regulation
advertisement

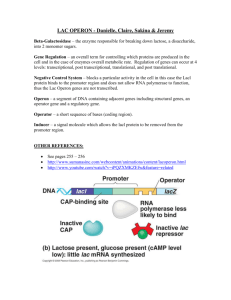
regulation c o u r s e l a y o u t introduction molecular biology biotechnology bioMEMS bioinformatics bio-modeling cells and e-cells transcription and regulation cell communication neural networks dna computing fractals and patterns the birds and the bees ….. and ants i n t r o d u c t i o n electronic pathway seoul subway tokyo subway pyrimidine pathway protein pathway from DNA to pathways biological information Two Types of Biological Information The genome, digital information Environmental, analog information genome information Two types of digital genome information Genes, the molecular machines of life Gene regulatory networks, specify the behavior of the genes what is systems biology? Biological System DNA Biomodules RNA Cells Networks Proteins a gene network a gene network in a physical network what is a genetic circuit? Jacob & Monod Model of the prokaryotic operon (1961) Repressor RNAP Inducer Gene A Promoter Operator what is a genetic circuit? Jacob & Monod Model of the prokaryotic operon (1961) “It is obvious from analysis of these [bacterial genetic regulatory] mechanisms that their known elements could be connected into a wide variety of ‘circuits’ endowed with any desired degree of stability” A Promoter Operator Gene A B Promoter Operator Gene B electronic circuits Basic electrical engineering (digital): A B And AB 00 01 10 11 C C 0 0 0 1 A B Or AB 00 01 10 11 C A B C 0 1 1 1 Nand AB 00 01 10 11 C C 1 1 1 0 A basic “flip-flop” = memory in1 Nand 1 out2 out1 Nand 2 in2 Stable states (with in1, in2 = 0): out1 out2 0 1 1 0 examples A genetic NAND Gate Gene A A genetic flip-flop out1 in1 in2 out2 basic genetic engineering How do you clone a gene? accessexcellence.com/AB/GG/plasmid.html genetic circuit engineering paradigm 1. Design Design “genetic circuitry” that demonstrates a rudimentary control behavior, such as oscillations, bistability (like the flip-flop), step activation, a spike, etc. 2. Simulate Build a simulation (deterministic or stochastic ODEs) encapsulating the design and examine its dynamic behavior (boundary conditions of different stability regimes, parameter sensitivity…). 3. Implement and Test Use the results of this simulation to pick genetic parts yielding the desired behavior and splice them together in a plasmid. Transform the plasmid into bacteria and observe the behavior of the system. Does it match predictions from the simulation? -- Back to 1 gene expression gene regulation mechanism Bacteria express only a subset of their genes at any given time. Expression of all genes constitutively in bacteria would be energetically inefficient. The genes that are expressed are essential for dealing with the current environmental conditions, such as the type of available food source. gene regulation mechanism Regulation of gene expression can occur at several levels: Transcriptional regulation: no mRNA is made. Translational regulation: control of whether or how fast an mRNA is translated. Post-translational regulation: a protein is made in an inactive form and later is activated. gene regulation Transcriptional control mechanism Translational control Post-translational control Lifespan of mRNA Onset of transcription Translation rate Protein Protein activation (by chemical modification) Ribosome DNA mRNA RNA polymerase Feedback inhibition (protein inhibits transcription of its own gene) Escherichia coli gene regulation mechanism Operon A controllable unit of transcription consisting of a number of structural genes transcribed together. Contains at least two distinct regions: the operator and the promoter. gene regulation mechanism Case study of the regulation of the lactose operon in E. coli E. coli utilizes glucose if it is available, but can metabolize other sugars if glucose is absent. gene Food source: regulation Glucose : Lactose mechanism Glucose : Lactose 1:3 Glucose : Lactose 1:1 70 60 50 40 30 20 10 0 3:1 29.5 14.0 43.5 26.5 39.0 13.5 0 1 2 3 4 5 0 1 2 3 4 Time (hours) 5 6 0 1 2 3 4 5 6 7 Second period of rapid growth with lactose as food source Initial period of rapid growth with glucose as food