Elements and the Periodic table and Bohr Rutherford Diagrams
advertisement

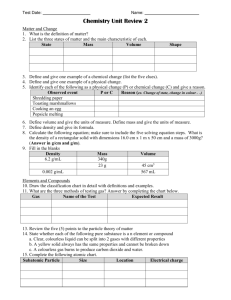
Elements and the Periodic Table Period • Period – Row of elements in periodic table Group • Group – column of elements in periodic table Alkali metals • Alkali metals – group one elements (not H) – Soft, highly reactive Alkaline earth metals • Alkaline earth metals – group two elements – Light, reactive Halogens • Halogens – group 17 elements – Most reactive group of periodic table Noble Gases • Noble Gases – group 18 elements – Very stable, rarely react with other chemicals Elements and the periodic table • 1) – green metalloids – Pink non-metals – Blue metals • 2) what is the exception to metals and nonmetals • 3) give two reason why this is where it is? Elements and the periodic table • 4) list four properties of metals –1 –2 -3 -4 • 5) List four properties of non-metals –1 –2 -3 -4 Elements and the periodic table • 6) Explain the term chemical family (group) Elements and the periodic table Family Alkali Metal Alkaline earth metal Noble gases halogens Location 2 elements in this family 2 properties of this family Elements and the Periodic Table • 8B) give a definition of each part- charge and location – Proton + , inside nucleus – Neutron neutral, inside nucleus – Electron - , inside nucleus Elements and the Periodic Table • 9) What is an ion? – A charged atom • 10) how is an ion formed? – Loses or gains electrons Bohr Rutherford Diagrams Bohr Rutherford Diagrams • Graphically illustrate the chemical structure of elements of the Periodic Table • Electrons are around nucleus in orbits • Each orbit can only contain a certain number of electrons (2 electrons in first orbit then 8 in each after) – # protons = atomic # – #neutrons = mass# - atomic# – #electrons = atomic # Bohr Rutherford Diagrams • Example Phosphorus Bohr Rutherford Diagrams • Example Fluorine Bohr Rutherford Diagrams Bohr Rutherford Diagrams Bohr Rutherford Diagrams Bohr Rutherford Diagrams