lecture 16
advertisement

What’s coming up???
•
•
•
•
•
•
•
•
•
•
•
•
•
Oct 25
Oct 27
Oct 29
Nov 1
Nov 3,5
Nov 8,10
Nov 12
Nov 15
Nov 17
Nov 19
Nov 22
Nov 24
Nov 26
The atmosphere, part 1
Midterm … No lecture
The atmosphere, part 2
Light, blackbodies, Bohr
Postulates of QM, p-in-a-box
Hydrogen and multi – e atoms
Multi-electron atoms
Periodic properties
Periodic properties
Valence-bond; Lewis structures
VSEPR
Hybrid orbitals; VSEPR
Hybrid orbitals; MO theory
Ch. 8
• Nov 29
MO theory
Ch. 12
•
•
bonding wrapup
Review for exam
Ch. 11,12
Dec 1
Dec 2
Ch. 8
Ch. 9
Ch. 9
Ch. 9
Ch.9,10
Ch. 10
Ch. 10
Ch. 11
Ch. 11
Ch. 11, 12
Ch. 12
The Final Exam
• December 13 (Monday)
• 9:00 – 12:00
• Cumulative (covers everything!!)
• Worth 50% of total mark
• Multiple choice
The Final Exam
• From my portion, you are responsible for:
– Chapter 8 … material from my lecture notes
– Chapter 9 … everything
– Chapter 10 … everything
– Chapter 11 … everything
– Chapter 12 … everything except 12.7
The Final Exam
• You will need to remember
– Relationship between photon energy and
frequency / wavelength
– De Broglie AND Heisenberg relationships
– Equations for energies of a particle-in-a-box
AND of the hydrogen atom
– VSEPR shapes AND hybribizations which
give them
COMBINATION OF ORBITALS
Remember, when we take linear combinations
of orbitals we get out as many as we put in.
Here, the sum of the 2 orbitals
1sA + 1sB = MO1
builds up electron density between nuclei.
90% probability
COMBINATION OF ORBITALS
1sA – 1sB = MO2
results in low electron density between nuclei
1sA + 1sB = MO1
builds up electron density between nuclei.
THE MO’s FORMED BY TWO 1s
ORBITALS
SUBTRACTION gives an….
Energy more positive than average of original orbitals
E
Energy of a
1s orbital in
a free atom
A
s1s*
s1s
B
Energy of
a 1s orbital
in a free
atom
ADDITION gives an….
Energy more negative than average of original orbitals
The bonding in H2
H
H2
H
s1s*
E
1s
1s
s1s
H
H2
H
s1s*
E
1s
1s
s1s
H2: (s1s)2
He2: (s1s)2(s1s*)2
The He2 molecule is not
a stable species.
He
He2
He
s1s*
E
1s
1s
s1s
The bonding effect of the (s1s)2 is cancelled by the
antibonding effect of (s1s*)2
BOND ORDER
A measure of bond strength and molecular stability.
If # of bonding electrons > # of antibonding electrons
the molecule is predicted to be stable
Bond
order
= 1/2 {
= 1/2 (n
# of bonding
# of antibonding
–
electrons(nb)
electrons (na)
b
- na)
A high bond order indicates high bond
energy and short bond length.
Consider H2+,H2,He2+,He2……….
}
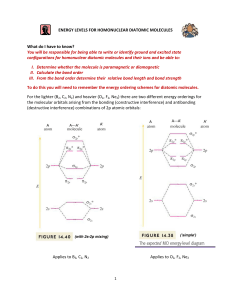
First row diatomic molecules and ions
H2
H2+
He2+
He2
Magnetism
Dia-
Para-
Para-
—
Bond order
1
½
½
0
Bond energy
(kJ/mol)
436
225
251
—
Bond length
(pm)
74
106
108
—
s1s*
E
s1s
ELECTRONS FOR DILITHIUM
s2s*
2s
2s
s2s
E
Li2
s1s*
1s
Put the electrons in the MO’s
1s
s1s
Electron configuration for DILITHIUM
s2s*
Li2
(s1s)2(s1s*)2(s2s)2
2s
2s
s2s
E
Bond Order = 1/2 (nb - na)
= 1/2(4 - 2) =1
1s
1s
s1s
A single bond.
Li2
(s2s)2
Only valence orbitals contribute to
molecular bonding
Li
Li2
Li
s2s*
E
2s
2s
s2s
(s1s)2(s1s*)2 assumed
Electron configuration for DIBERYLLIUM
Be2
Be
Be2
Be
s2s*
E
2s
2s
s2s
Configuration:
(s2s)2(s2s*)2
Bond order?
B2
The Boron atomic configuration is
1s22s22p1
So we expect B to use 2p orbitals to
form molecular orbitals.
How do we do that???
Combine them by addition and subtraction
BUT … remember there are 3 sets of p-orbitals
to combine
s molecular orbitals
SUBTRACT
s2p
-
* antibonding
-
+
+
ADD
-
+
s2p bonding
-
The p molecular orbitals.
p2p* antibonding
SUBTRACT
ADD
+
-
-
+
+
-
p2p bonding
The p molecular orbitals.
The M.O.’s formed by p orbitals
The p do not split as much as the s
because of weaker overlap.
s2p*
p2p*
2p
E
2p
p2p
s2p
Combine this with
the s-orbitals…..
