Topic 10. Organic chemistry
advertisement

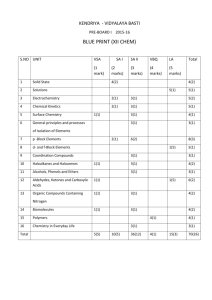
Topic 10 Organic chemistry • Inorganic chemistry? Chemistry of life (now biochemistry) Carbon chemistry • Introduction • Alkanes • Alkenes • Alkohols • Halogenalkanes • Reaction pathways 10.1 Carbon molecules • • • • Carbon has 4 valence electron’s (1s22s22p2) Tetrahedral structures if single bonds Planar triangular if double bond Linear if triple bonded Organic substances • Carbon is often (almost always) covalently bonded • Other common atoms is hydrogen, oxygen, nitrogen ….. • Molecules can be long chains or ring form • Hydrocarbons: Compounds only containing carbon and hydrogen HYDROCARBONS • Molecules containing only C and H • Alkanes, alkenes, alkynes, arenes, cycloalkanes Homologous series • • • • Same general formula Neighbouring members differ by –CH2Similar chemical properties Gradation in physical properties, such as boiling point The first straight-chain alkanes, general formula CnH2n+2 Name Molecular formula Structural formula Boiling point (oC) Methane CH4 CH4 - 162 Ethane C2H6 CH3-CH3 - 89 Propane C3H8 CH3-CH2-CH3 - 42 Butane C4H10 CH3-CH2-CH2-CH3 - 0,5 Pentane C5H12 CH3-CH2-CH2-CH2-CH3 36 Hexane C6H14 CH3-CH2-CH2-CH2-CH2-CH3 69 Explain the trend! Predict the boiling point of heptane! Different ways of representing structural formulas Skeletal formula Empirical formula? Isomers Same molecular formula but different structural formulas Butane have 2 isomers: • CH3-CH2-CH2-CH3 Butane • CH3-CH-(CH3)-CH3 Methyl-propane Methyl-group C H 3 H C C H C H C H C H C H C H 3 2 2 2 2 3 1 2 3 4 5 6 7 1. Find the longest carbon chain and you have the ”last name” of the compound: 7→ heptane 2. Find and name substituents: 1 metyl-group 3. Number the carbons in the chain so that substituents gets as low no. as possible: the methyl-group is attached at carbon no. 2 4. Put it together: 2-methyl-heptane Ethyl-group C H 3 Methyl-group C H 2 H C C H C H C H C H 3 2 3 1 2 3 4 5 C H 2 1. Find the longest carbon chain and you have the ”last name” of the compound: 5→ pentane 2. Find and name substituents: 1 metyl-group and 1 ethyl-group 3. Number the carbons in the chain so that substituents gets as low no. as possible: the methyl-group is attached at C no. 3, and the ethyl-group at C no. 2 4. Put it together: 3-etyl-2-methyl-pentane Which names? The names 2-methyl-propane 2, 3-dimethyl-butane 2-methyl-butane 2,2-dimethyl-butane 2,2-dimethyl-propane 2-methyl-pentane 10.2 Alkanes • Homologous series of a saturated hydrocarbon with only single bonds between carbon atoms • General formula CnH2n+2 • Low m.p. and b.p. due to only van der Waal´s forces • Often gases and liquids at room temperature • Low reactivity Alkanes have high bond entalpies and low reactivity Bond C-C C-H Bond enthalpy kJ/mol 348 412 Bond Si-Si Si-H Bond enthalpy kJ/mol 226 318 Ge-Ge Sn-Sn 188 151 Alkanes have low bond polarity and low reactivity Bond C-C C-H C-F Difference in electronegativity 0 0,4 1,5 C-Cl C-O C-N 1,0 1,0 0,5 The higher the difference in electronegativity, the higher the reactivity in reactions with e.g. nucleophiles (as we shall see later…) About hydrocarbons • Alkanes and other hydrocarbons are good fuels • Complete combustion: Hydrocarbons + oxygen Carbon dioxide + water +heat • If oxygen is limited then incomplete combustion: carbon monoxide, CO and elementary carbon, C, may be formed Combustion of octane • Complete (with plenty of O2): C8H18 + 25 O2 16 CO2 + 18 H2O • Incomplete (not enough O2): C8H18 + 9 O2 C + CO +2 CO2 + 9 H2O Reactivity • Alkanes can react with radicals- eg. chlorine in UV-light UV Cl-Cl 2 Cl• unpaired electron • Compounds with unpaired electrons are called free radicals and are very reactive UV • Cl-Cl 2 Cl• homolytic fission • A-B A+ + Bheterolytic fission The reaction of methane and chlorine by radical reactions 1. 2. 3. 4. 