Intro to Radiobiology
advertisement

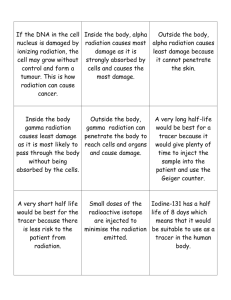
INTRODUCTION TO RADIOBIOLOGY By Professor Stelmark Sources of Ionizing Radiation Ionizing radiation is a special type of radiation that includes x-rays. Ionizing radiation is any type of radiation that is capable of removing an orbital electron from the atom with which it interacts. This type of interaction between radiation and matter is called ionization. Ionization occurs when an x-ray passes close to an orbital electron of an atom and transfers sufficient energy to the electron to remove it from the atom. The ionizing radiation may interact with and ionize additional atoms. The orbital electron and the atom from which it was separated are called an ion pair. The electron is a negative ion and the remaining atom is a positive ion. X-rays, gamma rays, and ultraviolet light are the only forms of electromagnetic radiation with sufficient energy to ionize. Some fastmoving particles (particles with high kinetic energy) are also capable of ionization. Examples are: alpha, beta +, beta - Many types of radiation are harmless, but ionizing radiation can injure humans. We are exposed to many sources of ionizing radiation . These sources can be divided into two main categories: 1. natural environmental radiation 2. man-made radiation. Natural environmental radiation consists of three components: cosmic rays, terrestrial radiation, and internally deposited radionuclides. • Cosmic rays are particulate and electromagnetic radiation emitted by the sun and stars. On Earth, the intensity of cosmic radiation increases with altitude and latitude. • Terrestrial radiation results from deposits of uranium, thorium, and other radionuclides in the Earth. The intensity is highly dependent on the geology of the local area. • Internally deposited radionuclides, mainly potassium-40 (40K), are natural metabolites. They have always been with us and contribute an equal dose to each of us. The largest source of natural environmental radiation is radon. Radon is a radioactive gas that is produced by the natural radioactive decay of uranium, which is present in trace quantities in the Earth. All Earth-based materials, such as concrete, bricks, and gypsum wallboard, contain radon. Radon emits alpha particles, which are not penetrating, and therefore contributes a radiation dose only to the lungs. Collectively, these sources of natural environmental radiation result in approximately 2 to 10 microroentgens (μR)/hr at waist level in the United States. This equals an annual exposure of approximately 20 milliroentgen (mR)/yr (0.2 milligray [mGy]/yr) along the Gulf Coast and Florida to 90 mR/yr (0.9 mGy/yr) or higher in the Rocky Mountain region. Diagnostic x-rays constitute the largest man-made source of ionizing radiation (39 mrem/yr) (0.39 mSv/yr). This estimate was made in 1990 by the National Council on Radiation Protection and Measurements (NCRP). More recent estimates put this source at nearly 320 mrem/yr (3.2 mSv/yr), with increases due principally to the increasing use of multislice spiral computed tomography (MSCT) and highlevel fluoroscopy. The currently accepted approximate annual dose resulting from medical applications of ionizing radiation is 50 mrem (0.5 mSv). In contrast to the natural environmental radiation dose, this level takes into account people who are not receiving a radiologic examination and those undergoing several within a year. Other sources of man-made radiation include nuclear power generation, research applications, industrial sources, and consumer items. Nuclear power stations and other industrial applications contribute very little to our radiation dose. Consumer products such as watch dials, exit signs, smoke detectors, camping lantern mantles, and airport surveillance systems contribute a few millirems to our annual radiation dose. The first x-ray fatality in the United States occurred in 1904. Unfortunately, radiation injuries occurred rather frequently in the early years. These injuries usually took the form of skin damage (sometimes severe), loss of hair, and anemia. Physicians and, more commonly, patients were injured, primarily because the low energy of radiation then available resulted in the necessity for long exposure times to obtain an acceptable radiograph. By about 1910, these acute injuries began to be controlled as the biologic effects of x-rays were scientifically investigated and reported. With the introduction of the Coolidge tube and the Snook transformer, the frequency of reports of injuries to superficial tissues decreased. The Pioneer Era (1895-1905)in which recognition of the gross somatic hazard occurred, and relatively simple means devised to cope. has already been briefly described, and needs only mention of an extraordinary x-ray protection pioneer W.H Rollins. William Herbert Rollins was a Boston dentist, who, during the period 1896-1904, made numerous original contributions to the emerging science of radiology.In 1896 July W.H Rollins developed a heavy glass plate to protect the eyes during dental radiography.Among his other contributions were several pertaining to radiation protection: • leaded tube housings, • collimators, • the development of high voltage tubes to limit patient dose. Rollins also performed a series of experiments that showed x-rays could kill guinea pigs. His experiments included exposure of a pregnant guinea pig which resulted in killing of the fetus and which led to Rollins expressing concern about the use of x-rays in pelvic