Heat Slides
advertisement

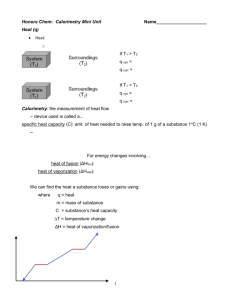
2 Temperature Temperature is a measure of how hot or cold an object is Ice Water Boiling Water Body Temp Room Temp Absolute Zero F = 9/5 C + 32 0o C 100o C 37o C 20o C -273o C 32o F 212o F 99o F 68o F -459o F K = C + 273 273o K 373o K 310o K 293o K 0o K Pressure and Temperature Pressure Gage Constant Air Volume Pressure Increases Pressure Decreases 0o C 100o C Ice Water Boiling Water Absolute Zero 100o C 0o C Boiling Water Ice Water Temperature (o C) -273o C 0o K Absolute Zero 373o K 273o K Quantity of Heat Quantity of Heat = Mass x Specific Heat x Temperature Change Q = m c ΔT 1 calorie is the amount of heat energy necessary to raise 1 gram of water 1 celsius degree. Specific Heat (Capacity) is the amount of heat energy necessary to raise 1 gram of a substance 1 celsius degree. 1 calorie = 4.18 joules 1 Calorie = 1,000 calories = 4,180 joules Calorimetry Hot Object Cold Object Heat Flow Heat Loss = Heat Gain mH cHΔTH = mC cCΔTC mH = mass of hot object cH = specific heat of hot object ΔTH = temperature change of hot object mC = mass of cold object cC = specific heat of cold object ΔTC = temperature change of cold object Sample Problem 1 500 g of metal at 90.0o C is placed in 200 g of water at 10.0o C. The specific heat capacity of the metal is .30 cal/goC. What is the final equilibrium temperature of the water and metal? mH = 500 g TH = 90.0oC CH = .30 cal/goC mC = 200 g TC = 10.0oC CC = 1.0 cal/goC TE = ? Hot Object Cold Object Heat Flow Heat Loss = Heat Gain mH cHΔTH = mC cCΔTC 500(.30)(90- TE) = 200(1.0)(TE - 10) 150 (90- TE) = 200 (TE - 10) 13500- 150TE = 200 TE - 2000 350TE = 15,500 TE = 44.3 oC Sample Problem 2 800 g of metal at 100.0o C is placed in 200 g of water at 10.0o C. The final equilibrium temperature of the water plus metal is 40.0oC. What is the specific heat capacity of the metal ? mH = 800 g TH = 100.0oC mC = 200 g TC = 10.0oC TE = 40.0oC CC = 1.00 cal/goC CH = __ cal/goC Hot Metal Cold Water Heat Loss = Heat Gain mH cHΔTH = mC cCΔTC 800 (CH)(100- 40) = 200(1.0)(40 - 10) 48,000 (CH) = 6,000 CH = .125 cal/goC Thermal Expansion Linear Expansion Lo ΔL ΔL = is proportional to ΔT ΔL = is proportional to Lo ΔL = depends on the type of material Volume Expansion ΔV = is proportional to ΔT ΔV = is proportional to Vo ΔV = depends on the type of material Linear Expansion The following materials are listed in alphabetical order. Arrange them in order of their rate of linear expansion per oC from lowest to highest. Aluminum Brass Copper Glass Steel 1. 2. 3. 4. 5. Aluminum 2.4 cm per km Brass 2.0 cm per km Copper 1.7 cm per km Steel 1.2 cm per km Glass 0.4-0.9 cm per km Expansion of Water Conductivity TH TC H L H is the heat flow between hot (TH) and cold ( TC) objects H increases as (TH – TC) increases H decreases as L increases H = depends on the type of material Top 7 Conductors of Heat The following metals are listed in alphabetical order. Arrange them in order of conductivity from best to worst. Aluminum Brass Copper Lead Mercury Silver Steel 1. 2. 3. 4. 5. 6. 7. Silver Copper Aluminum Brass Steel Lead Mercury 406 385 205 109 50 35 8 Top 10 Heat Insulators The following insulators are listed in alphabetical order. Arrange them in order of insulation from best to worst. Air Brick Concrete Cork Felt Fiberglass Glass Rock Wool Styrofoam Wood 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Styrofoam Air Cork Felt Fiberglass Rock Wool Wood Brick Concrete Glass .01 .024 .04 .04 .04 .04 .12 .6 .8 .9 Sea Breezes and Convection Currents Day During the day, the land is hotter and the air rises above the land and is replaced by the cooler air from above the water. Night During the night, the land cools faster and the air rises above the warmer water and is replaced by the cooler air from above the land. Convection Currents and the Earth Radiation • A good radiator of heat is a good absorber of heat. • Dark colored objects radiate and absorb heat better than light colored objects. • The rate of heat radiation increases with temperature. • The rate of heat radiation or absorption is proportional to the surface area of the object radiating or absorbing. Scaling and Heat Radiation S = 1 cm A= 6 cm2 A/V = 6 V= 1 cm3 S = 2 cm A = 24 cm2 A/V = 3 V= 8 cm3 S= 10 cm A = 600 cm2 V= 1000 cm3 A/V = .6 Change of State 2 Steam Ice Water Heat Heat 80 calories added to 1 g of ice at 0oC will convert the ice to 1gram of water at 0oC. 540 calories added to 1 g of water at 100oC will convert the water to 1gram of steam at 100oC. 3 Degrees Celsius Change of Phase 140 120 100 80 60 40 20 0 -20 0 -40 Water to Steam Ice to Water Ice Steam Series1 Water 20 40 60 time 80 100 The Human Body -Thermodynamic System External Work Food Heat Out Heat In Internal Work Energy Change = (Food +Heat In) – (External Work + Heat Out + Internal Work) Thermodynamics and Weight Loss External Work Food Heat Out Heat In Internal Work Energy Change = Food + Heat In – External Work - Heat Out - Internal Work If Energy Change = 0, then you maintain your body weight. If Energy Change > 0, then you gain weight. If Energy Change < 0, then you lose weight. If 1 once fat is equivalent to approximately 300 Calories of energy, we can calculate weight loss on a daily or monthly basis based on ΔU value.