3S_alfanoD1 - shireen
advertisement

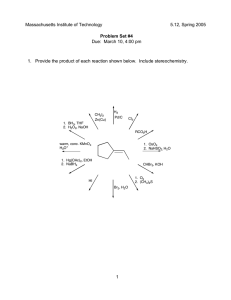
Acid and Base Reactions Shireen Rudina ingegneria biologica Equipe de dibattito Arrhenius Theory • In water, acids dissociate to form H+ and bases dissociate to form OHHCl + H2O H3O++ Cl- Concept Q: Which of these options are only acids? 1. HCl, NH3, HI 2. H2CO3, NaOH, HBr 3. H2CO3, NH4+, HCl 4. HI, KOH, HBr Arrhenius Strong/Weak Acids • Strong acid – Dissociates completely HCl HNO3 H2SO4 HBr HI HClO4 • Weak acids- everything else CH3COOH (aceto) NH4+ Arrhenius Strong/Weak Bases • Strong bases: LiOH NaOH KOH RbOH CsOH Ba(OH)2 Ca(OH)2 Sr(OH)2 • Weak bases: everything else In teams: Calculate the pH of a 0.05 M HBr solution 1. Is HBr a strong acid? How much does it dissociate? 2. What is [H+]? In teams: A 0.128 M solution of uric acid (HC5H3N4O3) has a pH of 2.39. Calculate the Ka of uric acid. 1. What is the equation for Ka? 2. What variables do we have? • http://www.youtube.com/watch?v=YqEK5ECc sDo • http://www.youtube.com/watch?v=vyOnGA0c mp0 • http://www.elmhurst.edu/~chm/vchembook/ 183neutral.html In teams: You have an unknown acid. When you put 1 M in water, you see a pH that corresponds to a specific dissociation percentage. • Find the Ka for the acid that dissociates: – Team 1: 1% – Team 2: 2% – Team 3: 0.1% • What acid do you have? Arrhenius Acid/Base Reaction Acid + Base = Salt + Water HBr + NaOH NaBr + H2O Neutralization A Neutralization Experiment Phenolphthalein: Purple if pH > 7 Colorless if pH<7 CO2 + H2O --> H2CO3 Ca(OH)2 + H2CO3 --> CaCO3 + 2 HOH What is the result of a neutralization reaction: NaOH + H2CO3? 1. NaH2 + H2O 2. NaH2 + H2O + CO3 3. NaHCO3 + H2O 4. NaCO3 + H2O Bronsted-Lowry Acid/Base Reaction Conjugate acids and bases





![pH = - log [H + ]](http://s2.studylib.net/store/data/005622524_1-002df1ea50d2a849b15deb604928664e-300x300.png)

