Types of Reaction and Prediction Reactions
advertisement

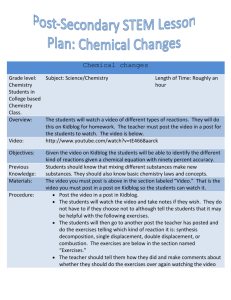
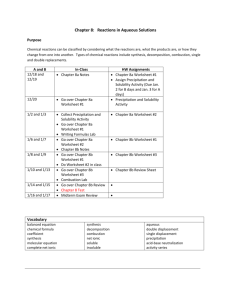
Types of Reaction and Predicting Reactions Concept Presentation Winnie Ho HS Science Instructors Janine Extavour & Marty Zatzman Goal of this presentation Grade 10 teachers: - Differences between Gr 10 and 11 and how to prepare our students Grade 11 teachers: - What Gr 10 students know and common roadblocks in this topic Teaching sequence in 11U • Unit 1- Matter, Chemical trends, and Chemical Bonding • Unit 2 - Chemical reaction Prior knowledge: - Ionic compounds and covalent molecules - Nomenclature - Balancing equation - Classifying reactions Curriculum expectation Grade 10 – Chemistry expectations on types of reaction C2.3 C3.5 investigate simple chemical reactions, including synthesis, decomposition, and displacement reactions, and represent them using a variety of formats (e.g., molecular models, word equations, balanced chemical equations) describe, on the basis of observation, the reactants in and products of a variety of chemical reactions, including synthesis, decomposition, and displacement reactions (e.g., reactions occurring when magnesium burns or in the production of oxygen from hydrogen peroxide; the reaction of iron and copper sulphate; reactions occurring when fossil fuels burn) Grade 11 – Chemical reaction (selected expectations on types of reaction) C2.4 predict the products of different types of synthesis and decomposition reactions (e.g., synthesis reactions in which simple compounds are formed; synthesis reactions of metallic or non-metallic oxides with water; decomposition reactions, in which a chemical compound is separated into several compounds) C2.5 predict the products of simple displacement reactions, using the metal activity series and the halogen series. C2.6 predict the products of double displacement reactions (e.g. formation of precipitates or gases; neutralization) C2.7 design an inquiry to demonstrate the difference between a complete and an incomplete combustion reaction C2. 10 plan and conduct an inquiry to demonstrate a simple displacement reaction, using elements from the metal activity series C3.1 identify various types of chemical reactions, including synthesis, decomposition, single displacement, double displacement, and combustion Comparison Grade 10 - Classifying all four reaction types + Neutralization Grade 11 - All four reaction types + Activity series + Solubility rules + Neutralization + Combustion + Nuclear reactions • Focus on lab inquiry • Prediction of products Diagnostic assessment 1. nitric acid + potassium hydroxide ____________+ water Type: ___________ 2. zinc + ___________ zinc nitrate solution + copper Type: ___________ 3. __________ acid + zinc carbonate zinc sulfate + water + carbon dioxide Type: ___________ 4. calcium + chlorine _____________________ Type: ___________ 5. magnesium + hydrochloric acid __________ + hydrogen Type: ___________ 6. propane + oxygen _____________________ + water Type: ___________ Reaction Type General Chemical Equation A + B AB Synthesis + Decomposition AB A + B + Sodium and Chlorine reacts to form Sodium chloride M + BC Single Displacement + AB + CD Double Displacement Combustion N + BC + + MC + BN AD + B + C + + CB + CxHy + _O2 _ CO2 +_ H2O Roadblock #1 Failure to connect symbols to the actual elements A + B AB A can represent an element, a diatomic molecule, or a compound. The ordering of the symbols is important. Can you find the mistakes? Macroscopic Microscopic Symbolic Roadblock #2 Predicting synthesis vs. combustion rxn Grade 10 – synthesis reaction Grade 11 –combustion reaction •Reaction of a substance with oxygen, producing oxides and energy (Nelson text) •Combustion of magnesium as an example of a synthesis reaction •Exothermic reaction Synthesis Reaction! Brad + Angelina Brangelina Tom + Katie TomKat Roadblock #3 Predicting single displacement reactions Zn(s) + CuSO4(aq)--> ZnSO4(aq) + Cu(s) Video Safety concerns: Demo: Li, Na, K with water Goggles, small quantity Alternatives M + BC MC + B M= metal element N + BC BN + C N= non-metal element Roadblock #4 Predicting double displacement reactions If a reaction does not produce a precipitate, a gas, or water, then it is not a DD reaction. Solubility table KOH(aq) + NH4Cl (aq)--> No Reaction KOH(aq) + NH4Cl (aq)--> no reaction Use of states symbols are important • More consistent usage of state symbols in grade 10 when writing out chemical equations. e.g., NaOH(aq) + HCl(aq) H2O (l) + NaCl (aq) Societal implications Synthesis reaction Rusting, Galvanization, Alloy Decomposition reaction Production of sodium using electrolysis of molten sodium chloride Single displacement reaction Extracting metals, smelting process, Bromine compound as fire retardant Double displacement reaction Geochemistry (solubility rules allows students to understand why some elements exist as compounds in nature), corrosion of statues Combustion Greenhouse effect Computer resources • Gizmos • Metals in aqueous solutions simulation Differentiated instruction • Computer simulation (modified versions of activities) • Video • Group work on making connections issues Thank you.