ANTI-CANCER THERAPY
advertisement

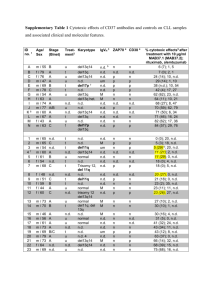
ANTI-CANCER THERAPY MONOCLONAL ANTIBODIES BY: FIROUZEH KAMALI Conventional Anti-Cancer Therapy Chemotherapy: Imperfect Systematic nature of cytoxicity Agents lack intrinsic anti-tumor selectivity Anti-proliferative mechanism on cells in cycle, rather than specific toxicity directed towards particular cancer cell Host toxicity: treatment discontinued at dose levels well below dose required to kill all viable tumor cells HISTORY Emil von Behring in 1890 Discovered antibodies Paul Ehrlich (16 years later) Coined phrase, “magic bullets and poisoned arrows”: use of antibodies to specifically target toxic substances in pathogenic substances Kohler and Milstein in 1975 Discovery of monoclonal antibodies (mAb) directed against well-characterized antigens Use of DNA bio-engineered technologies within last 25 years Rationale mAb as efficient carriers for delivery of antitumor agents Enhanced vascular permeability of circulating macromolecules for tumor tissue and subsequent accumulation in solid tumors Normal tissue: blood vessels have intact endothelial layer that permits passage of small molecules but not entry of macromolecules (like mAb) Tumor tissue: blood vessels leaky, so small and large molecules have access to malignant tissue -tumor tissue generally do not have a lymphatic drainage system; therefore, macromolecules are retained and can accumulate in solid tumors Patho-physiology of Tumor Tissue Angiogenesis Hypervasculature Impaired lymphatic drainage ***Due to these characteristics, tumors can be exploited for tumor-selective drug delivery**** Genetic Engineering Remove or modify effector functions of mAb: used to avoid unwanted side effects Use mAb in their natural, fragmented, chemically modified, or recombinant forms Use of phage display antibody libraries or transgenic animals Identify animals that make desired antibodies Animals must be immunized using the cellular antigens and immunization procedures used to generate conventional antibodies Perform cell fusions to generate clones and isolate stable clones, making mAb Most mAb used in the clinical setting were generated in mice Structure of Antibody Presently, all intact therapeutic antibodies are murine immunoglobulins of the IgG class Murine immunoglobulin = glycoprotein that has a Y-shaped structure: 2 identical polypeptide heavy chains and 2 identical light chains linked by an S-S bond Chimeric antibody = genetically engineered construct containing a mouse Fab portion and a human Fc portion 3 main components Two identical Fabs (fragment-antigen binding site): the arms of the Y An Fc (for fragment crystallizable), the stem of the Y Constant region responsible for triggering effector functions that eliminate the antigen-associated cells Constant region must be tailored to match requirements of the antibody (depending on which antigen you want it to bind to) IgG structure 3 MECHANISMS RESULTING IN APOPTOSIS Antigen cross-linking Activation of death receptors Blockade of ligand-receptor growth or survival pathways 1. Antigen Cross-Linking Target growth factor receptor Antagonize ligand-receptor signaling Growth-factor signaling mediated by the receptor tyrosine kinase is inhibited EGFR (epidermal growth factor receptor) IGF-1R (insulin-like growth factor-1 receptor) FGFR (fibroblast growth factor receptor) PDGFR (platelet-derived growth factor receptor) VEGFR (vascular endothelial growth factor) Results in arrest of tumor cell growth 2. Activation of death receptors Cross-link targeted surface antigens on tumor cells and antibody agonists that mimic ligand-mediated activation of specific receptors Response: intracellular Ca II ions increase Activate caspase-3 and caspase-9 (involved in cell apoptosis) APOPTOSIS PATHWAY 3. Delivery of Cytotoxic Agents Physically link antibodies to toxic substances for delivery Radio-immunoconjugates (aim of delivering radiation directly to the tumor) Toxin-immunoconjugates (deliver toxins intracellularly) Antibody-directed enzyme pro-drug therapy (ADEPT): localize enzymes to tumor cell surfaces General