Protein domain BioBricks
advertisement

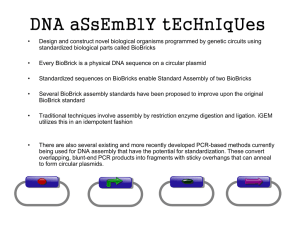
Protein domain BioBricks Perry Tsai June 19, 2006 Goals (1) Demonstrate the use of protein domains in BioBricks assembly (hDlg experiment) (2) Develop library of BioBrick’d protein domains (populate the registry) (3) Design and construct a novel protein (for its own sake or for other project) http://www.criduchat.asn.au/criduchat/Images/notebook.JPG The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf The pCMV mammalian expression vector http://www.stratagene.com/vectors/maps/pdf/pCMV-Tag3A.pdf Converting multiple cloning site to BioBricks site pCMV-Tag3A vector BamHI/XhoI digest BamHI sticky ends XhoI Converting multiple cloning site to BioBricks site BamHI BamHI/XhoI digest pCMV-Tag3A vector sticky ends XhoI + BioBricks linker 5’-GATCCTGAATTCGCGGCCGCTTCTAGAGTACTAGTAGCGGCCGCTGCAGC -3’ 3’GACTTAAGCGCCGGCGAAGATCTCATGATCATCGCCGGCGACGTCGAGCT -5’ BamHI EcoRI NotI XbaI SpeI NotI PstI XhoI Converting multiple cloning site to BioBricks site BamHI BamHI/XhoI digest pCMV-Tag3A vector sticky ends XhoI + BioBricks linker 5’-GATCCTGAATTCGCGGCCGCTTCTAGAGTACTAGTAGCGGCCGCTGCAGC -3’ 3’GACTTAAGCGCCGGCGAAGATCTCATGATCATCGCCGGCGACGTCGAGCT -5’ BamHI EcoRI NotI XbaI SpeI NotI ligase pCMV-Tag3A-BB vector BioBricks site pCMV-Tag3A-BB PstI XhoI Converting protein domains to BioBricks EcoRI NotI XbaI 5’ GTTTCTCCGAATTCGCGGCCGCTTCTAGAG[Part, 5’->3’ sense] Protein domain coding sequence 5’ 3’ 3’ 5’ [Part, 3’->5’ antisense]-ATGATCATCGCCGGCGACGTCCCTCTTTG 3’ SpeI NotI PstI Converting protein domains to BioBricks EcoRI NotI XbaI 5’ GTTTCTCCGAATTCGCGGCCGCTTCTAGAG[Part, 5’->3’ sense] Protein domain coding sequence 5’ 3’ 3’ 5’ [Part, 3’->5’ antisense]-ATGATCATCGCCGGCGACGTCCCTCTTTG 3’ SpeI NotI PstI PCR Protein domain coding sequence 5’ GTTTCTCCGAATTCGCGGCCGCTTCTAGAG 3’ CAAAGAGGCTTAAGCGCCGGCGAAGATCTC EcoRI NotI XbaI TACTAGTAGCGGCCGCTGCAGGGAGAAAC 3’ ATGATCATCGCCGGCGACGTCCCTCTTTG 5’ SpeI NotI PstI Converting protein domains to BioBricks EcoRI NotI XbaI 5’ GTTTCTCCGAATTCGCGGCCGCTTCTAGAG[Part, 5’->3’ sense] Protein domain coding sequence 5’ 3’ 3’ 5’ [Part, 3’->5’ antisense]-ATGATCATCGCCGGCGACGTCCCTCTTTG 3’ SpeI NotI PstI PCR Protein domain coding sequence 5’ GTTTCTCCGAATTCGCGGCCGCTTCTAGAG 3’ CAAAGAGGCTTAAGCGCCGGCGAAGATCTC EcoRI NotI XbaI TACTAGTAGCGGCCGCTGCAGGGAGAAAC 3’ ATGATCATCGCCGGCGACGTCCCTCTTTG 5’ SpeI NotI PstI PstI XhoI TOPO cloning Protein domain BioBrick TOPO vector Caveat: BioBricks restriction sites need to be removed from the coding sequence via site-directed mutagenesis. The hDlg protein • Human homolog of Drosophila Discs-large tumor suppressor • Alternatively spliced isoforms McLaughlin M, Hale R, Ellston D, Gaudet S, Lue RA, and Viel A. The distribution and function of alternatively spliced insertions in hDlg. J Biol Chem 2002 Feb 22; 277(8) 6406-12. The hDlg protein • Human homolog of Drosophila Discs-large tumor suppressor • Alternatively spliced isoforms • Insertions I2, I3, I4, I5 McLaughlin M, Hale R, Ellston D, Gaudet S, Lue RA, and Viel A. The distribution and function of alternatively spliced insertions in hDlg. J Biol Chem 2002 Feb 22; 277(8) 6406-12. The hDlg protein • Human homolog of Drosophila Discs-large tumor suppressor • Alternatively spliced isoforms • Insertions I2, I3, I4, I5 • I3 targets to plasma membrane; I2 targets to nucleus McLaughlin M, Hale R, Ellston D, Gaudet S, Lue RA, and Viel A. The distribution and function of alternatively spliced insertions in hDlg. J Biol Chem 2002 Feb 22; 277(8) 6406-12. Mix and match F11 PCR to create BioBrick parts 1-9 SG25 SG35 I2-I5-I4 Mix and match F11 PCR to create BioBrick parts 1-9 SG25 SG35 I2-I5-I4 Assemble into constructs Progress and agenda pCMV-Tag3BB vector and BioBrick parts 1-9 have been created, sequenced, and midiprepped. Assemblies have been attempted but not verified. Next, assemblies need to be completed and verified by sequencing. (3 wks) Then, constructs can be transfected into human epithelial cells, selected by kanamycin resistance. (2 wks) Finally, localization of hDlg isoforms will be tested by immunostaining for myc tag. (2 wks) Pros/Cons Achievable in 1-2 months by 2-3 people An observable proof of principle The principle has already been