practice test 1112 cp2 u5

Name ________________________________________________________ Period _____
Unit # 5 Practice Test: Intermolecular Forces
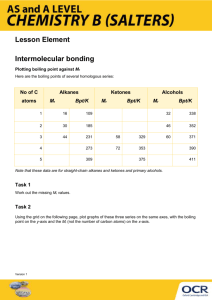
1. Explain the trend displayed in the boiling points in the table below (your answer must include reference to an intermolecular force and the reason for the trend).
Noble Gas
He
Ne
Ar
Kr
Boiling Point (K)
4.6
27.3
87.5
120.9
Xe 166.1
2. Which of the following substances is most likely to exist as a gas at room temperature and normal atmospheric pressure: P
4
O
10
, F
2
, AgCl, I
2
? WHY?
3. When the liquid metal mercury, Hg, is placed in a small tube, the meniscus actually curves upward, just the opposite of water. The reason is that - - -
A.
B. the cohesive force is greater than the adhesive force. the adhesive force is greater than the cohesive force.
C.
D.
E. the density of the mercury is much larger than water. the density of mercury is much greater where it is in contact with the glass. mercury is less volatile than water.
4. Explain why propane (C
3
H
8
) is a gas at room temperature and hexane (C
6
H
14
) is a liquid at room temperature.
5. Which of the following substances would you expect to have the lowest boiling point?
A. Barium chloride
B.
C.
D.
Ethanol, CH
3
CH
2
OH
Sodium acetate, NaC
2
H
3
O
2
Iron
E. Methane CH
4
6. Liquid A has a boiling point of 96˚C and liquid B has a boiling point of 68˚C. Which of the following is true?
A.
B.
C.
D.
A and B have the same volatility.
At 80 ˚C liquid A has a higher vapor pressure than B.
A is more volatile than B.
B must have weaker intermolecular forces of attraction.
7. Which of the following would exhibit hydrogen bonding?
A. CH
4
B. CH
3
OH C. H
2
S D. SiH
4
8 . What are some characteristics of high-viscosity liquids? Describe the properties itemized.
A. Strength of intermolecular forces
9. How do the boiling points of these substances compare? Rank them from the lowest to the boiling point. Defend your answer with scientific reasoning (reference specific intermolecular forces).
H
2
O, CH
3
Br, CH
4
, C
4
H
10
10. Below is a phase diagram for mystery liquid X. highest
Label the following on the phase diagram.
Critical point, Area of gas phase only, Triple point, Where subliming is in equilibrium with deposing
(opposite of subliming)
What is the normal (as in atmospheric pressure) melting point for mystery liquid X? ______________