Chapter 14 2015 - Franklin College

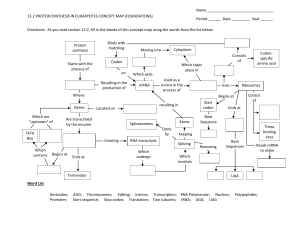
What is a gene?
• A sequence of DNA nucleotides that specifies the primary structure of a polypeptide chain (tells the cell how to make it)
• Genes-made of nucleotides
• Proteins-made of amino acids
• How does a nucleotide code (in the nucleus) specify an amino acid sequence
(in the cytoplasm)?
The Central Dogma
• DNA is transcribed into RNAcharacteristics of RNA
• RNA is translated into protein
• Advantages
• Exceptions
LE 17-4
DNA molecule
Gene 1
Gene 2
Gene 3
DNA strand
(template)
3
TRANSCRIPTION mRNA 5
TRANSLATION
Protein
Codon
Amino acid
5
3
The Genetic code-characteristics
• Triplet (3 nucleotides=codon=info for a specific amino acid);64 different codons (3 are stop codons)
• Universal
• Redundant (61 codons-20 amino acids)variability in third nucleotide of codon.
Advantages of a redundant code?
• Non-overlapping
• Exceptions (ciliates; mito/chloroplasts)
LE 17-5
Second mRNA base
Figure 17-06
Gene Expression
• If a gene is transcribed and the m-rna is translated (the gene is expressed); a protein is made. This often changes the phenotype of the cell that produces the protein.
• Differential gene expression is involved in embryonic development and cell specialization.
• Totipotency-each cell has the genetic information for an entire organism.
• Differential gene expression results in cell specialization (differentiation)
• Hormones often play a role in gene expression
Transcription
• The first step in gene expression
• Takes place in the nucleus
• Requirements
• A. RNA nucleotides
• B. DNA template (gene)
• C. Enzymes (RNA polymerase)
• Only one of the two DNA strands is copied
(template strand)
LE 17-7a-2
Promoter Transcription unit
5
3
Start point
RNA polymerase
DNA
Initiation
3
5
5
3
Unwound
DNA
RNA transcript
Template strand of DNA
3
5
LE 17-7a-3
Promoter Transcription unit
5
3
Start point
RNA polymerase
DNA
Initiation
3
5
5
3
Unwound
DNA
RNA transcript
Template strand of DNA
Elongation
Rewound
DNA
5
3
5
RNA transcript
3
3
5
3
5
LE 17-7a-4
Promoter Transcription unit
5
3
Start point
RNA polymerase
DNA
Initiation
3
5
5
3
Unwound
DNA
RNA transcript
Template strand of DNA
Elongation
5
3
Rewound
DNA
5
RNA transcript
3
Termination
3
5
3
5
5
3
5
Completed RNA transcript
3
3
5
LE 17-7b
Elongation
RNA polymerase
Non-template strand of DNA
RNA nucleotides
3
5
3
end
5
Newly made
RNA
Direction of transcription
(“downstream”)
Template strand of DNA
LE 17-8
5
3
5
3
Promoter
Eukaryotic promoters
TATA box
Start point Template
DNA strand
Several transcription factors
3
5
Transcription factors
3
5
Additional transcription factors
5
3
RNA polymerase II
Transcription factors
5
RNA transcript
3
5
Transcription initiation complex
Transcription-some important details
• Rate-30-60 nucleotides/second
• RNA polymerase (Many forms in eucaryotes, 3 basic types in bacteria: type I transcribes r-rna, type II-mrna, types III-trna)
• Promotors-(approximately 100 nucleotides)strong and weak promotors
• Eukaryotes-transcription factors needed to help
RNA polymerase to bind to TATA box (region of promotor 25 nucleotides upstream from initiation site).
RNA products of transcription
• m-rna
• t-rna
• r-rna
• sn-RNA (small nuclear)
• mi-Rna (micro)
• Si-rna (small interfering)
Not all genes code for proteins (m-rna)-Rrna and t-rna are obvious examples
• Actually, recent discoveries indicate that a large part of the eukaryotic genome is non-coding
RNA-Introns
• Small rna (micro rna and small interfering rna)play a crucial role in the regulation of gene expression involving both transcription and translation. Rna interference (Rnai)
• We’ll talk about regulation of gene expression in
Chapter 15.
