Primary Air Pollution
advertisement

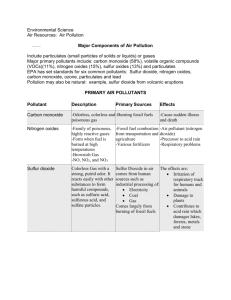
Primary Air Pollution “Bad Stuff” So What is Primary Air Pollution Anyways? DR. KNOW is ASHAMED! (and why do I care?) Recall that Air Pollutants are substances in the air at such concentration as to produce a harmful effect to humans, animals, vegetation, or materials. So What is Primary Air Pollution Anyways? DR. KNOW (and why do I care?) Air Pollutants are divided into two separate categories: Primary Air Pollutants is ASHAMED! Pollutants added directly to the atmosphere as a result of a given process. Secondary Air Pollutants Pollutants formed in the air as a result of chemical reactions. CO NOx Carbon Monoxide Nitrogen Oxides SOx Sulfur Oxides Particulates HCs Hydrocarbons The Big Five Primary Air Pollutants ? HC ? SOx So, No Cop, eH (C)Qi?CO NOx ? Particulates ? The Big Five Primary Air Pollutants :CΞO: As we already know, Carbon Monoxide is formed from the incomplete combustion of hydrocarbons in oxygendeprived environments. Carbon Monoxide is both toxic and undetectable to the human senses. Formed by 2 C(s) + O2(g) 2CO(g) FACT: 90 percent of all carbon monoxide production is from natural sources. SO What’s the big deal about man made carbon monoxide? Man Made CO is still a problem! Whereas natural sources of CO tend to be widely distributed, human production is localized and comes in periods of high emission rates Most of the human emission of carbon dioxide is produced in small areas, such as downtown areas. At rush hours, emissions are concentrated. In areas of high man made CO concentration, there is little natural reduction of CO. CO CO CO Nitrogen fixation ability is only affected at high levels of carbon monoxide. Plants are Happy. Nitrogen fixation ability is only affected at high levels of carbon monoxide. Hemoglobin, the oxygen carrier in animal blood cells, becomes unable to carry oxygen in the presence of carbon monoxide. Animals and humans can die of asphyxiation. CO: Metabolic Poison O2 bonds with hemoglobin to form oxyhemoglobin, which then distributes oxygen through the body. This process is easily reversible, so the hemoglobin can pick up more oxygen. CO bonds with hemoglobin to form carboxyhemoglobin. This bond, however, is much stronger than that of oxyhemoglobin, and the reaction is hence much more difficult to reverse, thus inhibiting the ability of the hemoglobin to carry oxygen. CO: Metabolic Poison Increasing carbon monoxide Effects of Carbon Monoxide Poisoning Heart beats faster Shortness of breath Headaches Fatigue, drowsiness Coma Respiratory failure Death No natural venues are so packed with CO that much more then headaches can occur. Coma and death from CO poisoning happens in enclosed areas. Death from carbon monoxide can be prevented by moving the victim from the enclosed area into fresh air. The reverse reaction of carbon monoxide and hemoglobin is facilitated by the presence of ample oxygen. The primary man made source of carbon monoxide is TO REDUCE PRODUCTION: theHOW combustion engine of CO the contemporary automobile, The Top Three by Methods but the problem is complicated the presence other 3. Investigation of new fuel burning techniques and engine pollutants. advancements 2. Development of new transportation technology 1. STOP DRIVING SO MUCH! Chemistry of a Cleaner Engine Thermal Exhaust Reactor Exhaust 2CO + O2 2 CO2 HC + O2 C2 + H2O(g) (not an exhaustive list) Air Less CO, Less Hydrocarbons But more greenhouse gasses Allows usage of leaner burning engines, which minimize nitrogen oxide emissions but have increased hydrocarbon and carbon monoxide outputs. Chemistry of a Cleaner Engine Catalytic Converters Exhaust Air compressor Cooling Apparatus Catalytic Converters have the amazing ability to convert two pollutants to mostly harmless substances. Platinumbased catalyst 2 CO +2 NO 2 CO2 +N2 They have, however, added six to eight hundred dollars to the price of a car and requires high octane, non-leaded, desulferized fuel. Nitrogen Oxides NOx There are eight known oxides of nitrogen. Some of these oxides natural, others are man made. B a s i c s The stability of the N-N triple bond in elemental nitrogen gas limits the natural production of nitrogen oxides. The three main Nitrogen Oxides present in the air are Nitrous Oxide, Nitric Oxide, and Nitrogen Dioxide. Formation: N2(g) +O2(g) 2NO(g) or N2(g) +O2(g) 2NO2(g) Nitrogen Oxides N2O NO NO2 Nitrous Oxide Dinitrogen Oxide Nitric Oxide Nitrogen Monoxide Nitrogen Oxide +1 +2 +4 Lewis Structure :N=N=O: :N=O: :O-N=O Properties Colorless, non-toxic, non-flammable gas with slight odor. Colorless, odorless, nonflammable, toxic gas Toxic, reddish-brown, non-flammable gas with strong odor Sources Decomposition of nitrogen containing compounds in soil and lightning. Majority from natural sources, twenty