Organic Chemistry
advertisement
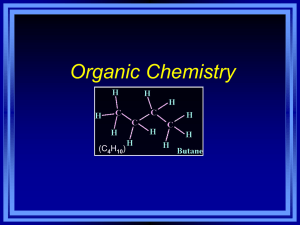
Organic Chemistry The Chemistry of Carbon Why call it Organic? • Fewer than 200 years ago, it was thought that only living organisms could synthesize carbon compounds. • So, they called carbon compounds “organic” How can carbon form 4 bonds? • Carbon is in the 4th group on the periodic table. • This means carbon has 4 valence electrons. • Carbon will want to bond with 4 other atoms to achieve the octet rule. Hydrocarbons • Hydrocarbons – any molecule that contains only hydrogen and carbon • Simple Hydrocarbons Methane and Ethane Fats are Hydrocarbons Group 1: Alkanes • Alkanes are hydrocarbons made of single bonds only. • Draw Methane • Draw Ethane • Draw Propane • Draw Butane Branched Alkanes – Each branch is called a SUBSTITUENT. Naming Branched Alkanes • Find the longest chain of carbons. • (8 – Octane) Naming Branched Alkanes • Number the longest chain starting with the end that will give substituents the smallest numbers. Naming Branched Alkanes • To name these substituents, start with the number. Add a dash. Add the type of substituent (drop the –ane ending and add the –yl ending) • 4-methyl, 4-ethyl, 5-propyl Naming Branched Alkanes • Add prefixes to indicate that the same substituent appeared more than once. • If there were two methyl groups on carbon 4, we would put 4,4 dimethyl Naming Branched Alkanes • List substituents in alphabetical order (ignore prefixes di, tri, tetra) • Combine all parts and use proper punctuation. Write the entire name without any spaces. Use commas to separate numbers and hyphens to separate numbers and words. 4-ethyl-4-methyl-5-propyloctance You Try! 4-ethyl-2-methylhexane Group 2: Alkenes • Alkenes are hydrocarbons that contain one or more double bonds. • The rules for naming is mostly the same, except for… – We drop the ending –ane and use –ene. – We use numbers to indicate the position of the double bond. 1-butene 3,3-dimethyl-1-butene 2-butene 4-methyl-2-pentene Group 3: Alkynes • Alkynes are hydrocarbons that contain one or more triple bonds. • The same rules apply with alkenes and alkynes. The only difference is that you drop the –ene ending and add –yne. Isomers • Isomers - compounds that have the same molecular formula, but have different structures. • Example: – C4H10 Stereoisomers • Stereoisomers are molecules in which the atoms are joined in the same order, but the positions of the atoms in space are different. Cis-Trans Isomers • Molecules with double bonds prevent rotating with respect to each other. • Because of this lack of rotation, the groups on either side of the double bond can have different orientations. Cis-Trans Isomers • Cis Configuration – Similar groups are on the same side of the double bond. Cis-Trans Isomers • Trans Configuration – Similar groups are on opposite sides of the double bond. Cis-Trans Significance • Cis-Trans configurations have different physical and chemical properties. Enantiomers • Enantiomers – Optical Isomers – Enantiomers have identical physical properties, but behave differently when they react. – Enantiomers have asymmetric carbons. Why are some enantiomers more effective as medicines? • Substrates and enzymes must fit correctly (like puzzle pieces) for the enzyme to function correctly. Certain enantiomers work better than their mirror image. Cyclic Hydrocarbons • Clycloalkane – Contains only single bonds Aromatic Hydrocarbons • An aromatic compound contains a benzene ring.