6-ce50182-defects-me..
advertisement

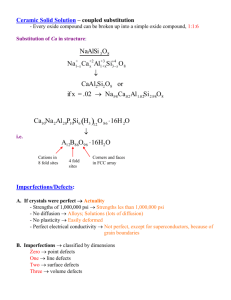
Materials Science Defects Defects in solids 1 mole Fe = 55.85g; V= 7.10 cm3 (D=7.87g/cm3); ~ 6 x 1023 atoms It would be nearly impossible to arrange so many atoms in exact 3D periodicity. So, formation of defects is not unexpected! ISSUES TO ADDRESS... • What types of defects arise in solids? • How do defects affect material properties? • Are defects undesirable? Defects in solids There is no such thing as a perfect crystal. What are these imperfections? Why are they important? Many of the important properties of materials are due to the presence of imperfections. Types of imperfections • Vacancy atoms • Interstitial atoms • Substitutional atoms Point defects (0D) • Dislocations Line defects (1D) • Grain Boundaries Area defects (2D) • Precipitates / cracks / porosity Volume defects (3D) Point defects (Intrinsic) • Vacancies -vacant atomic sites in a structure. Vacancy distortion of planes • Self-Interstitials- "extra" atoms positioned between atomic sites. selfinterstitial distortion of planes Intrinsic point defect concentration • Equilibrium concentration varies with temperature! No. of defects No. of potential defect sites. Activation energy -Q Nv = exp v kT N ÷ ÷ unitless; probability Absolute Boltzmann's constant Temperature Each lattice site is a potential vacancy site Point defects (Extrinsic) Two outcomes if impurity (B) added to host (A) - alloys: • Solid solution of B in A (i.e., random dist. of point defects) Insterstitial site can be calculated using simple geometry. OR Substitutional solid soln. (e.g., Cu in Ni) Interstitial solid soln. (e.g., C in Fe) Point defects (Extrinsic) - alloys • Solid solution of B in A plus particles of a new phase (usually for a larger amount of B) Second phase particle -different composition -often different structure. Point defects – summary Important for diffusion and mechanical properties Line defects in solids (1D) 1-D defect in which atoms are mispositioned. Line defects in Solids Linear Defects (Dislocations) Edge dislocation: Are one-dimensional defects around which atoms are misaligned extra half-plane of atoms inserted in a crystal structure b to dislocation line Screw dislocation: spiral planar ramp resulting from shear deformation b to dislocation line Burger’s vector, b: measure of lattice distortion Line defects in solids Screw dislocation Screw Dislocation Dislocation line Burgers vector b (a) Dislocations are visible in electron micrographs Dislocation motion • Incrementally breaking bonds • If dislocations don't move, deformation doesn't happen! (But it will fracture like a ceramic) Dislocations in materials • Metals: Disl. motion easier. -non-directional bonding -close-packed directions for slip. electron cloud • Covalent Ceramics (Si, diamond): Motion hard. -directional (angular) bonding • Ionic Ceramics (NaCl): Motion hard. -need to avoid ++ and -neighbors. ion cores Dislocation density Dislocation density: total dislocation length per unit volume of material … … or, the number of dislocations that intersect a unit area of a random section The dislocation density typically determines the strength of a material Metals (carefully solidified): 103 mm-2 Metals (heavily deformed): 109-1010 mm-2 Metals (heat treated): 105-106 mm-2 Ceramics: 102-104 mm-2 Single crystal silicon for ICs: 0.1-1 mm-2 Area defects (2D) Grain Boundaries regions between crystals transition from lattice of one region to that of the other slightly disordered low density in grain boundaries high mobility high diffusivity high chemical reactivity Area defects (2D) – grains Crystallites (grains) and grain boundaries. Vary considerably in size. Can be quite large ex: Large single crystal of quartz or diamond or Si ex: Aluminum light post or garbage can - see the individual grains Crystallites (grains) can be quite small (mm or less) – necessary to observe with a microscope. 