percent from the oxidation of nitrogen in automobiles. Secondary pollutant from nitric oxide Nitrogen Oxidation Number Odd number of electrons Odd number of electrons B a s i c s Nitrogen Oxides B a As with CO pollution, Human Activity produces only 10 Percent s of the total NOx pollution. Most human NO emissions are NO, but this is readily converted to NO in i the atmosphere. c The majority of NO and N O pollution is natural, from decomposition of nitrogen containing organisms, bacterial action and lightning strikes. s x 2 2 NOx NOx NOx NOx NOx NOx NOx Localization and high emission rates, however, exacerbate the problem. Nitrogen Oxides Both NO and NO2 are toxic to humans and animals. Similar effects, but NO2 is worse. Effects of NOx are most harsh in the young and the elderly. - Eye and nose irritation - Breathing difficulty, respiratory distress - Accumulation of fluid in the lungs (pulmonary edema) - Death NO or NO2 AAHHH (No living creatures were purposely harmed to study these effects. Study has come from low NOx pollution when smog is trapped near the earth) H e a l t h Nitrogen Oxides The catalytic converter and the thermal exhaust reactor discussed under CO pollution both do very well to prevent the NOx pollution. BUT in considering the control of NOx alone, it is important to note that the concentration of NOx creation is dependant upon the maximum temperature reached and the amount of oxygen gas available. Therefore, the engines that produce less NOx are those that have designs that either limit the amount of oxygen present in the reaction or recycle cooler exhaust so that the exhaust reaction is cooled. Just Say NO to Smog C o n t r o l This family of pollutants contains the infamous sulfur dioxide, produced in prestigious amounts, and the lesser produced sulfur trioxide. T h e L o w D o w n The vast majority of primary SOx pollution is anthropogenic (big word meaning man made) It is estimated that humans produce approximately 150 Million Tons of SO2 per year. That’s about fifty pounds per person! A large amount of SO2, however, does come as a natural secondary pollutant. About 200 Million Tons are produced as a natural secondary pollutant annually. Anthropogenic Sources T h e L o w D o w n Combustion of Impure The Major Sulfur Oxide Pollutor Coal: Low grade coal contains as much as seven percent sulfur impurity. Coal is polluted with both elemental sulfur, iron pyrite (FeS2), and organic sulfurs. Combustion causes these sulfurs to mix with oxygen, thus yielding Sulfur Oxides. Essentially: S(s) + O2(g SO2 Since sulfur is removed in the refinement of petroleum and natural gas, they are not major sources of sulfur oxide pollution. Anthropogenic Sources BOO! T h e L o w D o w n Smelting Plants Treat sulfur ores, since many ores exist as sulfides. I thought I SMELT somethin’ funny. The Reaction : 2 Cu2S(s) + 3 O2(g) 2 Cu2O(s) +2 SO2 Sulfuric Acid Plants Sulfur trioxide is produced by the oxidation of sulfur dioxide, and this is then reacted with water to form sulfuric acid. Some of the sulfur dioxide is released into the atmosphere rather than being properly and fully oxidized. As a Natural Secondary Pollutant T h e In nature, decay of organic material creates hydrogen sulfide gas. This reacts with oxygen to form sulfur dioxide. L o w It can also react with ozone: D o w n 2 H2S + 3 O2 2 SO2 + 2 H2O H2S + O3 SO2 + H2O “No Chemist ever goes undead. Being the bile of living decay is not only a degrading state of being for them, but they know that every day they are releasing hydrogen sulfide, which is going up into the air and producing pollution.” --Disowned HealthEffects Effects Health T h e L o w D o w n Sulfur oxides have no particular health effect of their own as primary pollutants, but when they mix with water they become acid rain and acid fog, and we well know by now that that ain’t good cool digs. The Chemistry of Controlling SOx pollution T h e L o w D o w n Purifying stuff before it’s burnt High sulfur coal is converted to synthetic natural gas (SNG) to remove the sulfur through gasification. Sulfur is converted to hydrogen sulfide, which is easily removed. S + H2 H2S Precumbustion removal of sulfur from petroleum and natural gas Hydrogen gas is bubbled through petroleum or natural gas so that becomes hydrogen sulfide, and this is acidic and easily removed. The Chemistry of Controlling SOx pollution T h e L o w D o w n Coal Washing A physical process by which coal is ground and washed with water. Iron Pyrite is dense, and sinks, thus removed easily. Organic sulfur, however, is chemically bonded to carbon and cannot be removed by this physical process. The Chemistry of Controlling SOx pollution T h e L o w D o w n After Burning Stuff Limestone injection or fluidized beds Powdered limestone, CaCO3, mixes with the coal in the combustion. Heat