exams of pregnant women. The Dormant Era (1905-1925) was a period of two decades in which applications of x-rays and radium along with the development of improved equipment seemed dominant. In the protection area, little overt progress was made, although latent effects of radiation exposures, particularly at low level, began to be recognized.One little known event that is of historical significance in health physics was reported at the October 1907 meeting of the American Roentgen Ray Society. At that meeting, Rome Vernon Wagner, an x-ray tube manufacturer, reported that in an effort to control his personal exposures, he had begun to carry a photographic plate in his pocket and to develop the plate each evening to determine if he had been exposed. This practice, which apparently did not come into widespread use until later, was clearly the forerunner of the film badge. Unfortunately, Wagner's concerns for his personal exposure came too late, for he had already developed cancer and died 6 months later in 1908. The year 1925 marked the start of what might be termed "Era of Progress" (1925-1945). In that year, Arthur Mutscheller,a German-American physicist,put forth the first tolerance dose or permissible exposure limit, equivalent to about 0.2 rem per day.Swedish physicist Rolf Sievert also put forth a tolerance dose10% of the skin erythema dose - in the same year. The other gains in radiation protection in this Era: the introduction of film badges for routine personnel monitoring, recognition of the genetic effects of xrays (for which Hermann Muller won the Nobel Prize in 1946), and the adoption of a unit for measuring radiation by the Second International Congress on Radiology in 1928. The definition and adoption of the Roentgen, as this unit was named, provided a physical basis for the quantitative measurement In 1942 Ernest O.Wollan ,a cosmic ray physicist at the University of Chicago formed a group named “health physicists”,for study and control radiation hazards.It was in the Manhattan District of the United States.Their activities included development of appropriate monitoring instruments, developing physical controls, administrative procedures, monitoring areas and radioactive waste disposal-- in short, the entire spectrum of modern day radiation protection problems. Many of the modern concepts of protection including the rem unit, which took into account the biological effectiveness of the radiation, and the maximum permissible concentration (MPC) for inhaled radioactivity were born from Manhattan District of united States. X-ray Interactions • • • • • Classical Compton Photoelectric Pair Production Photodisintegration Classical Interactions Classical interactions are also commonly known as coherent scattering or Thomson scattering. In this scattering event the incident x-ray photon interacts with an orbital electron of a tissue atom and changes direction. In this particular interaction the incident x-ray photon is of a rather low energy (generally less than 10 keV). When such low-energy incident photons interact with tissue atoms, they are not likely to ionize (remove orbital electrons from their shell). Instead, the atom absorbs the energy of this x-ray photon, causing excitation of the atom, and then immediately releases the energy in a new direction. Because the energy is reemitted in a new direction, it is now a scatter photon. It is of equal energy to the incident photon but travels in a new direction. Because of its low energy, most classical scatter photons are absorbed in the body through other interactions and do not contribute significantly to the image, but do add slightly to patient dose Compton scattering occurs throughout the diagnostic range. In this interaction, an incident x-ray photon enters a tissue atom, interacts with an orbital electron an outer-shell electron, and removes it from its shell. In doing so, the incident photon loses up to one third of its energy and is usually deflected in a new direction. This interaction ionizes the atom, making it unstable. Ionization in the body is significant because the atom is changed and may bond differently to other atoms, potentially causing biological damage. Compton scattering is one of the most prevalent interactions between x-ray photons and the human body in general diagnostic imaging and is responsible for most of the scatter that fogs the image. The probability of Compton scattering does not depend on the atomic number of atoms involved. Compton scattering may occur in both soft tissue and bone. The probability of Compton scattering is related to the energy of the photon. As x-ray photon energy increases, the probability of that photon penetrating a given tissue without interaction increases. However, with this increase in photon energy, the likelihood of Compton interactions relative to photoelectric interactions also increases. Photoelectric interactions occur throughout the diagnostic range (e.g., 20-120 kVp) and involve innershell orbital electrons of tissue atoms. For photoelectric events to occur, the incident x-ray photon energy must be equal to or greater than the orbital shell binding energy. In these events the incident x-ray photon interacts with the inner-shell electron of a tissue atom and removes it from orbit. In the process, the incident x-ray photon expends all of its energy and is totally absorbed Note that this absorption that constitutes photoelectric interactions contributes significantly to patient dose accrued with each diagnostic image. Although some