Elements and Compounds: Middle School Science
advertisement

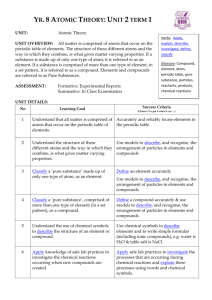
TEKS 6.5A: Know that an element is a pure substance represented by chemical symbols. TEKS 6.5C: Differentiate between elements and compounds on the most basic level. What are elements? • An element is a pure substance that cannot be broken down into any other substances. • Each element can be identified by its specific physical and chemical properties. • Each element is represented by a one- or two-letter symbol. • Carbon: C Oxygen: O • Hydrogen: H Aluminum: Al • Elements are made of particles called atoms. Elements have different properties because their atoms are different. 1. Apply Concepts Give an example of a common object made from a single element. TEKS 6.5A: Know that an element is a pure substance represented by chemical symbols. TEKS 6.5C: Differentiate between elements and compounds on the most basic level. What are compounds? • Atoms can be held together by a chemical bond. Two or more atoms combined by chemical bonds may form a particle called a molecule. • A compound is a substance made of two or more elements that are chemically combined in a set ratio. Molecules of a compound are always made up of the same ratio of atoms of each element. (contd.) TEKS 6.5A: Know that an element is a pure substance represented by chemical symbols. TEKS 6.5C: Differentiate between elements and compounds on the most basic level. • A chemical formula represents a compound. The numbers in a chemical formula show the ratio of atoms of each element. If there is no number, the number is assumed to be 1. • In the formula for carbon dioxide, CO2, the 2 means there are two atoms of oxygen (O) for each atom of carbon (C). • In the formula for carbon monoxide, CO, there is one atom of oxygen for each atom of carbon. • Compounds have different properties than the elements in them have. • Sulfur: yellow, powdery element • Silver: shiny, metal element • Silver sulfide (tarnish): black compound 2. Identify The formula for water is H2O. How many atoms of hydrogen are there for each atom of oxygen?