from the combustion decomposes the limestone into calcium oxide and carbon dioxide. Calcium oxide reacts with the SO2. CaO + SO2 CaSO3 or 2 CaO + 2 SO2 + O2 2 CaSO4 The Chemistry of Controlling SOx pollution T h e L o w D o w n Wet Scrubbers A wet-scrubber is a cleaning device that uses a liquid, usually water, to remove contaminants. Wet scrubbers are used to remove calcium sulfates, calcium oxides, extra sulfur dioxide, and particulates. The Chemistry of Controlling SOx pollution T h e L o w D o w n Wet Scrubbers Air + Coal + CaCO3 CO2 + SO2 +CaO + O2 CaO + H2O Alkaline Scrubber CO2 spray SO2 + CO2 CaO + CO2 + SO2 + CaSO4 Slurry Slurry Intermission •Pressing Questions? •Natural Needs? Particulates What are they? Particulates, also called aerosol, are solid particles or liquid droplets suspended or carried in the air. They are generally big enough to be seen. Smoke and soot produced by combustion. Dust from physical breakup of solid matter. Asbestos from industrial plants Metallic particles beryllium, mercury, arsenic, lead Particulates And their effects on health Unsurprisingly, most particulates invade the body of the animal through their process of respiration. The two genres of health effects, therefore, are directly related to the nature of the chemicals. BlahBlahosis is the scaring of lung tissue by small quantities of the chemical [blahblah], thus causing respiratory difficulties and increasing risk of cancer. Whatever Poisoning is caused by inhalation of particulates of the chemical [whatever], little tiny bits of a highly toxic chemicals which either cause damage in their tiny quantities or accumulate in the body to toxic levels. Particulates Asbestos and Beryllium Cause rather unpleasant respiratory conditions. Asbestos comes in a bunch of tiny crystals, which, when inhaled, and fart around in the lungs indefinitely. Asbestosis occurs: lung tissue is scarred and the lining of the lungs thicken. As a result, one experiences shortness of breath and is more prone to lung cancer. Beryllium is a toxic little devil of a chemical. When it gets in the lungs, it causes a problem called berylliosis. it inflames the tissue, causing pneumonia-like symptoms. Like asbestosis, it causes scarring of the lung tissue, and this in turn yields constant shortness of breath. Particulates Lead and Mercury These puppies are poison stuff. Lead, and more specifically, lead poisoning inhibits enzyme actions. Acute lead poisoning causes an array of problems in the human body from abdominal pain, nausea, numbness, muscle cramps, and brain damage. Mercury, also a metabolic poison, causes mercury poisoning, also known as Minamata disease. It reacts with and inhibits the human enzyme system, thus causing nerve damage and death. Particulates Being that particulates are just a bunch of little chunks of stuff floating around in the air, they are relatively easy to remove. Gravity settling chambers have a large tank which allows the solid dusts to settle, but this only works with large particles. Cyclone separators are essentially large scale centrifuges. The gas is sent through a spinning cylindrical camber. Centrifugal force causes the particles to move to the outsides and fall to the bottom while the gases escape from the top. Wet scrubbers are also effective. Baghouse filters are mentioned in the packet, but we know nothing about them. Particulates Electrostatic precipitators are particularly important in the removal the tiny particles. More than 98 percent of the particulate matter is removed. Gas Solids Hydrocarbons tend to exist as extremely volatile liquids. C1 to C4 are gases and C5 to C12 are liquids. Most are produced by nature, similar to NOx and CO pollution. Hydrocarbons are actually very similar in their nature as pollutants to those already mentioned. (makes it easy, but boring to learn and teach.) Natural Sources Geothermal Activity Bacterial Decomposition of Organic Matter Trees and Plants Anthropogenic Sources Creation of Petroleum Incomplete Combustion of Fuel Incomplete Combustion of Other Stuff Healthy Ef fects We are immune to aliphatic hydrocarbons unless they are in large amounts. eg. Methane may smell bad, but it ain’t killin’ ya Watch out for the aromatic compounds, though. They cause irritation of the mucous membrane and can be fatal in large amounts. (Dramatization) Ha Ha Oh cough ha! crap. ! Aromatic HC HC The Automobile Problem: A Summary -A Problem of Numbers The Cleaner Car -Non-Leaded Gas -Valves -Different Fuel to Air Ratios -Exhaust Recycling -Thermal Exhaust Reactors -Catalytic Converters Now that you have written all of this down, a summary of this information is available on the web. http://www.geocities.com/ib_homework Questions? Comments? Angry, Ranting, Foaming Criticisms? Fear not. If you did not speak now, a forum will be open on the website.