Expected orbital splitting:
The p do not split
as much
because of
weaker overlap.
s2p*
p2p*
2p
p2p
s2p
E
s2s*
2s
2p
But the s and p
along the
internuclear axis
DO interact
2s
s2s
This pushes the s2p up..
MODIFIED ENERGY LEVEL DIAGRAM
s2p*
p2p*
2p
E
s2p
p2p
s2s*
2s
s interaction
2p
Notice that the s2p and p2p
have changed places!!!!
2s
s2s
Now look at B2...
Electron configuration for B2
s2p*
B is [He] 2s22p1
p2p*
2p
E
s2p
p2p
2p
s2s*
2s
2s
s2s
Electron configuration for B2:
s2p*
(s1s)2(s1s*)2(s2s)2(s2s*)2(p2p)2
p2p*
2p
E
s2p
p2p
s2s*
2s
2p
Abbreviated configuration
(s2s)2(s2s*)2(p2p)2
2s
s2s
Bond order 1/2(nb - na)
s2p*
= 1/2(4 - 2) =1
p2p*
2p
E
s2p
p2p
2p
s2s*
2s
2s
s2s
Molecule is
predicted to be
stable and
paramagnetic.
Li2
E
2p
SECOND ROW DIATOMICS
B2 C2
N2
O2
s2p*
p2p*
s2p
p2p
F2
s2p*
p2p*
2p
2p
p2p
2p
s2p
s2s*
s2s*
2s
2s
s2s
2s
2s
s2s
Back to Oxygen
O
O
12 valence electrons
BO = 2 but PARAMAGNETIC
s2p*
p2p*
E
s2p
p2p
s2s*
s2s
BUT REMEMBER …THE LEWIS
STRUCTURE WAS DIAMAGNETIC
Second row diatomic molecules
NOTE SWITCH
OF LABELS
B2
C2
N2
O2
F2
Magnetism
Para-
Dia-
Dia-
Para-
Dia-
Bond order
1
2
3
2
1
Bond E. (kJ/mol) 290
620
942
495
154
Bond length(pm) 159
131
110
121
143
s2p*
p2p*
E
s2p or p2p
p2p or s2p
s2s*
s2s
Example: Give the electron configuration and bond order
for O2, O2+ , O2- & O22-. Place them in order of bond
strength and describe their magnetic properties.
Step 1:Determine the number of valence electrons in each:
O2 :
6 + 6 = 12
O2+ : 6 + 6 - 1 = 11
O2– : 6 + 6 + 1 = 13
O22- : 6 + 6 + 2 = 14
Step 2:
Determine the valence electrons configurations:
O2
O 2+
O 2–
s2p*
E
p2p*
p2p
s2p
s2s*
s2s
O2 :
O 2+ :
O 2– :
(s2s)2(s2s*)2 (s2p)2(p2p)4 (p2p*)2
O22-
O2
O 2+
O 2–
s2p*
E
p2p*
p2p
s2p
s2s*
s2s
O2 :
(s2s)2(s2s*)2 (s2p)2 (p2p)4(p2p*)2
O2+ : (s2s)2(s2s*)2 (s2p)2 (p2p)4(p2p*)1
O2– : (s2s)2(s2s*)2 (s2p)2 (p2p)4(p2p*)3
O22- : (s2s)2(s2s*)2 (s2p)2 (p2p)4(p2p*)4
O22-
Step 3:
Determine the bond orders of each species:
O2
s2p*
E
p2p*
p2p
s2p
s2s*
s2s
O2 :
B.O. = (8 - 4)/2 = 2
O2+ : B.O. = (8 - 3)/2 = 2.5
O2– : B.O. = (8 - 5)/2 = 1.5
O22- : B.O. = (8 - 6)/2 = 1
O 2+
O 2–
O22-
HETERONUCLEAR DIATOMICS
E
2p
s2p*
s2p*
p2p*
s2p
p2p
p2p*
2p
2p
p2p
2p
s2p
s2s*
s2s*
2s
2s
s2s
2s
2s
s2s
NITRIC OXIDE (NO)
Number of valence electrons:
5 + 6 = 11
USE THE MO DIAGRAM FOR HOMONUCLEAR
DIATOMIC MOLECULES WITH s-p INTERACTION
AS AN APPROXIMATION FOR < 12 ELECTRONS
s2p*
p2p*
E
s2p
p2p
s2s*
s2s
Put the electrons in…..
NITRIC OXIDE (NO)
s2p*
p2p*
E
s2p
p2p
s2s*
Bond order
83
2.5
2
Molecule is stable
and paramagnetic.
Experimental data agrees.
s2s
NO+ and CN-
NO+:
Number of valence electrons:
5 + 6 - 1 = 10
CN–:
Number of valence electrons:
4 + 5 + 1 = 10
ISOELECTRONIC
s2p*
p2p*
E
s2p
p2p
s2s*
s2s
Bond order
82
3.0
2
TRIPLE BOND
Ions are both stable
and diamagnetic.
Experimental data agrees.
CAN NeO EXIST?
How can we answer this question?
Check bond order……...
NeO:
Number of valence electrons:
s2p*
p2p*
E
p2p
s2p
s2s*
s2s
Bond order
8 + 6 = 14
86
1.0
2
SINGLE BOND
Therefore ….
It could exist.