0.75mm Fe-Cr alloy Volume defects (3D) Precipitates in Al alloys (deliberate) Cracks and damage (affects brittle materials) Porosity (in ceramics/ metals) Mechanical Properties ISSUES TO ADDRESS... • Stress and strain: What are they and why are they used instead of load and deformation? • Elastic behavior: When loads are small, how much deformation occurs? What materials deform least? • Plastic behavior: At what point does permanent deformation occur? What materials are most resistant to permanent deformation? • Toughness and ductility: What are they and how do we measure them? Stress-Strain curves Necking starts STRESS σUTS REGION I σYIELD l0 + le REGION II HARDENING OCCURS DISLOCATION MOTION AND GENERATION ! E REGION III σFAILURE or σFRACTURE Region I : Elastic Deformation Hooke’s Law Region II: Uniform Plastic Deformation Strain is uniform across material Region III: Non-uniform Plastic Deformation Deformation is limited to “neck” region l0 + l e + lp STRAIN l0 εYIELD εUTS Yield Strength : Comparison Metals/ Alloys Graphite/ Ceramics/ Semicond Polymers Composites/ fibers 2000 300 200 Al (6061) ag Steel (1020) hr Ti (pure) a Ta (pure) Cu (71500) hr 100 70 60 50 40 Al (6061) a 30 20 10 Tin (pure) ¨ dry PC Nylon 6,6 PET PVC humid PP HDPE LDPE in ceramic matrix and epoxy matrix composites, since in tension, fracture usually occurs before yield. 700 600 500 400 Ti (5Al-2.5Sn) a W (pure) Cu (71500) cw Mo (pure) Steel (4140) a Steel (1020) cd Hard to measure, 1000 Hard to measure , since in tension, fracture usually occurs before yield. Yield strength, sy (MPa) Steel (4140) qt Yield strength – summary Yield strength sy, the point at which stress is no longer proportional strain Above this level of stress permanent deformation occurs sy is a measure of resistance to plastic deformation (onset of dislocation motion in metals) Can be difficult to measure experimentally, but can use proportional limit (0.2% strain offset). Tensile Strength, TS • Maximum stress on engineering stress-strain curve. TS F = fracture or ultimate strength engineering stress sy Typical response of a metal Neck – acts as stress concentrator strain engineering strain • Metals: occurs when noticeable necking starts. • Polymers: occurs when polymer backbone chains are aligned and about to break. Tensile Strength : Comparison Metals/ Alloys Tensile strength, TS (MPa) 5000 3000 2000 1000 300 200 100 40 30 Graphite/ Ceramics/ Semicond Polymers C fibers Aramid fib E-glass fib Steel (4140) qt AFRE(|| fiber) GFRE(|| fiber) CFRE(|| fiber) Diamond W (pure) Ti (5Al-2.5Sn)aa Steel (4140)cw Si nitride Cu (71500) Cu (71500) hr Al oxide Steel (1020) ag Al (6061) a Ti (pure) Ta (pure) Al (6061) a Si crystal <100> Glass-soda Concrete Nylon 6,6 PC PET PVC PP HDPE 20 Composites/ fibers Graphite wood(|| fiber) GFRE( fiber) CFRE( fiber) AFRE( fiber) LDPE 10 wood ( 1 fiber) Tensile strength – summary Maximum stress that a structure can sustain Accompanied by plastic deformation and necking Design is to avoid this dangerous regime Ductility • Plastic tensile strain at failure: Lf - Lo x 100 %EL = Lo smaller %EL Engineering tensile stress, s larger %EL Lo Engineering tensile strain, e Examples: • Al 40% •Al2O3 <1% Definition: Degree of plastic deformation before failure. Lf Toughness • Energy to break a unit volume of material (J.m-3) • Resistance to crack propagation • Approximated by the area under the stress-strain curve. Engineering tensile stress, s small toughness (ceramics) large toughness (metals) very small toughness (unreinforced polymers) Engineering tensile strain, Brittle fracture: elastic energy Ductile fracture: elastic + plastic energy e Resilience - Elastic recovery Capacity of a material to absorb energy during elastic deformation and then on unloading to recover the energy Modulus of resilience U is strain energy per unit volume required to stress a material to the yield point i.e. area under stressstrain curve Resilience, Ur Ability of a material to store energy Energy stored best in elastic region Ur = ey 0 sde If we assume a linear stress-strain curve this simplifies to 1 Ur @ sy e y 2 Hardness Resistance of materials to localised plastic deformation (dents or scratches) Related to sy and E Quantitative analysis Small indenter forced into the surface and depth of indent is a measure of hardness E.g. Vickers hardness, small diamond indenter Arbitrary! Hardness • Large hardness means: --resistance to plastic deformation or cracking in compression. --better wear properties. apply known force measure size of indent after removing load e.g., 10 mm sphere D most plastics brasses Al alloys Smaller indents mean larger hardness. d easy to machine steels file hard cutting tools increasing hardness nitrided steels diamond Hardness: Measurement Summary • Stress and strain: These are size-independent measures of load and displacement, respectively. • Elastic behavior: This reversible behavior often shows a linear relation between stress and strain. To minimize deformation, select a material with a large elastic modulus (E). • Plastic behavior: This permanent deformation behavior occurs when the tensile (or compressive) uniaxial stress reaches sy. • Toughness: The energy needed to break a unit volume of material. • Ductility: The plastic strain at failure.