” Octet is usually the most stable electron configuration WHAT IS A COVALENT BOND? Shared pairs of electrons http://www.glencoe.com/sites/co mmon_assets/science/cmc/cim/an imations/ch8_3_1.swf SINGLE COVALENT BONDS Two atoms share one pair of electrons Sigma bond (σ) _ Either : or for a Lewis Structure · · H H·· H H+ or H-H MULTIPLE COVALENT BONDS Two atoms share more than one pair of electrons One pair is a sigma bond (σ)the others are pi (π)bonds Double bond shares 2 pair of electrons Triple bond shares 3 pair of electrons COMPARING BONDS Sigma (σ) Bonds Pi (π) Bonds Single Paired Centered Parallel s p orbital electrons One pair of electrons is shared orbital electrons Multiple pairs of electrons are shared MOLECULE A neutral group of atoms joined together by covalent bonds. DIATOMIC MOLECULES Molecules made up of two atoms. There are 7 diatomic molecules. H2, N2, O2, F2, Cl2, Br2, I2 DIATOMIC MOLECULES Br I N Cl H O F I couldn't exist without you! Oh, Ha Ha! HYDROGEN H· + ·H Hydrogen Atom Hydrogen Atom H:H Hydrogen molecule Shared electron pair The hydrogen molecule has a single covalent bond. The electronegativity of each hydrogenatom is the same, so the electrons are shared. MOLECULAR COMPOUNDS Compounds comprised of molecules. Ionic Compounds Molecular Compounds Crystal Lattice Molecule Metal with non-metal Non-metal with or polyatomic ions non-metal Solid Solid, liquid or gas Types of Elements Physical State Melting Point High > 300 C Solubility in Generally high water Electrical Good conductor conductivity of solution Low <300 C Generally low Poor to none VIDEO http://www.glencoe.com/sites/co mmon_assets/science/cmc/cim/an imations/ch8_1.swf LEWIS STRUCTURES AND OCTET Practice by drawing H2 O2 + N2 H2O CO2 + LEWIS STRUCTURES AND OCTET Practice by drawing H2 + - + LEWIS STRUCTURES AND OCTET Practice by drawing O2 σ - - - + - - - - - - - + - π - + - - - + - STRENGTH OF COVALENT BONDS Strength depends on distance of the atoms from each other With more bonds comes stronger bonds O2 is stronger than H2 single bond < Double bond < Triple bond CHEMICAL BONDS 22 CHEMICAL BONDS Electronegativity is an atom’s affinity for electrons. Differences in electronegativity dictate how electrons are distributed in covalent bonds. - nonpolar covalent bonds = equal sharing of electrons - polar covalent bonds = unequal sharing of electrons 23 BONDS AND ENERGY Endothermic reactions require additional energy for bonds to break Exothermic reactions release energy when the bonds break (spontaneous) Section 8.2 Chemistry Matter & Change NAMING MOLECULES 8.2 Main Idea Specific rules are used when naming binary molecular compounds, binary acids and oxyacids OBJECTIVES Translate molecular formulas into binary molecular names. Name acidic solutions. REVIEW VOCABULARY & CONCEPTS Ionic bond Covalent bond Formula unit Oxyanion Naming ionic substances Molecule NEW VOCABULARY & CONCEPTS Oxyacid Binary acid NAMING BINARY MOLECULAR COMPOUNDS 1. Name the first element using the entire name of the element. 2. The second element in the formula is named using the root and suffix “-ide.” 3. Prefixes are used to indicate the numbers of each element. COMMON PREFIXES Number of atoms 1 2 3 4 5 Prefix MonoDiTriTetraPenta- Number of atoms 6 7 8 9 10 Prefix HexaHeptaOctaNonaDeca- NAMING BINARY MOLECULAR COMPOUNDS Exceptions to the rules: The first element never uses “mono-” There is an understood 1 if nothing is specified. Awkward vowels can be dropped NAMING BINARY MOLECULAR COMPOUNDS EXCEPTION The first element never uses “mono-” There is an understood 1 if nothing is specified. CO2 is carbon dioxide not monocarbon dioxide CO is carbon monoxide, not monocarbon monoxide PRACTICE NAMING BINARY COMPOUNDS FORMULA SYSTEM NAME N2O dinitrogen monoxide NO nitrogen monoxide N2O3 dinitrogen trioxide NO2 nitrogen dioxide N2O4 dinitrogen tetroxide N2O5 dinitrogen pentoxide NO3 nitrogen trioxide NAMING BINARY ACIDS The first word has the prefix “hydro-” followed by the root of the second element followed by “-ic” The second word is “acid” HCl- hydrochloric acid PRACTICE NAMING BINARY ACIDS Hydrobromic acid Hydrophosphic acid Hydrosulfuric acid HF H2Se HI NAMING OXYACIDS First