source gene regulation mechanism Case study of the regulation of the lactose operon in E. coli Genes that encode enzymes needed to break other sugars down are negatively regulated. Example: enzymes required to metabolize lactose are only synthesized if glucose is depleted and lactose is available. In the absence of lactose, transcription of the genes that encode these enzymes is repressed. How does this occur? gene regulation mechanism Case study of the regulation of the lactose operon in E. coli All the loci required for lactose metabolism are groupe d together into an operon. The lacZ locus encodes -galactosidase enzyme, which breaks down lactose. The lacY locus encodes galactosidase permease, a transport protein for lactose. The function of the lacA locus is unknown. The lacI locus encodes a repressor that blocks transcription of the lac operon. gene regulation mechanism Regulatory function Cleaves lactose to glucose and galactose Regulatory protein ß-galactosidase Lacl Membrane transport protein-imports lactose Galactosidase permease LacY LacZ Section of E. coli chromosome lacl lacZ Observations about regulation of lacZ and lacY: (1) Lacl protein and glucose shut down transcription of lacZ and lacY (2) Lactose induces transcription of lacZ andlacY lacY Glucose Lactose E. coli Galactose Galactosidase permease Chromosome ß-galactosidase gene regulation mechanism Lac operon lacl promoter lacl Promoter lac operon Operator lacZ lacY lacA gene regulation mechanism Repression and induction of the lactose operon. The lac operon is under negative regulation, i.e. , normally, transcription is repressed. Glucose represses transcription of the lac operon. Glucose inhibits cAMP synthesis in the cells. At low cAMP levels, no cAMP is available to bind CAP. Unless CAP is bound to the CAP site in the promoter, no transcription occurs. gene regulation mechanism When no lactose is present, the repressor binds to DNA and blocks transcription. NO TRANSCRIPTION Functional repressor lacl lacZ RNA polymerase blocked Operator (binding site for repressor) lacY gene regulation mechanism Repressor plus lactose (an inducer) present. Transcription proceeds. Lactose TRANSCRIPTION BEGINS repressor lacl + mRNA Permease galactosidase lacZ lacY gene regulation mechanism Operons produce mRNAs that code for functionally related proteins. "Polycistronic" mRNA lacZ message RNA polymerase binds to promoter lacY message lacA message lacl promoter lacl Promoter Operator lacZ lacY lacA cell programming programming cell communities Diffusing signal E. coli proteins programming cell communities Program cells to perform various tasks using Intra-cellular circuits Digital & analog components Inter-cellular communication Control outgoing signals, process incoming signals programmed cell applications Biomedical combinatorial gene regulation with few inputs; tissue engineering Environmental sensing and effecting recognize and respond to complex environmental conditions Engineered crops toggle switches control expression of growth hormones, pesticides Cellular-scale fabrication cellular robots that manufacture complex scaffolds programmed cell applications pattern formation programmed cell applications analyte source reporter rings analyte source detection biological cell programming biological cell programming cellular logic protein expression basics RNA polymerase binds to promoter RNAP transcribes gene into messenger RNA Ribosome translates messenger RNA into protein RNA Polymerase DNA Z Promoter Z Gene protein expression basics RNA polymerase binds to promoter RNAP transcribes gene into messenger RNA Ribosome translates messenger RNA into protein RNA Polymerase DNA Z Promoter Z Gene protein expression basics RNA polymerase (RNAP) binds to promoter RNAP transcribes gene into messenger RNA Ribosome translates messenger RNA into protein Transcription RNA Polymerase DNA Z Promoter Z Gene Messenger RNA protein expression basics RNA polymerase binds to promoter RNAP transcribes gene into messenger RNA Ribosome translates messenger RNA into protein Translation Z RNA Polymerase Protein Transcription Messenger RNA DNA Z Promoter Z Gene regulation