Ribosomal RNA and ribosomes
• R-rna; one of two important components of ribosomes (other is protein-some of the proteins are enzymes). 60% r-rna; 40% protein.
• Ribosomes consist of 2 subunits
• Ribosomes needed to translate proteins
• “workbench of protein synthesis”
• Position t-rna (which is attached to a specific amino acid) on the codon of a m-rna
• Result is the synthesis of a protein (whose amino acid sequence is specified by the m-rna; which is transcribed from a gene)
LE 17-16b
P site (Peptidyl-tRNA binding site)
E site
(Exit site)
A site (AminoacyltRNA binding site)
E P A mRNA binding site
Schematic model showing binding sites
Large subunit
Small subunit
LE 17-16a tRNA molecules
Growing polypeptide
Exit tunnel
Large subunit
E P
A
5
mRNA
3
Computer model of functioning ribosome
Small subunit
Ribosomal –rna processing
T-rna
• Single polynucleotide chain folded into a complex 3-D shape (inter-chain H bonding). 75-80 nucleotides in length
• Binds a specific amino acid (involvement of amino-acyl-trna-synthetase
• Attaches to a specific m-rna codon via its anticodon
• How many different t-rna’s are there? 61?
Actually only 45 (wobble)
LE 17-14a
Amino acid attachment site
3
5
Hydrogen bonds
Anticodon
Two-dimensional structure
5
Amino acid attachment site
3
Hydrogen bonds
Anticodon
Three-dimensional structure
3
5
Anticodon
Symbol used in this book
“Charging” t-rna with its specific amino acid
• “charging” enzyme-amino acyl t-rna synthetase (20 different enzymes)
• Requires ATP
LE 17-15
Amino acid
Aminoacyl-tRNA synthetase (enzyme)
Pyrophosphate
Phosphates tRNA
AMP
Aminoacyl tRNA
(an “activated amino acid”)
Messenger Rna (m-rna)
• Contains the information for the primary sequence of a polypeptide chain
• Consists of codons
• Binds to ribosomes
• T-rna binds to m-rna (codon/anticodon)
LE 17-13
Polypeptide
Amino acids
Ribosome tRNA with amino acid attached
5
mRNA
Codons tRNA
Anticodon
3
Translation
• Codons (m-rna) read by ribosomes/t-rna
• Polypeptide chain produced
• 3 steps in translation-
• A. initiation
• B. elongation
• C. termination
• Translation is a process that consumes a tremendous amount of energy (ATP and GTP)
LE 17-16c
Amino end mRNA
E
Growing polypeptide
Next amino acid to be added to polypeptide chain tRNA
3
5
Codons
Schematic model with mRNA and tRNA
Translation-Initiation
• Initiation codon is AUG
• T-rna that bonds to AUG has an anticodon
UAC-this carries the amino acid methionine
• Requires a GTP molecule
• Requires proteins called initiation factors.
LE 17-17
Large ribosomal subunit
P site
Initiator tRNA
5
mRNA
Start codon mRNA binding site
3
Small ribosomal subunit
GTP GDP
E A
5
Translation initiation complex
3
Translation-Elongation
• The elongation cycle takes about 60 milliseconds
• During elongation, one m-rna codon is read and then the ribosomes moves down the message to the next codon.
• Binding of incoming t-rna to the A site of the ribosome requires a GTP
• Translocation-requires a GTP
LE 17-18
Amino end of polypeptide
Ribosome ready for next aminoacyl tRNA mRNA
5
E
P site
A site
3
2 GTP
2 GDP
E
P A
E
P A
GDP
GTP
E
P A
Translation-Termination
• When the ribosome reaches a termination codon, it causes the m-rna/ribosome complex to separate
• No t-rna binds to the termination codon.
• Release factors
• Newly made polypeptide chain is released
(folds into its characteristic 3-D shape)
LE 17-19
Release factor
Free polypeptide
5
3
3
5
Stop codon
(UAG, UAA, or UGA)
When a ribosome reaches a stop codon on mRNA, the A site of the ribosome accepts a protein called a release factor instead of tRNA.
