Various Career Options Available
advertisement

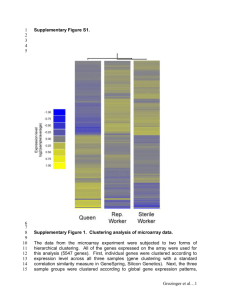
Introduction to Microarray Dr G. P. S. Raghava Molecular Biology Overview Cell Nucleus Chromosome Protein Gene (mRNA), single strand Gene (DNA) Measuring Gene Expression Idea: measure the amount of mRNA to see which genes are being expressed in (used by) the cell. Measuring protein would be more direct, but is currently harder. (RT) The Goals Basic Understanding – Arrays can take a snap shot of which subset of genes in a cell is actively making proteins – Heat shock experiments Medical diagnosis – Microarrays can indicate where mutations lie that might be linked to a disease. Still others are used to determine if a person’s genetic profile would make him or her more or less susceptible to drug side effects – 1999 – A genechip containing 6800 human genes was used distinguish between myeloid leukemia and lympholastic leukemia using a set of 50 genes that have different activity levels Drug design – Pharmaceutical firms are in a rush to translate the human genome results into new products Potential profits are huge First, though, they must figure out what the genes do, how they interact, and how they relate to diseases. – Evaluation, Specificity, Response Microarray Potential Applications Biological discovery – new and better molecular diagnostics – new molecular targets for therapy – finding and refining biological pathways Recent examples – molecular diagnosis of leukemia, breast cancer, ... – appropriate treatment for genetic signature – potential new drug targets History 1980s: antibody-based assay (protein chip?) ~1991: high-density DNA-synthetic chemistry (Affymetrix/oligo chips) ~1995: microspotting (Stanford Univ/cDNA chips) replacing porous surface with solid surface replacing radioactive label with fluorescent label improvement on sensitivity What is a DNA Microarray? genes or gene fragments attached to a substrate (glass) Tens of thousands of spots/genes =entire genome in 1 experiment A Revolution in Biology Hybridized slide Two dyes Image analyzed Gene Expression Microarrays The main types of gene expression microarrays: Short oligonucleotide arrays (Affymetrix); cDNA or spotted arrays (Brown/Botstein). Long oligonucleotide arrays (Agilent Inkjet); Terms/Jargons Stanford/cDNA chip Affymetrix/oligo chip one slide/experiment one chip/experiment one spot 1 gene => one spot one probe/feature/cell or few spots(replica) control: control spots 1 gene => many probes (20~25 control: two mers) fluorescent dyes control: match and (Cy3/Cy5) mismatch cells. Affymetrix Microarrays Raw image 1.28cm 50um ~107 oligonucleotides, half Perfectly Match mRNA (PM), half have one Mismatch (MM) Raw gene expression is intensity difference: PM - MM DNA Microarrays Each probe consists of thousands of strands of identical oglionucleotides – The DNA sequences at each probe represent important genes (or parts of genes) Printing Systems – Ex: HP, Corning Inc. – Printing systems can build lengths of DNA up to 60 nucleotides long – 1.28 x 1.28+ cm glass wafer Each “print head” has a ~100 m diameter and are separated by ~100 m. ( 5,000 – 20,000 probes) Photolithographic Chips – Ex: Affymetix – 1.28 x 1.28 cm glass/silicon wafer 24 x 24 m probe site ( 500,000 probes) – Lengths of DNA up to 25 nucleotides long – Requires a new set of masks for each new array type GeneChip The Process Poly-A RNA Cells AAAA 10% Biotin-labeled Uracil Antisense cRNA IVT L L L (In-vitro Transcription) cDNA Fragment (heat, Mg2+) Labeled fragments L L L Hybridize Wash/stain Scan Hybridization and Staining Biotin Labeled cRNA GeneChip Hybridized Array L L + L L L L L + L L L L L SAPE Streptavidinphycoerythrin Microarray Data First, the Problems: 1. The fabrication process is not error free 2. Probes have a maximum length 25-60 nucleotides 3. Biologic processes such as hybridization are stochastic 4. Background light may skew the fluorescence 5. How do we decide if/how strongly a particular gene is being expressed? Solutions to these problems are still in their infancy Affymetrix “Gene chip” system Uses 25 base oligos synthesized in place on a chip (20 pairs of oligos for each gene) RNA labeled and scanned in a single “color” – one sample per chip Can have as many as 20,000 genes on a chip Arrays get smaller every year (more genes) Chips are expensive Proprietary system: “black box” software, can only use their chips cDNA Microarray Technologies Spot cloned cDNAs onto a glass microscope slide – usually PCR amplified segments of plasmids