proven. (Silver lab) Not creative Not iGEM-y http://www.physlink.com/estore/cart/item_images/780_xl.jpg Protein domain BioBricks library Develop a BioBricks library of protein domains within the Registry Mix and match protein domains Model alternatively spliced proteins, like in hDlg experiment Add, remove, or replace domains for altered functionality Fuse domains together into chimeric proteins Design new proteins from characterized domains http://parts.mit.edu/registry/index.php/Image:Registry-head-c2g.png Types of domains Receptor (iron, aspartate) Kinase (tyrosine, serine) Transmembrane (OmpX) Localization (nuclear, membrane) Targeting (intercell) DNA binding (repressor) http://olenka.med.virginia.edu/mcsg/images/structures/2AU5x500r.jpg Sources of domains Genomes (E. coli, human, yeast) cDNA, clone libraries Synthesis Mutation Evolution http://olenka.med.virginia.edu/mcsg/images/structures/2AU5x500r.jpg Agenda Find out what domains Silver lab has BioBrick’d and what may be added to the Registry. Research other interesting domains, design primers, order primers, DNA sources, and plasmids. (2 wks) BioBrick parts from chosen protein domains, add BioBricks sites to plasmids if necessary, clone into plasmids. . (3 wks) Modify parts as needed (remove restriction sites, mutations). (1 wk) Verify parts by sequencing. (1 wk) Pros/Cons Flexible timeline and goals Estimated goal of 10-15 parts in 2 months by 3-4 people Independent chances at success and failure Potential to plan for more parts if ahead of schedule Important for future iGEM teams, future protein domain research Silver lab experience Not terribly exciting Not creative, except choice/modification of parts http://www.physlink.com/estore/cart/item_images/780_xl.jpg Designing/constructing a novel protein Use BioBrick’d protein domains to build a new protein Can be any desired protein, or a protein useful for another project http://www.cygnus-x1.net/links/lcars/blueprints/star-trek-the-motion-picture-sheet-4.jpg Chimeric protein examples LamB secretion signal + mature bovine somatotropin Aspartate chemoreceptor + EnvZ activator of OmpC transcription Moe GR, Bollag GE, and Koshland DE Jr. Transmembrane signaling by a chimera of the Escherichia coli aspartate receptor and the human insulin receptor. Proc Natl Acad Sci U S A 1989 Aug; 86(15) 5683-7. Iron receptor + beta-galactosidase Utsumi R, Brissette RE, Rampersaud A, Forst SA, Oosawa K, and Inouye M. Activation of bacterial porin gene expression by a chimeric signal transducer in response to aspartate. Science 1989 Sep 15; 245(4923) 1246-9. Aspartate chemoreceptor + human insulin receptor kinase Klein BK, Hill SR, Devine CS, Rowold E, Smith CE, Galosy S, and Olins PO. Secretion of active bovine somatotropin in Escherichia coli. Biotechnology (N Y) 1991 Sep; 9(9) 869-72. Coulton JW, Mason P, Cameron DR, Carmel G, Jean R, and Rode HN. Protein fusions of beta-galactosidase to the ferrichrome-iron receptor of Escherichia coli K-12. J Bacteriol 1986 Jan; 165(1) 181-92. Maltose-binding protein + SH2 tyrosine kinase substrate Sierke SL and Koland JG. SH2 domain proteins as high-affinity receptor tyrosine kinase substrates. http://www.planetadnd.com/interactive_books/chimera.gif Agenda Design a novel protein, and research necessary domains. Design primers. Order DNA source, primer, plasmid. (1 wk) BioBrick domains, add BioBrick site to plasmid if necessary, clone into plasmid. (2 wks) Modify parts as needed (remove restriction sites) (1 wk) Verify parts by sequencing, while beginning assembly. (1 wk) Complete assembly, and verify by sequencing. (2 wks) Test functionality of construct. (1 wk) Pros/Cons Creative exercise Applicable and useful to many potential projects Difficult to ensure success with a novel protein Protein domains may not be so modular Tighter timeline because of BioBricking + assembly. http://www.physlink.com/estore/cart/item_images/780_xl.jpg Final thoughts 3 projects: (1) hDlg, (2) library, (3) novel protein Interestingness? 1 < 2 < 3 1 is “easily” achievable 2 & 3 have overlapping agendas; feasible with 4-5 people working over 2 months. 2 & 3 require further research 1 & 2 are independent projects, while 3 may depend on 2, and another project may depend on 3