5
3
The release factor hydrolyzes the bond between the tRNA in the
P site and the last amino acid of the polypeptide chain. The polypeptide is thus freed from the ribosome.
The two ribosomal subunits and the other components of the assembly dissociate.
Summary of energy demands for protein synthesis
• A rough estimate is that for every amino acid incorporated into a polypeptide chain, 3
ATP/GTP are consumed
A. Charging the amino acid (1 ATP)
B. Binding of incoming t-rna into the A site (1
GTP)
C. Translocation (1 GTP)
D. So a small protein (120 amino acids in length) would cost the cell 360 ATP/GTP to make (the equivalent of 12 glucose molecules going through aerobic cell respiration)
Polyribosomes
• A single ribosome can translate an average-sized polypeptide in about 1 minute
• Several ribosomes can translate the same message one after the other.
• Increases the efficiency of protein production
LE 17-20a
Growing polypeptides
Completed polypeptide
Incoming ribosomal subunits
Start of mRNA
(5
end)
End of mRNA
(3
end)
An mRNA molecule is generally translated simultaneously by several ribosomes in clusters called polyribosomes.
LE 17-20b
Ribosomes mRNA
0.1 m m
This micrograph shows a large polyribosome in a prokaryotic cell (TEM).
M-rna modifications
• Eukaryotic M-rna is modified extensively after transcription (while its still in the nucleus)
• These modifications include
A.Polyadenylationadded to 3’ end of m-rna
B. 5’ cap
C. Intron removal
M-RNA modifications
• Poly A tail
• A. added to the 3’ end of the m-rna
• B.30-200 Adenine nucleotides
• C. roles-regulation of transport of m-rna out of the nucleus; regulation of degradation of m-rna in the cytoplasm; helps m-rna attach to small ribosomal subunit
M-RNA modifications (continued)
• 5’ cap
• A. Modified guanine nucleotide stuck onto
5’ end of m-rna
• B. Roles- positioning of m-rna on small ribosomal subunit in initiation; protects m-rna from degradation
LE 17-9
Protein-coding segment
5
5
Cap 5
UTR
Start codon Stop codon
Polyadenylation signal
3
UTR
Poly-A tail
Introns
• Many eukaryotic genes have nucleotide sequences that don’t code for amino acids
(Introns)
• Introns separate coding sequences (exons).
Split genes
• Introns must be removed from the m-rna before it is translated (introns have nucleotide sequences that indicate splicing sites)
• Splicesomes are molecular machines that remove introns from m-rna
LE 17-11-1
5
Exon 1
RNA transcript (pre-mRNA)
Intron
Protein snRNA
Exon 2
Other proteins snRNPs
Spliceosome
LE 17-11-2
5
Spliceosome
Spliceosome components
5
Exon 1 mRNA
Exon 2
Cut-out intron
Significance of introns
• Why would chromosomes carry around extra DNA that isn’t used in the final mrna?
A. Expensive to maintain (energy).
B. Splicing out introns is a risky business
(what if it’s done incorrectly)
C. With these disadvantages, there must be an advantage or natural selection would not favor this arrangement
Benefits of Introns
• Evolution of protein diversity
• One gene can be alternatively spliced in a number of different ways to form several different types of m-rna (alternative splicing)
• Human antibody genes-about 500 genes can code for billions of different antibody molecules because of alternative splicing.
Figure 15.12
DNA
Exons
1 2 3 4
Troponin T gene
5
Primary
RNA transcript
1 2 3 4 5 mRNA 1 2 3 5
RNA splicing or 1 2 4 5
Summary of Transcription and Translation
Mutation
• An alteration in the nucleotide sequence of a DNA molecule (chromosome)
• Chromosomal mutations (duplications; deletions; inversions)
• Point mutations-alterations of one or a few nucleotides in a gene
Point mutations
• Spontaneous mutations
• Induced mutations
• Consequences of mutations-
• A. no effect-”silent mutations”
• B. harmful mutations-(may be lethal)
• C. beneficial mutations (rare)
Spontaneous mutations
• Base pairing errors; why aren’t they corrected by DNA repair enzymes?
