File
advertisement

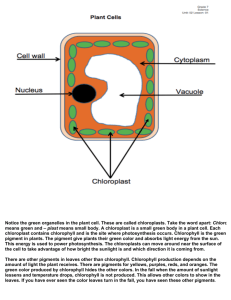
Chromatography Lab Objective: To separate and identify the colors of the plant extracted pigments by using the paper chromatography technique, indicate the distance of each pigment from the starting line, and discuss about the result and the fluorescence emitted. Introduction: In photosynthesis process, light reaction occurs to produce ATP and NADPH which later be converted carbohydrate in Calvin cycle reaction. The production cannot exist without energy input. The main source of energy comes in a type of solar energy. The energy is captured by three major pigments in plant. Those are chlorophyll A, chlorophyll B, and carotenoid. These pigments absorb different wavelength, which calls the absorption spectrum. These pigments absorb visible light, and the ones that do not get absorbed will reflect back. That is why we see different colors. Hypothesis: Three different colors are expected to appear on a watercolor paper. Those are yellow from carotenoid, green and light green from chlorophyll A and chlorophyll B. The color which will move furthest is yellow. It is because of the non-polarity of the chlorophyll. Three pigments have different polarity, which causes them to move in various distances. Materials: - 1 x 250 mL Beaker - A strip of watercolor paper - Glass pipette - Scissors - Ruler - Pencil - 9:1 petroleum ether-acetone solvent Procedure: 1. Cut the watercolor paper paper into 2.5cm x 8cm. 2. 3. 4. 5. 6. 7. Draw one line 1.5cm away from the end of the paper and another one 0.5cm from the other side. Draw a dot in the middle of the 1.5cm away line. drop one drop of the extracted pigment at that dot and wait until it dries, repeat this method for 5 times (concentrating the dot). Pour 9:1 petroleum ether-acetone solvent into the beaker, makes sure that it is not high enough to touch the spot. Put the paper and leave it until the solvent is absorbed to the 0.5cm away line. Count the number of separate pigments and measure the distance from the starting line. Result: Identify pigments table Color Distance from the starting line Pigment Orange 5 cm Carotenoid Blue-greenish 3 cm Chlorophyll A Green 1.7 cm Chlorophyll B Analysis: 1. How many substances are on the Chromatogram? 4 which are: - The extracted chlorophyll from plant’s leaves - Solution (9:1 petroleum ether:acetone solvent) - The separated colours of pigment (Carotenoids, Chlorophyll A and Chlorophyll B) - pencil line 2. What colours are they? There are yellow from the carotenoid, blue color from the chlorophyll A and green from the chlorophyll B. 3. Which color moved furthest? The yellow color which is carotenoids moved furthest. Conclusion: From this experiment, we were able to prove that plants have more than one type of pigments due to the presence of more than one color on the testing paper and the result has supported the hypothesis. Each of the color on the testing paper represents 1 type of pigment in plants. The blue-greenish one represents chlorophyll a, green color represents chlorophyll b, and the orange-yellowish one represents carotenoids. In addition, more different pigments help plants to get more energy from different wavelength of light. For instance, Carotenoid is responsible for absorbing sunlight with wavelength that Chlorophyll a cannot. Fluorescence is a kind of radiation that emitted by an electron when it falls back to its ground state. We see Fluorescence in the extracted pigment because when we shine light through them, the electron gets to the excited state. After that it falls back to the ground state and releasing fluorescence. Unlike the extracted pigment, In the leaves electron doesn't fall back to the ground state because there is an acceptor that catches electron and take them through electron transport chain. So, that is why we cannot see fluorescence ,when we shine light through them. If this experiment is to be repeated, we might use chromatography paper instead of watercolor paper. Because it is better in extracting colors than drawing paper. If we had more time, we would also be able to define other possible pigments that could be extracted and do the experiment more about how they are different that make some of them move further except from their polarity. We would also repeat the experiment at least three times for data accuracy.