chemicals and chemical change

CHEMICALS AND
CHEMICAL CHANGE
Chapter 5.1
BLM 5.1a, 5.1b
Chemicals and chemical Change
CHEMISTRY – is the study of matter, its changes and its properties
MATTER – is anything that has mass and takes up space
ATOM – the smallest particle of matter
PHYSICAL CHANGE – a change in a substance which does not produce a new substance.
• Easy to reverse
• Ex. Melting ice
CHEMICAL CHANGE – a change in a substance which does produce a new substance.
• Difficult to reverse
• Ex. Match burning
Classification of matter
Classification of matter
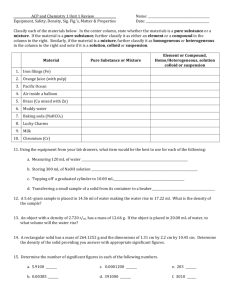
Classify each substance as 1) A pure substance or a mixture, and 2) An element, molecule, heterogeneous mixture or homogeneous: a) Glass of milk b) Spoonful of sugar c) A bar of gold d) Orange juice with pulp
Science 10 – Nov. 3 rd
Classify each reaction as a physical or chemical reaction: a) Butter melting b) Cheese molding c) Breaking a chocolate bar in half d) Burning toast
Science 10 – Nov. 3 rd
Classify each substance as 1) A pure substance or a mixture, and 2) An element, molecule, heterogeneous mixture or homogeneous: a) Glass of milk b) Spoonful of sugar c) A bar of gold d) Orange juice with pulp
Physical Reaction vs. Chemical
Reaction
Physical reactions are easy to reverse while chemical reactions are not.
• Ex “When ice melts is it easy to reverse that reaction? Burning a match?”
Physical Trait vs. Chemical Trait
Physical traits are either simple observations or things having to do with physical reactions.
• Color, smell, size, mass, boiling point, melting point.
Chemical traits have to do with chemical reactions.
• Will it catch on fire?
• Ability to react with other chemicals.
• Stable or unstable.
Science 10 – Nov. 1 st
1) Classify each as 1) Pure substance or mixture, then 2) Element, compound, homogeneous mixture or heterogeneous mixture .
a) b) c) d) e)
Sugar
Copper wire
Air
Orange juice w/ pulp
Orange juice w/o pulp
Science 10 – Nov. 1 st
2) Classify each as a physical change or a chemical change: a) b) c) d) e)
Food rotting
Breaking a carrot in half
Water boiling
Making toast
Dissolving salt into water
Science 10 – Nov. 1 st
3) Classify each as a physical trait or chemical trait: a) b) c) d) e)
Water freezes at 0 o C
Ethanol is a clear liquid
Gold does not react with acid
Aluminum is a malleable metal
Mercury metal is a liquid at room temperature
HOMEWORK QUESTIONS
Page 175
QUESTIONS: #1,2,4,5,6,7
Explain in your own words the difference between element, atom, molecule and compound.
Work on project
SOLUTIONS TO HOMEWORK
QUESTIONS
Q1 – classify each of the following as a pure substance or a mixture
A) soapy water
MIXTURE
B) hydrogen gas
PURE SUBSTANCE
C) sodium chloride
PURE SUBSTANCE
QUESTION 2
Classify each of the following as an element or a compound.
A) hydrogen
ELEMENT
B) potassium carbonate
COMPOUND
C) water
COMPOUND
D) Mg
ELEMENT
QUESTION 4
Classify each of the following as a physical property or a chemical property.
A) Gasoline is a clear pink solution
PHYSICAL PROPERTY
B) Gasoline burns in air
CHEMICAL PROPERTY
C) Water boils at 100 0 C.
PHYSICAL PROPERTY
D) electric current can split water into hydrogen and oxygen gases
CHEMICAL PROPERTY
QUESTION 5
When aluminum metal is added to hydrobromic acid, hydrogen gas and an aluminum bromide solution are formed.
A) What kind of change has occurred?
A chemical change has occurred because new substances were made.
QUESTION 5
When aluminum metal is added to hydrobromic acid, hydrogen gas and an aluminum bromide solution are formed
B) Which substances are the reactants and which are the products?
Reactants – Aluminum and Acid
Products – Hydrogen + Aluminum Bromide
QUESTION 6
Describe the chemical tests that can be used to identify the following gases.
A) hydrogen a flaming wooden splint causes a “pop”
B) Oxygen a glowing wooden splint relights (bursts into flame)
QUESTION 6
C) Carbon Dioxide
A burning wooden splint extinguishes, OR when the gas is bubbled through limewater, the limewater changes from a clear, colourless solution to a cloudy white liquid (a precipitate is formed)
QUESTION 6
d) Water Vapour
Changes the colour of cobalt chloride test paper from blue to pink
QUESTION 7
When sodium carbonate is added to water, the sodium carbonate dissolves.
When hydrochloric acid is added to the solution, the solution fizzes. What kinds of changes have occurred?
QUESTION 7 - ANSWER
When sodium carbonate dissolves in water it is a physical change
A chemical change happens when gas is formed after hydrochloric acid is added
( fizz / Bubbles )