• Effects:
• A. no effect-silent mutation (redundancy of genetic code; alteration of a non-critical amino acid)
• B. Positive effect-rare
• C. negative effect-missense mutations; nonsense mutations
LE 17-24a
Wild-type mRNA
5
Protein
Amino end
Stop
Carboxyl end
3
LE 17-24b
Base-pair substitution
No effect on amino acid sequence
U instead of C
Missense
Nonsense
U instead of A
A instead of G
Stop
Stop
Stop
Sickle cell anemia
• Results of a spontaneous missense mutation
• Result-altered hemoglobin molecule
• Effect-Depends on the environmental conditions and number of copies of the defective gene you inherited.
LE 17-23
3
Wild-type hemoglobin DNA
5
3
Mutant hemoglobin DNA
5
5
mRNA
Normal hemoglobin
3
5
mRNA
Sickle-cell hemoglobin
3
Induced mutations
• Caused by environmental damage
• Radiation (UV)- T-T dimers; excision repair enzymes; xerdoerma pigmentosa
• Chemicals-Common result-base pair addition or deletion
• Result of addition or deletion (frame shift mutation)-missense or nonsense
• Worst scenario-addition/deletion of 1 or 2 nucleotides at the beginning of a gene
LE 17-25
Wild type mRNA
Protein
5
3
Stop
Carboxyl end Amino end
Base-pair insertion or deletion
Frameshift causing immediate nonsense
Extra U
Stop
Frameshift causing extensive missense Missing
Insertion or deletion of 3 nucleotides: no frameshift but extra or missing amino acid
Missing
Stop
Mutations and Cancer
• Many mutations make cells cancerous
• 90% of known carcinogens are mutagens
• Ames test-screens potential chemicals for being carcinogens by seeing if they are mutagens
• Bacteria are the test subjects in the Ames test.
Employee Resource Manual
• Plasmids
• Restriction Endonucleases
• Agarose Gel Electrophoresis
Plasmids
• Small extrachromosomal pieces of DNA found in some bacterial species
• May carry additional genes (such as antibiotic resistance)
• Can be genetically modified and used as vectors for genetic engineering
PUC 18-Plasmid
Restriction Endonucleases
• Produced by some bacteria as a defense against virus infection
• Cleave DNA at specific bases sequences
(different recognition site for each different enzyme)
• Can be used to join DNA from 2 different sources (plasmid DNA and genomic DNA)
ECOR1
Agarose Gel Electrophoresis
• Separates DNA based upon size differences
• DNA is pulled through a gel by an electric current
• (-) charged DNA is pulled to the positive pole of the apparatus.
• Smaller pieces of DNA migrate through the gel faster than larger pieces of DNA
Agarose Gel Electrophoresis
Procell
Procell in Action
What is your first job assignment?
• Clone the H gene (use a bacteria to make copies of the gene for us)
What kind of bacteria do we use to clone the H gene?
• E.coli (lacZ(-), amp sensitve)
Where is the H gene located?
• Lambda virus
How do you get the cloned gene into the bacteria so the bacteria can copy it?
• Transform E.coli (lacZ(-), amp sensitve) with PUC 18-lambda plasmid (heat shock and osmotic shock)
Lambda virus genes have been inserted into the plasmids here
How do you get lambda genes into
PUC 18 plasmid?
• Incubate PUC-18 and lambda with EcoRI, ligate products
How many different plasmids do you get when you mix PUC 18 and lambda, both of which have been
ECOR1 and then ligated?
7
Would all 7 of the plasmids be recombinant (have lambda DNA)?
No!
How do you tell if bacteria have been transformed successfully with PUC-18 plasmid?
• They will grow on amp agar.
How can you distinguish whether plasmids that transformed bacteria were recombinant (lambda and PUC-18) or nonrecombinant (pUC-18 only)?
Plate the transformed cells on Xgal-amp agar
E.Coli transformed
With nonrecombinant
Plasmids (PUC-18)
E.Coli transformed
With recombinant
Plasmids (PUC-18/lambda)