Practice Lewis Structures
advertisement

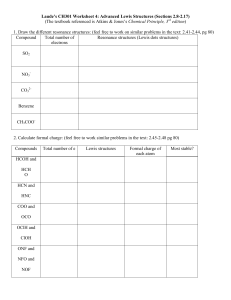
Expanding on Lewis Structures Lewis Structures • You can use lewis structures to predict some patterns in bonding. • Example : Nitrogen • Lewis Structure: • How many unpaired electrons? • 3 • How many bonds will it form? • 3 Practice Lewis Structures • Draw the Lewis Structures for: • SeF2 • SnCl4 • BF3 • Octet rule exception • B is fine with 3 bonds – it has 3 valence, so 3 bonds are expected • F NEVER FORMS A DOUBLE BOND Practice Lewis Structures • Draw NH3 and BF3 • When they bond, the nitrogen will donate it’s lone electron pair to the boron • Coordinate Covalent Bond • When a bonding pair of electrons originates on one of the bonded atoms • Both electrons in a bond come from a single atom Practice Lewis Structures • Draw the Lewis Structures for: • NO • Odd electron molecules can’t obey the octet rule… why? • ClO2 • Free Radicals – chemical species with an unpaired electron Practice Lewis Structures • Draw the Lewis Structures for: • SF4 • Expanded octet… empty 3d orbitals in S • I3 • F3• Can’t happen… no “d” orbitals to hold electrons • Only elements of the 3rd or higher periods in the periodic table may form compounds or ions in which an octet is exceeded Practice Lewis Structures • • • • SiF5PF5 ClF3 XeF2 Resonance • Draw CO3-2 • Each structure is equally likely, “TRUE” CO3-2 is an average of the 3 structures Resonance • Draw Ozone (O3) • One lewis structure cannot accurately describe ozone Resonance • Resonance • when a single Lewis structure fails to describe accurately the actual electronic structure • Resonance Structures alternative structures • Actual structure is a composite : resonance hybrid Resonance Practice • C6H6 • H2O4-2 • OCN- Writing Formal Charges • Every atom has an assigned formal charge: Formal Charge = [# of valence e-] – [(# of nonbonding electrons)+1/2(# of bonding electrons)] Writing Formal Charges • Formal charges must add up to the overall charge • Large formal charges are unlikely, and negative formal charges “want” to reside on very electronegative elements Writing Formal Charges • Calculate the formal charge for: • CN• SO3-2