through repression Repressor proteins can bind to the promoter and block the RNA polymerase from performing transcription The DNA site near the promoter recognized by the repressor is called an operator The target gene can code for another repression protein enabling regulatory cascades RNA Polymerase Transcription Translation R DNA Binding R R Promoter R Gene Z Promoter & Operator Z Gene transcription-based inverter Protein concentrations are analogous to electrical wires Proteins are not physically isolated, so unique wires require unique proteins R 1 0 R Z 0 1 simple inverter model Chemical Equations Repressor Binding Protein Synthesis Protein Decay R + O RO KR+R = (O)(R)/(RO) OO+Z kx Z kdeg Z R Total Concentration Equations Total Operator (OT) = (O) + (RO) Total Repressor (RT) = (R) + (RO) (R) if (RT) >> (O) R Operator Z Gene simple inverter model Transfer Function Derivation (O) (OT) d(Z) dt (Z) (O) = (O) + (RO) 1 = 1 1 + (RO)/(O) = 1 + (R)/KR+R Z R = kx • (O) – kdeg • (Z) = 0 at equilibrium = kx kdeg (O) = kx kdeg • (OT) 1 + (R)/KR+R R Operator Z Gene simple inverter model Chemical Equations Protein Synthesis Protein Decay R + O RO KR+R OO+Z kx Z kdeg Total Concentration Equations = (O)(R)/(RO) Output Protein Concentration Repressor Binding 1 Total Operator (OT) = (O) + (RO) Total Repressor (RT) = (R) + (RO) (R) if (RT) >> (O) 0.8 0.6 0.4 0.2 0 0 0.2 0.4 0.6 0.8 Input Protein Concentration 1 cooperativity Cooperative DNA binding is where the binding of one protein increases the likelihood of a second protein binding Cooperativity adds more non-linearity to the system Increases switching sensitivity Improves robustness to noise Transcription Translation RNA Polymerase R Cooperative DNA Binding R R R Promoter R Gene Z Promoter & Operator Z Gene cooperative inverter model Chemical Equations Coop Binding Protein Synthesis Protein Decay R + R + O R2O KR2O = (O)(R)2/(R2O) OO+Z kx Z kdeg Z R Total Concentration Equations Total Operator (OT) = (O) + (R2O) Total Repressor (RT) = (R) + 2•(R2O) (R) if (RT) >> (O) R R Operator Z Gene cooperative inverter model Transfer Function Derivation (O) (OT) d(Z) dt (Z) = (O) (O) + (RO) = 1 1 + (RO)/(O) = 1 1 + (R)2/KR20 = kx • (O) – kdeg • (Z) = 0 at equilibrium = kx kdeg (O) = kx kdeg • Z R (OT) R R 1 + (R)2/KR+R Operator Z Gene Cooperative Non-Linearity cooperative inverter model Chemical Equations Protein Synthesis Protein Decay R + R + O R2O KR2O = (O)(R)2/(R2O) OO+Z kx Z kdeg Total Concentration Equations Output Protein Concetration Coop Binding 1 Total Operator (OT) = (O) + (R2O) Total Repressor (RT) = (R) + 2•(R2O) (R) if (RT) >> (O) No Coop Coop 0.8 0.6 0.4 0.2 0 0 0.2 0.4 0.6 0.8 Input Protein Concentration 1 cellular logic summary Current systems are limited to less than a dozen gates Three inverter ring oscillator [ Elowitz00 ] RS latch [ Gardner00 ] Inter-cell communication [ Weiss01 ] A natural repressor-based logic technology presents serious scalability issues Scavenging natural repressor proteins is time consuming Matching natural repressor proteins to work together is difficult Sophisticated synthetic biological systems require a scalable cellular logic technology with good cooperativity Zinc-finger proteins can be engineered to create many unique proteins relatively easily Zinc-finger proteins can be fused with dimerization domains to increase cooperativity A cellular logic technology of only zinc-finger proteins should hopefully be easier to characterize in vivo logic circuits E. coli logic gates a genetic ci rcuit buildi ng block logic circuit based on inverters Proteins are the wires/signals Promoter + decay implement the gates NAND gate is a universal logic element: any (finite) digital circuit can be built! NAND and NOT gate X XY Y X X x y NAND 0 0 1 0 1 1 1 0 1 1 1 0 x NOT 0 1 1 0 logic circuit based on inverters X X R1 Z Y NAND NOT = R1 Z gene R1 Y gene gene why di gital? We know how to program with it Signal restoration + modularity = robust complex circuits Cells do it Phage λ cI repressor: Lysis or Lysogeny? [Ptashne, A Genetic Switch, 1992] Circuit simulation of phage λ [McAdams & Shapiro, Science, 1995] Also working on combining