CH3 Sec1
advertisement

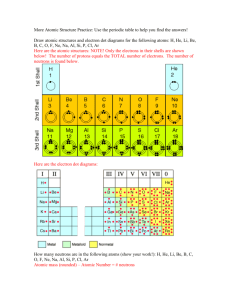
CH3 Sec1 Matter and Substances • Everything is made up of matter. • Matter is anything that has mass and takes up space. • Matter is made up of Atoms • Atoms are the smallest unit of matter • Atoms have positively charged center (core) surrounded my negatively charged region Atomic Structure Made up of three kinds of particles: Protons – (+) positively charged Electrons – (-) negatively charged Neutrons – No charge •Protons and neutrons make up the core (center or nucleus) •Electrons move around the nucleus in an electron cloud Elements • Are made up of atoms that have the same number of protons • Isotopes are atoms of an element that have different numbers of neutrons Periodic Table Determining # of electrons, protons, and neutrons in atom • Atomic # = # protons • Protons = electrons • Atomic mass = p + n + e Determining # of electrons, protons, and neutrons in atom • Atomic # = # protons (27) • Protons = electrons (27) • Atomic mass = p + n + e – 32 neutrons – e is 1/200th size of p & n does not impact mass • Now you try a few…. Chemical Bonds • Electron cloud of an atom may have levels • Innermost level can hold only 2 electrons • Levels farther from the nucleus can usually hold 8 electrons • Electrons in the outermost level (shell) are called valence electrons, this is where atoms bind Chemical Bonds • Opposites attract • Atoms come together and form a chemical bond • Making the atoms more stable they want an even number of electrons in each level, forming a neutral charge • A compound are created when atoms from two or more different elements bond • Ions are atoms that have a + or - charge as a result of gaining or losing electrons Covalent Bond and Ionic Bond Covalent Bond Electron pairs are shared Ionic Bond Electrons are transferred Molecules are produced Ions are produced Bond is the attraction for shared electrons Bond is the attractions between ions of the opposite charge Example: Water Molecule Example: Table Salt Na+ClH20 Table salt is an Ionic Compound because the positive Na (sodium) ion loses an electron to negative Cl (chloride) ion. Positive to Negative. ---- + Polarity • Molecules charges on opposite ends are polar – One end is positive the other opposite end is negative. Solubility • Polar molecules that attract opposite charges • Water dissolves polar molecules such as salt and sugar – Nature’s Solvent • Not able to break the bonds of non-polar substances like wax, oil, or grease – Important characteristic of our cell membranes Hydrogen Bonds • Play important role • Holds DNA strands together • Type of weak covalent bond