word consists of the root of the oxyanion (with prefixes if needed) followed by a suffix as specified If oxyanion ends in Oxyanion ending New suffix for acid -ate -ic -ite -ous The second word is always “acid.” NAMING OXYACIDS Relationshi General p name Oxyanion name Example name Example formula One more oxygen than (root)ic Per(root)ic acid Perchlorate Perchloric acid HClO4 Root(ic) acid chlorate Chloric acid HClO3 Root(ous) acid Chlorite Chlorous acid HClO2 One less oxygen than root(ic) Two less Hypo(root)ou Hypochlorou oxygens than Hypochlorite s acid s acid root(ic) HClO NAMING OXYACIDS First word consists of the root of the oxyanion (with prefixes if needed) followed by a suffix as specified The second word is always “acid.” Compound Oxyanion Acid suffix Acid name HClO3 Chlorate -ic Chloric acid HClO2 Chlorite -ous Chlorous acid HNO3 Nitrate -ic Nitric Acid HNO2 Nitrite -ous Nitrous Acid Look at the formula of the molecule Is it an acid? Yes Is there oxygen present in the compound? No Hydro(root) ic acid Yes No Name the first element using a prefix if necessary Name the second element indicating the number of atoms and changing the suffix to –ide. Root + -ic if the anion ends in –ate, or Root + -ous if the anion ends in –ite, then acid PRACTICE NAMING OXYACIDS Acid Formula Acid Name H2CO3 Carbonic Acid HBrO3 Bromic Acid H2C Carbonic Acid CH3COOH or CH3CO2H Acetic acid H3PO4 Phosphoric acid H2SO4 Sulfuric acid H2SO3 Sulfurous Acid CAN YOU Translate molecular formulas into binary molecular names. Name acidic solutions. Section 8.3 Chemistry Matter & Change MOLECULAR STRUCTURE 8.3 Main Idea Structural formulas show the relative positions of atoms within a molecule. OBJECTIVES List the basic steps used to draw Lewis structures. Explain why resonance occurs and identify resonance structures. Identify three exceptions to the octet rule and name molecules in which these exceptions occur. REVIEW VOCABULARY & CONCEPTS Ionic bond Covalent bond Lewis-dot structure Octet NEW VOCABULARY & CONCEPTS Structural formula Resonance Coordinate covalent bond Space filling model Ball and stick model MOLECULAR STRUCTURES Many different ways to depict the same thing Molecular formula Structural formula Lewis Structure Space-filling model Ball-and-stick model MOLECULAR FORMULA Indicates number of each element in a molecule H2O C6H12O6 NH3 STRUCTURAL FORMULAS Molecular model that shows the relative positions of the atoms O H H LEWIS STRUCTURE Shows shared electrons and lone pairs Shared pairs are usually depicted as lines Lone pairs may be lines or dots SPACE-FILLING MODEL Atoms are shown in relative size and position accounting for lone pairs BALL AND STICK MODELS Show relative position of atoms and bonds Easier to see double and triple bonds and bond angles HINTS FOR LEWIS STRUCTURES 1. Carbon is usually in the middle 2. Group 1 and 17 elements are always at ends 3. Atoms that are less numerous are usually in the middle (~Polyatomic Ions) SIMPLE PRACTICE FOR LEWIS STRUCTURES H2O HCl CH3F ORGANIC PRACTICE WITH LEWIS STRUCTURES Methane CH4 Ethane C2H6 Propane C3H8 TRICKIER PRACTICE CO2 HCN BH3 NH4+ TRICKIER ORGANIC PRACTICE C2H6 C2H4 C2H2 ethane ethene ethyne RESONANCE STRUCTURES More than one valid Lewis structure is possible Differ in position of electrons, not position of atoms HINTS FOR FORMING LEWIS STRUCTURES Carbon is usually in the middle Group 1 and 17 elements are always at ends Atoms that are less numerous are usually in the middle Hydrogen always forms one single bond Oxygen has two bonding electrons and two lone pairs Nitrogen has three bonding electron and one lone pair Group 13 elements have three bonding electrons and zero lone pairs Elements in groups 1, 2, 13 may break octet by having fewer electrons Phosphorus is a mess! REALLY HARD PCl5 SF6 ORGANIC CHEATS Common groups are often listed differently in organic molecules Ex: we write NH2CH2COOH instead of C2NO2H5 CAN YOU List the basic steps used to draw Lewis structures. Explain why resonance occurs and identify resonance structures. Identify three exceptions to the octet rule and name molecules in which these exceptions occur. Section 8.4 Chemistry Matter and Change MOLECULAR