absorption is necessary to create an x-ray image, it is the radiographer's responsibility to select technical factors that strike a balance between image quality (needed absorption and transmission of x-ray photons to produce a good image) and patient dose. The probability of photoelectric events is directly proportional to the third power of the atomic number of the absorber. If more photoelectric events are needed to make a particular structure visible on a radiographic image (i.e., tissues to be examined do not have high atomic number atoms), contrast agents such as barium or iodine are added. These agents have high atomic numbers and thereby increase the number of photoelectric events in these tissues. Protective shielding is another way of using photoelectric interactions. Lead has a very high atomic number and is used as a shielding material because the odds are great that photons will be absorbed by it. Pair production occurs only with very-high-energy photons of 1.02 MeV or greater. The interaction occurs when the incident x-ray photon has enough energy to escape interaction with the orbital electrons and interact with the nucleus of the tissue atom. In this interaction, two particles are produced: a positron (positively charged electron) and an electron. For these particles to exist, they must each have energy of 0.51 MeV (the energy equivalent of an electron). If the photon has energy greater than 1.02 MeV, it is shared between the two as kinetic energy. The last type of interaction between x-rays and matter is called photodisintegration. Photons with extremely high energies of more than 10 MeV may strike the nucleus of the atom and make it unstable. In photodisintegration the nucleus of the atom involved regains stability by ejecting a nuclear particle such as a proton, neutron, or alpha particle. Like pair production, photodisintegration does not occur in radiography because the energy levels required far exceed the kVp range used in diagnostic x-ray production. Other types of Ionizing Radiation • Alpha • Beta • Gamma Radioactivity Atoms with unstable nuclei are constantly changing as a result of the imbalance of energy within the nucleus. When the nucleus loses a neutron, it gives off energy and is said to be radioactive. Radioactivity is the release of energy and matter that results from changes in the nucleus of an atom. Alpha particles penetrability Alpha particles are highly ionizing (e.g. deposits energy over a short distance). Since alpha particles lose energy over a short distance, they cannot travel far in most media. For example, the range of a 5 MeV alpha particle in air is only 3.5 cm. Consequently, alpha particles will not normally penetrate the outermost layer of the skin. Therefore, alpha particles pose little external radiation field hazard. Shielding of alpha particles is easily accomplished with minimal amounts of shielding. Beta particles penetrability Beta particles are less ionizing than alpha particles. The range of beta particles depends on the energy, and some have enough to be of concern regarding external exposure. A 1 MeV beta particle can travel approximately 12 feet in air. Energetic beta particles can penetrate into the body and deposit dose to internal structures near the surface. Since beta particles are less ionizing than alpha particles, greater shielding is required. Low Z materials are selected as beta particle shields to take care of X-ray emissions associated with slowing down of beta particles while they travel in a medium. Gamma radiation penetrability Gamma rays are the least ionizing of the three forms discussed. A 1 MeV gamma ray can travel an average of 130 meters in air. Since gamma radiation can travel far in air, it poses a significant external radiation hazard. Further, if ingested, it may pose an internal radiation hazard. Shielding of gamma rays is normally accomplished with high atomic number materials such as lead. Carbon dating Nuclear Power Plant As of July 2008, there were more than 430 operating nuclear power plants and, together, they provided about 15 percent of the world's electricity in 2007. Of these 31 countries, some depend more on nuclear power than others. For instance, in France about 77 percent of the country's electricity comes from nuclear power. Lithuania comes in second, with an impressive 65 percent. In the United States, 104 nuclear power plants supply 20 percent of the electricity overall, with some states benefiting more than others. Despite all the cosmic energy that the word "nuclear" invokes, power plants that depend on atomic energy don't operate that differently from a typical coal-burning power plant. Both heat water into pressurized steam, which drives a turbine generator. The key difference between the two plants is the method of heating the water. While older plants burn fossil fuels, nuclear plants depend on the heat that occurs during nuclear fission , To make a nuclear fission it is necessary to bombard the split material with thermal neutrons. After the fission there there are two new atoms and and two or three free neutrons. This free neutrons make a fission of other atoms and so it is a nuclear chain reaction. Nuclear Power Station Nuclear medicine procedures use pharmaceuticals that have been labeled with radionuclides (radiopharmaceuticals). In diagnosis, radioactive substances are administered to patients and the radiation emitted is detected. The diagnostic tests involve the formation of an image using a gamma camera or positron emission tomography ,