AP BIO Unit 3 TEST REVIEW - KEY
advertisement

Unit 3 TEST REVIEW
ENERGY, ENZYMES, CELLULAR RESIPRATION & PHOTOSYNTHESIS
AP Biology Textbook Chapters 8, 9 & 10
ENERGETICS, METABOLISM & ENZYMES – CHAPTER 8
Chemical reactions in Metabolic Processes
1] What is the primary source of ENERGY INPUTS for all living things?
The Sun.
a) What is a producer, or an autotroph? Give several examples from
Kingdom Plantae or Moneran? A producer is an organism that makes its
own food from either light (photoautotrophs), or inorganic reduced
compounds like methane and ammonia
(chemolithoautotrophs). Photoautotrophs – tulips, oak trees.
Chemolithoautrotrophs (H2S, S0, Fe2+, NO2−or NH3) – Nitrobacter,
Nitrosomas, Acidithiobacillus ferrooxidans.
b) What is a consumer, or a heterotroph? Give several examples
from Kingdom Animalia, Protista or Fungi? A consumer is an
organism that derives its energy from ingesting [Kingdom Animlia] or
absorbing [Kingdom Fungi] other organisms. An example of a heterotroph is
an amoeba, a human, and a morel mushroom.
c) How does this energy travel from producers to consumers?
Producers form the base or foundation of all trophic pyramids.
Producers capture energy from the Sun, coupled with carbon dioxide, and
transform it into biologically active compounds like glucose. First level
consumers, herbivores, then ingest those producers. The herbivores are
eaten by secondary consumers. Secondary consumers are eaten by tertiary
consumers. Only 10% of the energy that is available actually makes it to the
next level consumer due to losses due to metabolism and heat dissipation.
2] Define METABOLISM.
The sum of all chemical reactions that occur in the body. the set of life-sustaining
chemical transformations within the cells of living organisms. These enzyme-catalyzed
reactions allow organisms to grow and reproduce, maintain their structures, and
respond to their environments. The word metabolism can also refer to all chemical
reactions that occur in living organisms, including digestion and the transport of
substances into and between different cells.
a) The reactions of biological life form bonds between molecules
through the process of dehydration synthesis.
This is called a(n) anabolic reaction, and it
requires energy, making it endergonic.
b) The reactions of biological life break bonds between molecules
through the process of hydrolysis.
This is called a(n) catabolic reaction, and it releases energy, making
it exergonic.
Often these two types of reactions are coupled together, why?
The exergonic reaction can power the endergonic reaction creating
efficiencies from an energy perspective.
3] ENERGY:
a) First Law of Thermodynamics:
Energy can neither be created nor destroyed, it can only be transformed.
b) Second Law of Thermodynamics:
The universe tends toward disorder, because of increases in entropy due to
dissipation of energy and to dispersal of matter and energy.
c) Gibb’s Free Energy:
The Gibbs free energy of a system at any moment in time is defined as the
enthalpy of the system minus the product of the temperature times the
entropy of the system. G = H - TS. The Gibbs free energy of the system is a state
function because it is defined in terms of thermodynamic properties that are
state functions.
4] ENZYMES are biological catalysts:
a) Are composed of globular proteins
b) Naming convention? [- ase] is added to the substrate that it acts upon –
for example sucrase is an enzymes that acts upon sucrose; Peptidases are
enzymes that act upon peptide bonds, and lactase breaks down lactose.
Give several examples: catalase (breaks down peroxide into water and
oxygen), amylase breaks down starches into simple sugars.
c) Primary function:
Reduce activation energy in chemical reactions, in order to facilitate
necessary metabolic reactions to take place.
d) Characteristics:
1) Substrate-specific (describe & Illustrate)
Enzymes are shaped in specific way to uniquely interact with
one particular substrate.
2) Enzymes remain unchanged as a result of the reaction
Enzymes remain unchanged chemically and conformationally
after being used in a chemical reaction. This allows them to
be re-used again rapidly by the body.