analog & digital circuitry why di gital? BioCircuit CAD SPICE http://bwrc.eecs.berkeley.edu/classes/icbook/SPICE/ BioCircuit steady state CAD dynamics BioSPICE a prototype biocircuit CAD tool simulates protein and chemical concentrations intracellular circuits, intercellular communication single cells, small cell aggregates intercellular genetic circuit elements translation RBS input mRNA RBS output mRNA ribosome ribosome transcription operator promoter RNAp modeling a biochemical inverter input repressor promoter output a BioSPICE inverter simulation input repressor promoter output smallest memory: RS-latch flip-flop 0 1 1 0 The output a of the R-S latch can be set to 1 by momentarily setting S to 0 while keeping R at 1. When S is set back to 1 the output a stays at 1. 1 0 0 1 Conversely, the output a can be set to 0 by keeping S at 1 and momentarily setting R to 0. When R is set back to 1, the output a stays at 0. RS-latch flip-flop truth table R 0 0 1 1 Q = R + ~Q ~Q = S + Q R S S 0 1 0 1 Q(n+1) Q(n) 1 0 0 Q ~Q ~Q(n+1) ~Q(n) 0 1 0 RS-latch timing diagram 1 R 0 1 S 0 1 Q 0 1 ~Q 0 R S - l a t c h d a n g e ro u s t ra ns i ti on R S 10 10 0 0 Q ~Q proof of concept in BioSPICE RS-Latch (“flip-flop”) Ring oscillator _ [R] _ [S] [A] _ R A [B] time (x100 sec) [B] _ S B [C] [A] time (x100 sec) time (x100 sec) They work in vivo Flip-flop [Gardner & Collins, 2000] Ring oscillator [Elowitz & Leibler, 2000] However, cells are very complex environments Current modeling techniques poorly predict behavior Work in BioSPICE simulations [Weiss, Homsy, Nagpal, 1998] the IMPLIES gate Inducers that inactivate repressors: IPTG (Isopropylthio-ß-galactoside) Lac repressor aTc (Anhydrotetracycline) Tet repressor Use as a logical IMPLIES gate: (NOT R) OR I Repressor Inducer Output Repressor 0 0 1 1 Inducer 0 1 0 1 Output 1 1 0 1 the IMPLIES gate RNAP active repressor inactive repressor inducer no transcription RNAP promoter operator gene promoter operator gene transcription the toggle pIKE = lac/tet pTAK = lac/cIts [Gardner & Collins, 2000] switch the toggle switch promoter protein coding sequence [Gardner & Collins, 2000] the ring [Elowitz, Leibler 2000] oscillator the ring oscillator The repressilator is a cyclic negative-feedback loop composed of three repressor genes and their corresponding promoters, as shown schematically in the centre of the left-hand plasmid. It uses PLlacO1 and PLtetO1, which are strong, tightly repressible promoters containing lac and tet operators, respectively6, as well as PR, the right promoter from phage l. The stability of the three repressors is reduced by the presence of destruction tags (denoted `lite'). The compatible reporter plasmid (right) expresses an intermediate-stability GFP variant11 (gfpaav). In both plasmids, transcriptional units are isolated from neighbouring regions by T1 terminators from the E. coli rrnB operon (black boxes). The repressilator network the ring oscillator evaluation of the ring oscillator Comparison of the repressilator dynamics exhibited by sibling cells. In each case, the fluorescence timecourse of the cell depicted in the Fig is redrawn in red as a referen ce, and two of its siblings are shown in blue and green. [Elowitz, Leibler 2000] evaluation of the ring oscillator a, Siblings exhibiting post-septation phase delays relative to the reference cell. b, Examples where phase is approximately maintained but amplitude varies significantly after division. c, Examples of reduced period (green) and long delay (blue). d, Two other examples of oscillatory cells from data obtained in different experiments, under conditions similar to those of a±c. There is a large variability in period and amplitude of oscillations. e, f, Examples of negative control experiments. e, Cells containing the repressilator were disrupted by growth in media containing 50mM IPTG. f, Cells containing only the reporter plasmid. evaluation of the ring oscillator Reliable long-term oscillation doesn’t work yet: Will matching gates help? Need to better understand noise Need better models for circuit design [Elowitz, Leibler 2000] three repressors LacI is a repressor protein made from the