Robin Wood Desmond Tutu HIV Centre University of Cape Town
advertisement

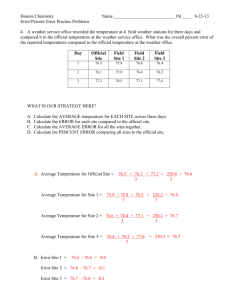
Cape Town AIDS Cohort-CTAC Electronic Data Sets Robin Wood Desmond Tutu HIV Centre University of Cape Town Photograph of Masiphumelele CTAC Data Sets • Natural history cohort 1984-2000 • Hospital-based ART trials 1996-2003 • Community-based ART programmes 2002-2003 • Web-based ART Register Natural History Cohort 1984-2000 • Epi-info based – Flow charts of 2088 outpatients – Laboratory, clinical data, OI prophylaxis • Pros – Doctors like flow charts of CD4 & WHO stages but not perceived non-useful data! • Cons – Data transfer, LTFU, missed hospitalisations & deaths required other data sources • Outputs – OI frequency, Kaplan-Meier survival Incidence rate/100 patient yrs CTAC: Incidence Rates of Opportunistic Infections 70 60 50 40 30 20 10 0 >350 200-350 <200 S. bact Toxo Cryptosp CMV MAI Crypto Wasting KS Enceph PCP HSV OC TB CD4+ T-cells/mm3 CTAC 2002 Hospital-based ART Trials 1996-2003 • Excel spread sheet of CRF data – Lab, clinical events, hospitalisations, deaths • Pros: – Trial data good quality, variable CD4 entry criteria, frequent visit intervals • Cons: – Extracting data laborious • Outputs: – Changed disease progression – Resource utilisation TB incidence rates & cases prevented per 100 pys of HAART 25 Cases /100pys 20 15 HAART Naïve 10 5 0 CD4 >350 CD4 200-350 CD4<200 WHO3&4 WHO 1&2 Cases saved 95% CI 1.3 9.4 11.3 (0.3-2.9) (3.8-14.3) (6.2-19.1) 18.8 (13.2-26.1) Badri, Wilson, Wood Lancet 2002 Progression-free survival of Triple vs NOART: WHO stage 1&2 Cumulative Proportion Surviving (Kaplan-Meier) Proportion of patients staying in stage1&2 Complete Censored stage1&2, p<0.00001 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 0 200 400 600 800 1000 1200 1400 1600 NOART Triple Time Category N Patients progressing to to higher stages Patients not progressing To higher stages P-value Triple 128 6 (4.7%) 122 (95.3%) <0.0001 NoART 140 52 (37.1%) 88 (62.9%) Progression-free survival of Triple vs NOART: WHO stage 3 Cumulative Proportion Surviving (Kaplan-Meier) Proportion of patients staying in stage3 Complete Censored Triple vs NOART, stage 3, p<0.001 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 200 400 600 800 1000 1200 1400 1600 1800 2000 2200 Triple NOART Time Category N Patients progressing to higher stages Patients not progressing to higher stages P-value Triple 85 13 (15.3%) 72 (84.7%) 0.014 No ART 155 46 (29.7%) 109 (70.3%) Progression-free survival of Triple vs NOART: WHO stage 4 Cumulative Proportion Surviving (Kaplan-Meier) Proportion of patients staying in stage4 Complete Censored Triple vs NOART, p=0.009stage 4, 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0 200 400 600 800 1000 1200 1400 1600 1800 Triple NOART Time Category N Patients progressing to higher stages Patients not progressing to higher stages P-value Triple 35 4 (11.4%) 31 (88.6%) 0.19 NoART 142 30 (21.1%) 112 (78.9%) HAART impact on hospitalisation (1995-2000) Days/yr (95% CI) OR (95% CI) AV cost PY (95% CI) HAART 0.7 (0.55-0.82) 0.2 (0.14-0.27) 525 (392-694) No ART 4.4 (3.9-4.9) 1 3,351 (2873-3888) HAART 1.4 (1.25-1.65) 0.13 (0.11-0.15) 1,113 (980-1293) No ART 10.9 (10.29-11.46) 1 8,512 (8065-8982) HAART 6.3 (5.58-6.98) 0.4 (0.38-0.49) 4,899 (4374-5471) No ART 14.6 (13.81-15.33) 1 11,412 (10824-12016) WHO Stages 1&2 WHO Stage 3 WHO Stage 4 Usapho Lwethu Community ART Data sources and follow up • Patient live in recruitment area & must have attended local HIV clinic for > 6 months • Household support by “Sizophila monitor” • Schedule 1 NNRTI, schedule 2 PI/r • Single referral hospital • Post mortem is requested on all deaths • Access to national mortality data base via “ID number” Community ART Programme 2002-2003 • Sequel database, data capture sheets – Laboratory, clinical, QOL, adherence data • Pros – Fixed visit schedule, LTFU rate low but deaths high, data captured on “lap-top” • Cons – Needs data administrator, active search for missing data • Outputs – Programme status reports of CD4, VL, survival etc Clinical Trials vs Community Clinic Clinical trials n=299 Community n=103 Median CD4 230 cells/ul 82 cells/ul WHO stage 3&4 49.8% 92.9% Treatment ART Regimen NNRTI 58.2% PI 39.1% Triple NRTI 2.7% NNRTI 100% Results at 16 weeks VL<400 copies/ml 75.5% (CI 71-80) 94.2% (CI 88-100) LTFU 5 1 Deaths 2 (0.69%) 7 (6.8%) Monthly drug costs of d4T, 3TC, EFV in study period from Sept 02-May 03 1400 $159 US$ cost calculated @ R8 per $ 1200 Rand 1000 800 $73 600 $41 400 200 0 Initial 4 months 8 months EFV 3TC d4T Proportional Costs for 250 Patients 18% 2% Drug Staff Monitoring Safety 48% 32% Cost per patient R8856 per annum Aerial Photograph of Masiphumele Pilot National ART Register • Web-based minimum data from ART programme linked to drug supply – ID number – Clinical stage – Current drug prescriptions • Linked to laboratory & pharmacy data – CD4 & Viral load monitoring – Drug monitoring Proposed Register Outputs • Patient retention, number on Rx, LTFU and discontinuing • Drug switching patterns • Time to first failure (MTCT exposure?) • Survival (benefit if compare with natural history model!) • Drug accountability, dispensing to patients & “leakage” • Laboratory outcomes CD4/viral suppression at time-points, resistance ACKNOWLEGEMENTS FUNDING AGENCIES NIH Cipra-SA BMS “Secure the Future” Foundation Hannan Crusaid Trusts Heiser Foundation Doris Duke Foundation UCT AIDS COHORT-CTAC Motasim Badri, Desmond Tutu HIV Centre, UCT Eduard Beck, McGill University, Montreal, Canada Linda-Gail Bekker, Desmond Tutu HIV Centre, UCT Gary Maartens, Dept of Medicine, Groote Schuur Hospital, UCT Kwesi Matoti, PAWC Gugulethu Catherine Orrell, Desmond Tutu HIV Centre, UCT Larissa Reader, Desmond Tutu HIV Centre, UCT Eve Sabotski, PAWC South Peninsula Douglas Wilson, Dept of Medicine, Somerset Hospital, UCT Photograph of Sizophila Team Sizophila Team April 2003