3) Catalyze reactions in both the forward and backwards
direction
The direction of net activity is determined by the substrate
concentrations and other factors. The net direction of an
enzyme reaction can be driven in the forward direction by
keeping the product concentration low (by its removal or
conversion to another product).
4) Enzyme efficiency can be affected by multiple factors such
as pH [pepsin low pH – trypsin more neutral pH], salinity and
temperature [loss of 3D shape, disrupts bonds between
enzyme and substrate]. When an enzyme’s chemical bonds
are disturbed and its shape is altered it experiences a
conformational change leaving it unable to function. This is
called denaturing.
5) Lock and key vs Induced-fit model – Describe and compare,
which is a more accurate model given our current
understanding.
6) Draw an enzyme catalyzed reaction. Label the
following: substrate(s), enzyme, active site,
enzyme-substrate complex, and the product(s)
ENZYME REGULATION:
e) Compounds that help enzymes – Activators:
1) Co-factors: non-protein, INORGANIC molecules & ions that
assist enzymes SUCH AS: Mg, K, Ca, Zn, Fe, & Cu
BOUND within an enzyme molecule {Mg in chlorophyll
or Fe in hemoglobin.
2) Coenzymes: ORGANIC cofactors that usually accept or
donate some part of a reaction – often electrons. Some
vitamins are co-enzymes or components of co-enzymes.
NAD: niacin, B3
FAD: riboflavin B2
Coenzyme A
3) Allosteric activators: bind & keep enzymes in the active
form
f) Compounds that regulate enzymatic action – Inhibitors:
1) Competitive [i.e. penicillin – blocks enzyme used to build cell
walls, disulfiram/Anabuse]
Inhibitor & substrate compete for the active site; can be
overcome by increasing the concentration of substrate
2) Non-competitive {Allosteric sites} [i.e. cyanide
poisoning – irreversible inhibitor of cytochrome c – an enzyme
needed in cellular respiration, some cancer drugs – inhibit DNA
synthesis]
Inhibitor binds to a site other than the active site – which
causes a change in shape of the active site
3) Feedback inhibition
Product is used by the next step in the pathway; final product
is an inhibitor for an earlier step. Secondary benefit, no
unnecessary accumulation of products [efficient].
5] ATP Adenosine Triphosphate, common source of activation energy for
metabolic reactions – “energy currency molecule.”
a) Composition? Adenosine = adenine + ribose; with three
phosphate groups attached
b) What is hydrolyzed to release energy? The phosphate bond
between the 2nd and 3rd phosphate group.
c) It is re-activated by a dehydration reaction. Draw this.
What is this specific synthesis reaction called? Phosphorylation
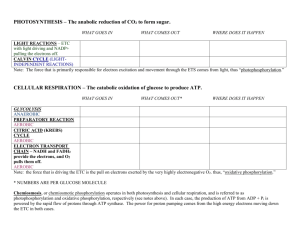
CELLULAR RESPIRATION - CHAPTER 9 [OXIDATION – EXERGONIC]
6] Harvesting stored energy – energy is stored in organic molecules.
What is the overall reaction for the breakdown of glucose to yield ATP?
a) [in words]
Glucose + Oxygen -> Water + Carbon dioxide + Energy
b) [chemical shorthand]
C6H12O6 + 6 O2 6 H2O + 6 CO2 + 36 ATP
This process is really many processes – organisms are “burning” organic
fuels in a series of step-by-step enzyme-controlled reactions.
7] Oxidation-Reduction Reactions break bonds and move electrons from
one molecule to another. As electrons move, they carry energy with them.
That energy is stored in another bond, released as heat or used to make
ATP:
Compare & contrast:
a) Oxidation [OIL]:
b) Reduction [RIG]:
adding O
- removing O
removing H
- adding H
loss of electrons
- gaining electrons
releases energy
- stores energy
exergonic
- endergonic
c) Re-write the reaction for cellular respiration showing the electron
movement. Show which compounds are oxidized, and which are
reduced. Electron carriers move electrons by shuttling H atoms
around – NAD+ NADH (reduced)
FAD 2+ FADH (reduced)
oxidation
ATP
C6H12O+
6 6O2 6CO2+ 6H2O+
reduction
8] CELLULAR RESIRATION:
Four metabolic stages:
a) GLYCOLYSIS – the decomposition of glucose to pyruvate –
substrate level phosphorylation [when a phosphate group and its
associated energy are transferred to ADP to form ATP]
Site: Cytosol
Reactants: 1 molecule of glucose, can occur in the presence or
absence of oxygen.