SHAPES Main Idea The VSEPR model is used to determine molecular shape OBJECTIVES Summarize VSEPR bonding theory Predict the shape of, and bond angles in, a molecule Define hybridization REVIEW VOCABULARY & CONCEPTS Atomic orbital s orbital p orbital NEW VOCABULARY & CONCEPTS VSEPR theory Hybridization Trigonal planar Trigonal pyramidal Tetrahedral Trigonal bipyramidal Linear Bent Octahedral VSEPR MODEL Valence Shell Electron Pair Repulsion Electrons repel each other Electrons stay as far away from each other as possible Lone pairs occupy more space than shared electrons HYBRIDIZATION Occurs because hybrid orbitals are more stable in some cases, notably carbon Remodel the “hotel” to make the “rooms” equal CARBON HYBRIDIZATION C: 1s22s22p2 1s22sp3 GETTING STARTED Begin with Lewis Structure Start with a tetrahedral and modify as needed Remember to use 3-D space not 2-D space VIDEO http://www.glencoe.com/sites/co mmon_assets/science/cmc/cim/an imations/ch8_2.swf CH4: A TYPICAL TETRAHEDRAL TETRAHEDRAL SHAPE Four atoms are coming from the center atom at equal angles 109.5° angle TRIGONAL PYRAMIDAL Three atoms coming from central atom and one lone pair Bond Lone angle 107° pairs occupy more space than shared pairs BENT Two atoms coming from central atom and two lone pairs 104.5° bond angle SUMMARY OF COMMON MOLECULAR SHAPES Molecul Tota Share e l d pair pairs s Lon e pair s Hybrid orbital Molecular shape BeCl2 2 2 0 sp Linear AlCl3 3 3 0 sp2 Trigonal planar CCl4 4 4 0 sp3 Tetrahedral NH3 4 3 1 sp3 Trigonal pyramidal H2O 4 2 2 sp3 Bent NbBr5 5 5 0 sp3d Trigonal bipyramidal SF 6 6 0 sp3d2 Octahedral TETRAHEDRAL BASED SHAPES Tetrahedral Trigonal Pyramidal Bent NON-TETRAHEDRAL BASED SHAPES Linear Trigonal Planar FEWER ELECTRONS THAN OCTET! NON-TETRAHEDRAL BASED SHAPES Trigonal Bipyramidal Octahedral MORE ELECTRONS THAN OCTET! CAN YOU… Summarize VSEPR bonding theory Predict the shape of, and bond angles in, a molecule Define hybridization Chapter 8.5 Chemistry Matter and Change ELECTRONEGATIVITY AND POLARITY Main Idea A chemical bond’s character is related to each atom’s attraction for the electrons in the bond. OBJECTIVES Describe how electronegativity is used to determine bond type. Compare and contrast polar and nonpolar covalent bonds and polar and nonpolar molecules Generalize about the characteristics of covalently bonded compounds REVIEW VOCABULARY & CONCEPTS Electronegativity Electron affinity Ionic bond Covalent bond NEW VOCABULARY & CONCEPTS Polar covalent bond Electronegativity difference Molecular polarity Intermolecular forces Solubility Dipole ELECTRON AFFINITY, ELECTRONEGATIVITY, AND BOND CHARACTER Electronegativity difference determines the character of the bond between atoms Most bonds are partly ionic and partly covalent Diatomics are 100% covalent Electronegativity difference Bond Character > 1.7 Mostly ionic 0.4 - 1.7 Polar covalent < 0.4 Mostly covalent 0 Nonpolar covalent BOND CHARACTER IONIC BONDS Created when the electronegativity difference is greater than 1.7 Elements are far apart on periodic table Example: Na and Cl Na = 0.9 Cl = 3.0 Difference is 2.1 ionic bond Electrons are transferred from Na to Cl NON-POLAR COVALENT BONDS Created when electronegativity difference is 0 Usually atoms of the same element Diatomics COVALENT BONDS Created when electronegativity difference is < 0.4 Elements are usually close together on the periodic table IBr POLAR-COVALENT BONDS Created when electronegativity difference is between 0.4 and 1.7 Most bonds fall into this category Electrons are shared unequally H2O POLAR COVALENT BONDS Molecules have a partial positive and a partial negative side DIPOLE INTERACTIONS Reactions between oppositely charged ends of a polar molecule POLAR COVALENT BONDS Polar bonds that are equal in all directions in a molecule create non-polar molecules SOLUBILITY RULES Like dissolves like Polar substances dissolve polar substances Non-polar substances dissolve non-polar substances. CAN YOU Describe how electronegativity is used to determine bond type. Compare and contrast polar and nonpolar covalent bonds and polar and nonpolar molecules Generalize about the characteristics of covalently bonded compounds