STEM7 Heat Expansion Lesson
advertisement

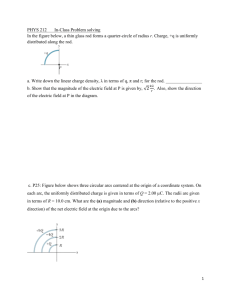
HEAT EXPANSION/thermal expansion Properties of matter STUDENT INFORMATION OBJECTIVES: Students will heat five kinds of metal rods to see how much each one expands. The students will discover why engineers and designers must consider expansion when planning metal products. RELATED CAREERS: Chemist Welder Metal Fabricator Construction Engineer Machinist KEY WORDS AND DEFINITIONS: 1. 2. 3. 4. Thermal – Pertaining to heat Expansion – To increase in length or size. Contraction – To shrink or reduce in length or size. Stress – To put pressure or strain on something. MATERIALS NEEDED: Thermal expander jig 8 inch aluminum rod 8 inch brass rod 8 inch copper rod 8 inch cold rolled steel rod 8 inch stainless steel rod Student worksheet Protective eye wear Propane torch Tongs or pliers Cooling pan Striker for torch Clock or watch RELATED INFORMATION: If you have ever been close enough to a railroad track to see where one length of track ends and another begins, you might have noticed that the gap between the two lengths of track appeared wider on cold days. On very hot days, the gap appeared to be much smaller. When the railroad track was designed, the designers realized that when metal was heated it would expand and increase in length. If the track lengths were placed snugly against one another, they would begin to bow and bend during the hot summer weather. Can you imagine what would happen to the train as it travels over the bow in the track? The Wheels of the train could slip off the track and cause the train to stop or even crash. Thermal expansion takes place in other areas as well. The explosion of the gases in an automobile engine creates great heat within the engine. Because a great amount of heat is generated, the engine parts will experience thermal expansion. Bridges made of metal, that get you from one side of the river to the other, and buildings constructed of steel and other materials, must be designed with expansion joints that allow the structure to expand on hot days, and contract on cold days. There are many types of building materials used in construction, and each one expands a t a different rate. JOB TO BE DONE: In this activity you will see examples of thermal expansion by conducting an experiment using the propane torch, various metal rods, the thermal expansion jig, protective eye wear, clock or watch, and paper and pencil to record your findings. SAFETY RULES FOR THIS ACTIVITY: Always wear safety glasses Do not touch the metal of=r the jig or the rods after they have been heated. Always work in an area that is free of fire hazards and materials. Get the instructor’s permission before you proceed with the activity. Do not paly around with the equipment; you may hurt yourself or others. If materials are broken or lost, report this to the instructor. PROCEDURE TO FOLLOW: 1. Acquire the activity worksheet. 2. Get the thermal expansion tester workstation. NOTE: There are five different types of ½ inch rods that you will test: A. Copper B. Aluminum C. Brass D. Stainless Steel E. Cold Rolled Mild Steel The number is stamped near the end of each rod. 3. Using the pliers, place the first rod on the expansion jig supports. 4. Turn the adjusting screw opposite the gauge so it pushes the rod lightly against the dial indicator. 5. Then turn the dial housing on the indicator until the zero on the dial face lines up with the needle. 6. While holding the striker in the front of the torch tip, open the torch valve just a VERY SMALL AMOUNT. Strike the striker to light the torch. NOTE: If the torch doesn’t light, you may have opened the valve to much. 7. As the torch heats the rod, watch the needle on the dial move up. NOTE: Each mark on the dial represents .001 of an inch. If the needle moves 23 marks or lines, this would be .023 or 23 thousands of an inch. 8. Time the heating of the rod for 2 minutes. The needle will have slowed down it’s movement. Turn off the torch. 9. On the worksheet, record the dial reading for the type of metal that was tested. 10. Use the pliers to remove the heated rod from the jig. Place it in its slot in the rack to cool. CAUTION: Be very careful that you do not burn yourself or others around you. 11. Loosen the adjusting screw. 12. Place the next rod on the support brackets and repeat the operation following steps 4-11. Continue this process until you have tested each of the rods and recorded the information on the student worksheet. 13. Leave the last rod tested on the jig and watch the needle move backwards as the material cools. When the dial indicator has returned to zero the rod has returned to the starting temperature 14. After you have completed the activity, be sure the torch is turned off. All the tools and equipment should be returned to the proper place 15. Complete the Activity worksheet. STUDENT WORKSHEET/EVALUATION HEAT EXPANSION Name_____________________________ Period______________ Date______________________ Directions: In the blank to the left, write the correct answer to the statement or question. ___________________________ 1. Expansion of rod #1 (copper) ___________________________ 2. Expansion of rod #2 (aluminum) ___________________________ 3. Expansion of rod #3 (brass) ___________________________ 4. Expansion of rod #4 (stainless steel) ___________________________ 5 Expansion of rod #5 (cold rolled steel) ___________________________ 6. Which rod showed the greatest expansion? ___________________________ 7. Which rod showed the least expansion? ___________________________ 8. Besides the railroad track, name one other steel structure that would need expansion joints. ___________________________ 9. Name two careers that deal with thermal expansion. ___________________________ Directions: write the answer to the question with one or more statements.10. Explain what causes metal to expand and contract. 10. ___________________________________________________________________________________________ Talking points while doing demo Fire Triangle Types of fire extinguishers Classes of fire extinguishers Fire prevention based on fire triangle and removing any of the components Alcohol and heat rising Residue left after burn is water 70/30 not the kind you drink warning cup and vacume demo Dial indicator and accuracy of .001 Human hair as example is .004 Metals that burn Gases /materials burn in different colors Like throwing Christmas wrap in fire Turn the torch upside down to show gas pressure affects Heat cone of the flame Materials/Metals conduct heat differently Function of torch tip and holes in tip Demonstrate air/oxygen added and convection by holding hands over holes What are we measuring with this apparatus Measure linear expansion Watch the temp rise and get a complete temp range through laser thermometer? Which heat tip puts out more heat, based on design. What else does the design of the tip do—spreads/shapes the heat This deals only with expansion of metals. Not expansion of other matter Expansion joint examples School hallway Bridge Road cutting Side walk and pushing up Railroad/trax 1. Heat and Temperature: Various temperature scales; Celsius, Fahrenheit, Kelvin. 2. Linear Thermal Expansion: The linear thermal expansion coefficient. Expansion leads to a need for expansion joints in railroad rails to avoid bulging on hot days. Thermal expansion is demonstrated by heating and cooling different metal rods. An important application of thermal expansion is bi-metals which are used in thermostats, safety devices and thermometers, as demonstrated. 3. Cubical Thermal Expansion: The fractional change in volume with temperature is given by the coefficient of cubical expansion. A mercury thermometer. 4. Shrink Fitting: Shrink fitting is a technique that makes use of the thermal expansion by heating one object (of two). After the two objects are assembled, they cool, and the fit is perfect and "for ever". 5. Cubical Thermal Expansion of Water: Water has a maximum density at 4 degrees Celsius; between 0 and 4 degrees Celsius the cubic thermal expansion coefficient is negative so the water expands as it cools below 4 degrees Celsius. The density of ice is about 8% lower than water, so ice cubes and icebergs float in water.