lacI gene, the lactose inhibitor gene of E. coli. TetR is a repressor protein made from the tetR gene. CI is a repressor protein made from the cI gene of phage. Each one of these, with its cognate promoter, will stop production of whatever gene is ‘downstream’ from the promoter. ring oscillator with mismatched inverters A = original cI/λP(R) B = repressor binding 3X weaker C = transcription 2X stronger device physics in steady state “Ideal” inverter “gain” [output] 0 [input] Transfer curve gain (flat,steep,flat) adequate noise margins 1 Curve can be achieved with certain dna-binding proteins Inverters with these properties can be used to build complex circuits measuring a transfer curve Construct a circuit that allows: Control and observation of input protein levels Simultaneous observation of resulting output levels inverter CFP R “drive” gene Also, need to normalize CFP vs YFP YFP output gene flow cytometry (FACS) drive input levels by varying inducer lacI [high] 0 P(lacIq) IPTG (uM) YFP P(lac) (Off) 0 IPTG P(lacIq) 100 lacI IPTG P(lac) promoter protein coding sequence YFP 1000 measuring a transfer curve for lacI/p(lac) 0 tetR lacI CFP [high] P(R) (Off) aTc P(R) YFP P(lac) P(LtetO-1) measure TC tetR P(lac) aTc P(Ltet-O1) lacI CFP YFP transfer curve data points 10 undefined 1,400 1,400 1,200 1,200 1,200 1,000 1,000 1,000 800 600 Events 1,400 Events Events 01 800 600 800 600 400 400 400 200 200 200 0 0 1 10 100 1,000 Fluorescence (FL1) 1 ng/ml aTc 10,000 0 1 10 100 1,000 Fluorescence (FL1) 10 ng/ml aTc 10,000 1 10 100 1,000 Fluorescence (FL1) 100 ng/ml aTc 10,000 lacl/p(lac) transfer curve lacI CFP [high] P(R) (Off) P(LtetO-1) YFP 1000 P(lac) gain = 4.72 aTc Output (YFP) 0 tetR 100 10 1 1 10 100 Input (Norm alized CFP) 1000 evaluating the transfer curve Gain / Signal restoration Noise margins 1,400 1,000 1,000 100 Events Fluorescence 1,200 high gain 800 600 10 400 200 1 30 ng/ml aTc 3 ng/ml aTc 0 0.1 1.0 10.0 100.0 1 aTc (ng/ml) *note: graphing vs. aTc (i.e. transfer curve of 2 gates) 10 100 Fluorescence 1,000 applications some possibilities “Forward Engineering” as a means of learning about natural genetic regulation. Biotechnology Experimental systems Validation of models forward engineering Reductionism + Simulation = reverse engineering. Main Difficulty: system is WAY to complex reductionism will never be finished when it is, models/ parameter-space will be too huge we don’t have much intuition for parallelism, processes interacting at different scales... Possible modes of attack: Engineering math: sensitivity analysis, control theory “Complex Systems” analysis forward engineering Forward Engineering Approach: “We learned more about how birds fly from trying to build airplanes than from studying structural anatomy of birds.” - ?(ai) Try to build something that has same functionality as system under study. Learn what some of the critical component requirements are, what the main design challenges. Generate testable hypotheses about how natural genetic regulation functions. Use forward and reverse engineering techniques in parallel. biotechnology Genetic engineering applications: production of antibiotics and other drugs production of proteins for: detergents, solvents, aminos… bioremediation Metabolic Control Analysis, directed evolution and other techniques used to optimize design of metabolic pathways for given task. Genetic circuit engineering could yield finer more sophisticated control. Genetic circuits as sensors. experimental systems Perhaps genetic circuits can be used as clever assays/probes, similar to the Yeast Two-Hybrid system used to detect interacting proteins. A Transcription Factor Fuse domains to putative interacting proteins DNA Activation Binding Activation fish Is TF active? GFP Or Genetic circuits could be used to examine a system’s response to complex controllable inputs. bait DNA Binding validation of modeling techniques Many competing techniques for modeling biochemical systems: kinetics-based, stochastic kinetics, graph theoretical, discrete-event… Ultimate gold-standard would be to design a system using a simulation technique, build it, and verify predictions of model.