Label 2 RNA samples with 2 different colors of flourescent dye - control vs. experimental Mix two labeled RNAs and hybridize to the chip Make two scans - one for each color Combine the images to calculate ratios of amounts of each RNA that bind to each spot cDNA microarrays Compare the genetic expression in two samples of cells PRINT cDNA from one gene on each spot SAMPLES cDNA labelled red/green e.g. treatment / control normal / tumor tissue HYBRIDIZE Add equal amounts of labelled cDNA samples to microarray. SCAN Laser Detector “Long Oligos” Like cDNAs, but instead of using a cloned gene, design a 40-70 base probe to represent each gene Relies on genome sequence database and bioinformatics Reduces cross hybridization Cheaper and possibly more sensitive than Affy. system Images from scanner Resolution – standard 10m [currently, max 5m] – 100m spot on chip = 10 pixels in diameter Image format – TIFF (tagged image file format) 16 bit (65’536 levels of grey) – 1cm x 1cm image at 16 bit = 2Mb (uncompressed) – other formats exist e.g.. SCN (used at Stanford University) Separate image for each fluorescent sample – channel 1, channel 2, etc. Processing of images Addressing or gridding – Assigning coordinates to each of the spots Segmentation – Classification of pixels either as foreground or as background Intensity determination for each spot – Foreground fluorescence intensity pairs (R, G) – Background intensities – Quality measures Images in analysis software The two 16-bit images (Cy3, Cy5) are compressed into 8bit images Display fluorescence intensities for both wavelengths using a 24-bit RGB overlay image RGB image : – Blue values (B) are set to 0 – Red values (R) are used for Cy5 intensities – Green values (G) are used for Cy3 intensities Qualitative representation of results Images : examples Pseudo-colour overlay Cy3 Cy5 Spot colour Signal strength Gene expression yellow Control = perturbed unchanged red Control < perturbed induced green Control > perturbed repressed Quantification of expression For each spot on the slide we calculate Red intensity = Rfg - Rbg (fg = foreground, bg = background) and Green intensity = Gfg - Gbg and combine them in the log (base 2) ratio Log2( Red intensity / Green intensity) Gene Expression Data On p genes for n slides: p is O(10,000), n is O(10-100), but growing, Slides Genes 1 2 3 4 5 slide 1 slide 2 slide 3 slide 4 slide 5 … 0.46 -0.10 0.15 -0.45 -0.06 0.30 0.49 0.74 -1.03 1.06 0.80 0.24 0.04 -0.79 1.35 1.51 0.06 0.10 -0.56 1.09 0.90 0.46 0.20 -0.32 -1.09 ... ... ... ... ... Gene expression level of gene 5 in slide 4 = Log2( Red intensity / Green intensity) These values are conventionally displayed on a red (>0) yellow (0) green (<0) scale. Biological question Differentially expressed genes Sample class prediction etc. Experimental design Microarray experiment 16-bit TIFF files Image analysis (Rfg, Rbg), (Gfg, Gbg) Normalization R, G Estimation Testing Clustering Biological verification and interpretation Discrimination Quality control (-> Flag) How good are foreground and background measurements ? – Variability measures in pixel values within each spot mask – Spot size – Circularity measure – Relative signal to background intensity – Dapple: b-value : fraction of background intensities less than the median foreground intensity p-score : extend to which the position of a spot deviates from a rigid rectangular grid Flag spots based on these criteria Replication Why? • To reduce variability • To increase generalizability What is it? • Duplicate spots • Duplicate slides Technical replicates Biological replicates Practical Application of DNA Microarrays DNA Microarrays are used to study gene activity (expression) – What proteins are being actively produced by a group of cells? “Which genes are being expressed?” How? – When a cell is making a protein, it translates the genes (made of DNA) which code for the protein into RNA used in its production – The RNA present in a cell can be extracted – If a gene has been expressed in a cell RNA will bind to “a copy of itself” on the array RNA with no complementary site will wash off the array – The RNA can be “tagged” with a fluorescent dye to determine its presence DNA microarrays provide a high throughput technique for quantifying the presence of specific RNA sequences Analysis and Management of Microarray Data Magnitude of Data – Experiments 50 000 genes in human 320 cell types 2000 compunds 3 times points 2 concentrations 2 replicates – Data Volume 4*1011 data-points 1015 = 1 petaB of Data Thanks