Process: requires 2 ATP, produces 4 ATP produced by substrate level
phosphorylation, and 2 NADH – a coenzyme & energy rich molecule
Products: 2 molecules of Pyruvate & net 2 ATP
b) Pyruvate oxidation – ‘the link reaction’
2 Pyruvate + coenzyme A 2 Acetyl CoA + 2 CO2 + 2 NADH
c) KREB’S CYCLE, A.K.A. CITRIC ACID CYCLE
Site: Matrix – fluid material that fills the area inside the inner
membrane of the mitochondria
Reactants: 2 (Acetyl CoA + oxaloacetate citrate).
Process: There are seven intermediate products
Products: 2 (3 NADH, 1 FADH2 (coenzymes – accepting electrons
during a reaction), 1 ATP & 2 CO2 (carbon dioxide we exhale))
d) OXIDATIVE PHOSPHORYLATION =
ELECTRON TRANSPORT CHAIN + CHEMIOSMOSIS
oxidative phosphorylation – the process of producing ATP from
NADH and FADH – as electrons pass along the ETC.
Site: cristae, inner membrane of the mitochondria
Reactants: 6 O2, 10 NADH, 2 FADH2
Process: electrons pass from NADH and FADH2, from one carrier
protein to another.
What are cytochromes? A cytochrome is a 100 amino acid
sequenced hemeprotein – carrier protein, catalyze redox reactions
Products: 34 ATP & 6 H2O
CHEMIOSMOSIS: mechanism of ATP generation that occurs when energy is
stored in the form of a proton gradient across a membrane.
1] Kreb’s cycle [produces NADH & FADH2 in the matrix.
2] Electrons are removed from NADH & FADH2 – protein complexes in the
inner membrane remove electrons from these two molecules. Protein
protein….
3] H+ ions (protons) are transported from the matrix to the inter
membrane compartment.
4] A pH (decreases/more acidic, as H+ increases) and electrical gradient
are created across the membrane
5] ATP synthase generates ATP, by phosphorylating ADP, powered by the
diffusion of H+ moving down its concentration gradient.
Stages of
Cellular
Respiration
Where is it
occurring?
SITE
Glycolysis
Cytoplasm
What is
needed?
‘INPUTS’
1
Glucose
What is
produced?
How
Is oxygen
many ATP required?
are
‘OUTPUTS’ produced?
2 Pyruvate
Net 2
No
2 ATP
2 NADH
Link Reaction
Mitochondria
2
pyruvate
2 Acetyl
CoA
0
No
2
No
34
Yes
2 Carbon
Dioxide
Krebs Cycle
(Citric Acid
Cycle)
Inner
Mitochondrial
Matrix
2 Acetyl
CoA
2 NADH
4 Carbon
dioxide
2 ATP
6 NADH
Oxidative
Phosphorylation
[Electron
Transport Chain
(ETC) +
Chemiosmosis]
2 FADH2
Inner
6 Oxygen 34 ATP
Mitochondrial (diatomic, 6 water
Membrane gaseous) molecules
10 NADH
2 FADH2
PHOTOSYNTHESIS - CHAPTER 10 REDUCTION=ENDERGONIC
9] PLANTS need to do several things in order to survive:
a) Collect light energy
Site: LEAVES
Structure, cross -section detail (draw & label):
1) CHLOROPLASTS - Photosensitive structures within leaves
(draw & label internal structures)
Outer membrane – phospholipid bilayer
Intermembrane space – area between the inner & outer
membrane
Inner membrane – another phospholipid bilayer
Stroma – fluid material that fills the area of the inner
membrane of the chloroplast
Thylakoids – suspended within the stroma are pancake-like
membranes called thylakoids which contain protein
complexes PSII & PSI cytochromes and other electron carriers
of the light-dependent reactions
Grana – stack of thylakoid membranes.