Drug Delivery System Drug molecules bound to macromolecule through spacer molecule Drug released from macromolecule after cellular uptake of the conjugate Targeting moiety = monoclonal antibody TOXIN IMMUNOCONJUGATES Cell surface antigen must internalize upon mAb binding When drug is released, it interferes with protein synthesis to induce apoptosis 3 methods to attach cytotoxic drug to variable regions of mAb a. Couple drug to lysine moieties in the mAb b. Generation of aldehyde groups by oxidizing the carbohydrate region and subsequent reaction with amino-containing drugs or drug derivatives c. Couple drugs to sulfhydryl groups by selectively reducing the interchain disulfides near the Fc region of the mAb Direct attachment of mAb to drug by S-S bonding Immunoconjugate BR96-doxorubicin conjugate (BR96-DOX) Promising toxinimmunoconjugate mouse/human chimeric mAb Targets antigen overexpressed on surface of human carcinoma cells of breast, colon, lung, and ovary Disulfide reduction attaches mAb to drug, BR96 Dose that can be safely administered every 3 weeks is insufficient Other examples of toxinimmunoconjugates KS1/4-MTX Conjugate of methotrexate (MTX) Coupling of MTX to the lysine moieties of the mAb No significant clinical response KS1/4-DAVLB Conjugate of vinca alkaloid derivatives Vinca alkaloid derivatives attached to amino groups of lysine residues on KS1/4 mAb No significant clinical response Why are these toxinimmunoconjugates unsuccessful? Cause gastrointestinal toxicity Inner regions of solid tumors poorly vascularized and have low blood flow (reduce amount of immunoconjugate reaching these parts of the tumor) Antigen expression is heterogenous on tumor cells Restricts the amount of cells that can be effectively targeted by antibody conjugates ADEPT ENZYMES (Antibodydirected enzyme pro-drug therapy) Chemically link the mAb to the enzyme of interest; can also be a fusion protein produced recombinantly with the antibody variable region genes and the gene coding the enzyme Convert subsequently administered anti-cancer pro-drugs into active anti-tumor agents Upon binding to targeted enzymes, it is converted into active drug Anti-growth factor mAb Therapy Angiogenesis Formation of nascent blood vessels VEGF One of the most upregulated antigens in cancer Protect endothelial cells from apoptosis via activation of PKC pathways and upregulation of anti-apoptotic proteins such as Bcl-2 Activity mediated by tyrosine kinase receptors, VEGFR 1 and VEGFR 2 Functions indirectly as survival factor for tumor cells Inhibit VEGF signaling Block the receptor Inhibits tumor growth and metastasis Deprives tumors of nutrient-providing blood vessels RITUXIMAB (Rituxan) 1st therapeutic mAb approved by FDA in 1997 High-level expression of the gene encoding Rituximab was found a mouse-chimeric mAb Contains the human IgG1 and murine variable regions that target CD20 B-cell antigen CD20 antigen function: cell cycle progression Binding Rituximab to CD-20 causes: autophosphorylation, activation of serine/tyrosine protein kinases, and induction of oncogene expression --induces apoptosis Response rates of 50% to 70% in follicular lymphomas Response rates of 90% to 100% when used in combination with various chemotherpay procedures Concluded that the dose of 4, once-weekly 375 mg/m squared IV infusions of Rituximab was safe and effective in patients with relapse or refractory B non-Hodgkin’s lymphoma Toxic effects of Rituximab Short-lived mild reactions to infusion after first treatment: fever, chills, rigors, rash, and nausea Factors affecting pharmacokinetic parameters Circulating target antigens (which can lead to rapid clearance) Antigen-antibody internalization in cells (which affect serum clearance and half-life) Antibody size and domains with the Fc region Fragments have shorter half-lives and more rapid clearance rates than their full-sized immunoglobulins FUTURE Researchers hope to define the optimal combinations of the use of mAb with conventional chemotherapeutic agents and with radiation therapy Determine best therapy candidates and expand clinical trials to other tumor types