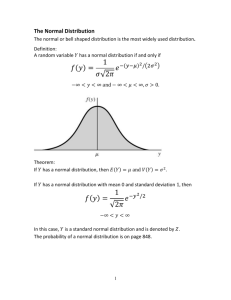
Trending GMP Issues In API Manufacturing Operations
advertisement

Trending GMP Issues In API Manufacturing Operations CHARLES I. AHN, USFDA ASSISTANT COUNTRY DIRECTOR, CHINA SHANGHAI OFFICE Trending GMP Issues API Mfg. Operations Quality System Infra-structure Still Being Implemented Double Standards Quality System Concept Not Deep-rooted Process Validation User/Process Requirement Assessment New Systems, New Equipment-Not Enough Support Monitoring, Recording Activities Handling & Investigation of Deviations and OOS Process Validation Protocol Define Process Identify &Qualify Controlled Parameters Identify & Qualify Monitored Parameters Describe Evaluation Criteria Establish Metrics No More Three Passing Batches On-going Effort, Continuous Assessment of Variables Case Study API Final Drying Process Drying Process Vacuum > 0.08MPa Drying Temp: 70~90oC Dryng Time: 7~8 Hours Historical Range Narrower 75~78oC and 7 Hours Fixed Time Based on R&D Validation Data Firm Considers the Process Validated Validation Issues Blender/Dryer Jacketed with Coil Circulating Heated Water Source of Heat: Water Coil Heat-exchange in “Steam Box” with Manual Valve to Steam Generator No Correlation Established between Jacket Temp. and Steam Box Temp. The Firm Uses Both Jacket and Steam Box Temperature Readings as Process Control Parameters Excessively Wide Temperature and Drying Time Ranges Specified Validation Consideration Need to Correlate Jacket Temperature and Heat Source Temperature One Can Only “Control” Source Temperature; and Jacket Temperature is Dependent on the Source Drying Temp. Should Be A Narrow Target Range Exhaust Air Temperature and End-point Determination Drying Curve Material’s Initial Wetness, Potential Variable(?) Assumption of Efficient Vacuum Deviation Investigation: November, 2008 A Drying Failure Deviation Was Noted During Final Drying Process Temperature Fell Below Limit For A Short-while Did Not Indicate How Long, What Caused Deviation LOD Testing Confirmed Drying Failure Re-drying and Re-milling per Modified Instruction No Investigation Into Cause of Temperature Fluctuation – Only Operator Error (?) No Stability Profile Deviation Investigation: November, 2008 F/U Action to Deviation: November, 2008 Deviation Report, March, 2009 Another Drying Failure Loss on Drying Test Result 1.12% (Limit 0.5%) Operator Error- Drying Time 6.5 Hrs. (>7 Hrs) Operator Re-trained Drying Process (and Milling) Repeated 3.5 Hrs. API Released upon Retesting No Concern for Other Quality attributes No Concern for Adequacy of Controls, or Process Reprocessing Procedure(?), Stability No Look-back of November Deviation Event OOS Report, March, 2009 Deviation Investigation Upon OOS Confirm F/U Action: March, 2009 QS Feedback And Process Consideration The Drying Process Might Not Be Robust Brief Fluctuation of Temperature and Shortened Drying Time Resulted in Drying Failure Drying Time and Drying Temperature Critical Parameters Capturing Relevant Production Data, Out-of-Trend Events Useful and Necessary to Evaluate Process Data Should Not Be Filed Away, But Collected in Systematic Way to Allow Trending and Quality Assessment Quality Management System Discrepancy Case Study- Water System Validation Water System Appearance May Deceive Your Eyes Sand Bed, Carbon Bed, 10m Filter, 1st RO, Intermediate Storage, 2nd RO, Purified Water, UV Conductivity Reading Monitored At Pre-treatment, 1st RO, and 2nd RO. Reading at the 1st RO Invariably Spikes Up When the System Starts Up on Demand; Water Discharged Until Reading Back to Normal Has the Water Purification System Been Validated? Handling of Deviations Typical Observations The Firm’s Quality Management System - Handling and Investigating Deviations, Out-of-Specification Data, and Out-of-Trend Events Is Inadequate. The firm’s Identification and Documentation of Events, Investigation Activities, Conclusions, and F/U Actions Are Incomplete and Not Supported by Factual Findings. Actions Retrospective, Not Prospective. Example of Deviation Handling A Seed Culture Lot XXXXX for Neomycin Sulfate Was Contaminated and Discarded. The Firm’s Investigation and Disposition of the Deviation Event Concluded the Cause of Contamination Was Likely Due to Cracked Viewing Glass and Microbial Ingression During the Seed Culture Growth Step However, There Is No Documentation of Sequence of Event Supporting Such Conclusion. Time of Breakage, Identity of Contamination, Lookback Example of Deviation Handling Final API Fermentation Lot YYY, Streptomycin, Was Contaminated and Discarded. The Firm’s Investigation Concluded That the Cooling Water Coil Running Through the Fermentation Vessel Was Discovered Cracked Along the Welded Connection While Cleaning the Vessel After the Operation. No Further Investigation for Other Potential Root Causes or Look-back of Previous Batches Were Noted to Have Been Conducted. Example of Deviation Handling Deviation Report ZZZ Addressed a Yield of Erythromycin Lot xxx Calculated to be 85%, Outside the Established Range of 88-97%. It Was Observed During Drying of Crystallized Material, Part of It Was Adhering to the Surface of Dryer. Recovered the Material Amounting to 35kgs, Tested for Specifications, Determined to Be Acceptable, Redried to 29kgs, and Used in the Subsequent Step. Example of Deviation Handling Supply Chain Issue Sterile Filtration Failure. Attributed to Failure of Membrane Pre-filter. Sample Membrane Filter Units Sent to the Vendor for Evaluation. Bubble Point Testing Performed by Vendor Confirmed That Filter Units Were Indeed Not Rated for 0.2μ Pore Size But Was Actually Performing at 0.3μ Range. Vendor Explained It Was Unable to Test The Filter Lot at The Time of Shipping Due to Temporary Unavailability of Testing Apparatus. Processing Steps Temporary Storage Typical API Manufacturing Process Is Broken Down Into a Number of Discrete Steps, Requiring Temporary Storage of Processed Materials Prior to Start of the Subsequent Step. Not (Necessarily) a Stability Issue But Must Establish a Procedure to Minimize Contamination or to Maintain Suitability to Continue Other Common Issues Stability Indicating Analytical Methods Use of In-House (Working) Reference Standard Qualification of Working Standard Loose Analyst Work-sheets and Accountability