b) Store light energy – ultimately in the form of glucose and/or
starch, after photosynthesis is complete.
c) Obtain inorganic building blocks from the environment
Gas exchange - Need carbon dioxide, CO2 – uptake from (draw &
label): Stomata
Need water, H2O – uptake from: ROOTS
Other necessary nutrients: N, P, K, S, Mg (Chlorophyll), Fe
Uptake from: ROOTS
d) Produce all organic molecules necessary for growth
PRODUCTION OF CARBOHYDRATES DETAILED –> PHOTOSYNTHESIS
Write the overall reaction for photosynthesis:
10] PHOTOSYNTHESIS
a) LIGHT REACTION, photophosphorylation, using light energy to
generate ATP and NADPH to power the Calvin cycle – 7 steps
including Photosystem II & Photosystem I, and the splitting of
water:
Site: Chloroplast, specifically the THYLAKOID MEMBRANE
Reactants: Light from the Sun + Water
Process: Photosystem II with P680 [Non-cyclic Phosphorylation - 7
steps – PS II, Primary electron acceptor, ETC, phosphorylation, PS I,
NADPH, Splitting of water] [Cyclic Phosphorylation – when the
electrons energized in PS I are recycle – energized electrons from
PS I join with protein carriers and generate ATP as they pass along
an electron transport chain.] Photosystem I with P700
Products: Oxygen & NADPH, ATP to power the Calvin cycle
b) CALVIN CYCLE – the light independent or “DARK REACTION”,
fixes atmospheric CO2 into an organic molecule, glucose, that can
be used in biological systems.
Site: STROMA of the chloroplast
Reactants: ATP + 6 Carbon Dioxide
Process: Carbon fixation, carbon dioxide combines with Rubisco
to produce an intermediate PGA [3 carbon atoms so-called C3
photosynthesis.
Reduction – 12 ATP and 12 NADPH are used to convert 12 PGA to 12
G3P.
Regeneration – 6 ATP are used to convert 10 G3P to 6 RuBP
Carbohydrate Synthesis – remaining 2 G3P are used to build glucose
Products: 2 G3P --- precursor to Glucose
11] CHEMIOSMOSIS IN BOTH CHLOROPLASTS & MITOCHONDRIA –
the making of ATP that occurs when energy is stored in the form of a
proton concentration gradient across a membrane. The process in
chloroplasts is analogous to ATP generation in mitochondria:
12] C4 PROCESS SPATIAL SEPARATION of steps – forms a four carbon
compound as its first product MALATE
a) Occurs in what types of environments? HOT & DRY Why? Because
plants need to close their stoma for all or part of the day to
conserve water, thus reducing the amount of carbon dioxide they can
acquire. Examples: Tropical grasses – corn, sugarcane
13] CAM – Crassulacean Acid Modification [named after the plant family
Crassulaceae – the succulents] TEMPORAL SEPARATION of steps
Used by plants that close their stomata completely during the day
Examples: Cacti, pineapple, agave & succulents
COMPARE C4 and CAM
IN SUMMARY:
Both are characterized by:
preliminary incorporation of CO2 into organic acids, followed
by
transfer of CO2 to the Calvin cycle [ALL USE THE CALVIN CYCLE
eventually!]
BOTH are two evolutionary solutions to the problem of
maintaining photosynthesis with stoma that are partially or
completely closed on hot dry days.
14] CHEMOAUTOTROPHS
a) Define – use inorganic substance to fuel themselves – hydrogen
sulfide, elemental sulfur, ferrous iron, molecular hydrogen and ammonia,
to name a few. Most are bacteria or Archaea that reside in what we would
consider to be extremely harsh environments like hydrothermal vents.
b) Examples – methanogens, halophiles, sulfur oxidizers, ‘nitrogen
fixing bacteria”