2 Cl• + CH4 CH3Cl + HCl 2 Cl• + CH3Cl CH2Cl2 +HCl 2 Cl• + CH2Cl2 CHCl3 + HCl 2 Cl• + CHCl3 CCl4 + HCl • A mixture of chlorinated methanes is achieved • Radical reactions involve an initiation step, one or more propagation steps, and a termination step Radical reactions 10.3 Alkenes • Homologous series of unsaturated hydrocarbons with one or more double bonds between carbon atoms • General formula CnH2n • Low m.p. and b.p. due to only van der Waal´s forces • Often gases and liquids at room temperature Ethene CH2=CH2 Propene CH3-CH=CH2 Butene CH3-CH2-CH=CH2 1-Butene or But-1-ene CH3-CH2=CH-CH3 2-Butene or But-2-ene Pentene CH3-CH2-CH2-CH=CH2 1-Pentene or Pent-1-ene CH3-CH2-CH2=CH-CH3 2-Pentene or Pent-2-ene Reactions of alkenes • Reactive double bonds • Low activation energy • Addition and polymerization reactions Addition reactions with bromine and hydrogen chloride • H2C=CH2 + Br2 colourless red/brown H2BrC-CH2Br Spontaneous at NTP colourless Used as proof of C-C-double bonds • H2C=CH2 + HCl H3C-CH2Cl Spontaneous at NTP Addition reactions with hydrogen and water • H2C=CH2 + H2 H3C-CH3 E.g. Ni-catalyst. Industrially important when transformation of vegetable oil to margarine • H2C=CH2 + H2O H3C-CH2OH Catalyst: H2SO4, H3PO4 or Al2O3 ~300oC, 7 MPa. At 1 atm the reversed reaction is favoured. Synthesis of alcohols Polymerisation reactions • Alkenes forming plastics, making plastics • Radical reactions involving Cl2 and UV-light • Initiation: UV Cl-Cl 2 Cl• • Propagation; adding monomers to a long chain, e.g. H2C=CH2 + Cl• •H2C-CH2Cl monomer •H2C-CH2Cl + H2C=CH2 •H2C-CH2-H2C-CH2Cl •H2C-CH2-H2C-CH2Cl + H2C=CH2 •H2C-CH2-H2C-CH2H2C-CH2Cl Termination Two radicals meet and a bond is formed. R-CH2• + R’-CH2• R-CH2-CH2-R’ • The polymer is ready! Addition polymerisation reactions (I) High pressure n Ethene monomer Cl n Chloroethene monomer n Propene monomer * n * Repeating unit of polyethene, PE n * * Cl Repeating unit of polychloroethene (polyvinylchloride, PVC) * n * Repeating unit of polypropene (PP) Addition polymerisation reactions (II) n n * P h e n y le t h e n e m o n o m e r R e p e a t in g u n it o f p o ly p h e n y le t h e n e ( p o ly s t y r e n e , P S ) F F n F F F T e t r a f lu o r e t h e n e m o n o m e r * * F n * F F R e p e a t in g u n it o f p o ly t e t r a f lu o r e t h e n e ( P T F E ) Teflon Benzene ring • Identify when present in structural formula, phenyl ring Functional groups • C-C double and triple bonds, phenyl ring • Other elements bonded in different ways with the carbon chain; alcohol, aldehyde, keton, carboxylic acid, amine, ester, halide • Give the molecule other chemical and physical properties 10.4 Alcohols • The functional group –OH • Name: stem + the suffix –anol (or as prefix: hydroxy) • H-bonds => higher b.p., smaller ones (C1-C3) are water-soluble • Methanol CH3OH Wood spirit, formed by pyrolysis of wood. Highly toxic! • Ethanol CH3-CH2-OH Alcohol, formed during fermentation of sugar. Technically very important; In drinks, as a solvent, desinfectant and fuel Propanol • CH3-CH2-CH2-OH 1-propanol or propan-1-ol a primary alcohol • CH3-CHOH-CH3 2-propanol or propan-2-ol a secondary alcohol Isopropanol, used as windscreen de-icer Butanol • CH3-CH2-CH2-CH2-OH 1-butanol or butan-1-ol Non-water soluble • CH3-CCH3OH-CH3 2-metyl-2-propanol or 2-metylpropan-2-ol, a tertiary alcohol Water soluble Combustion Alcohol + Oxygen Carbon dioxide + water CH3-CH2-OH + 3 O2 2 CO2 + 3 H2O Redox reactions in organic chemistry • The carbon with a functional group (eg. –OH) will be oxidised first • Oxidation: Add oxygen and/or remove hydrogen from the carbon • Reduction: Add hydrogen and/or remove oxygen from the carbon oxidation reduction Alcohol Aldehyde CH4 CH3OH HCHO H-COOH CO2 Methane Methanol Methanal Methanoic acid Carbon dioxide H H H C H H H C H Carboxylic acid O O H H O C H H O C O H C O The more bonds to oxygen, the higher oxidation state of a carbon Oxidation of alcohols • Primary alcohol Aldehyde Carboxylic acid • Secondary alcohol Ketone • Tertiary alcohol no oxidation (unless C-Cbonds are broken) K2Cr2O7, Potassium dichromate, a common oxidizing agent H H C H H H C O H H C H O H C H H C H CH3CH2OH + Cr2O72- + H+ CH3CHO C O H H CH3COOH + Cr3+ + H2O Reactive. Stable. Distill of when formed Reflux Other oxidizing agents KMnO4, CuO, CuSO4 O Aldehyde • The functional group –CHO or • Name: stem + suffix: -anal • Dipoles => slightly higher bp’s, smaller ones are water-soluble etc. • Quite reactive compounds • Methanal H-CHO • Ethanal CH3-CHO • Formed by light oxidation of primary alcohols Ketone • The functional group –CO- or • Name: stem + suffix: -anone • Dipoles => slightly higher bp’s, smaller ones are water-soluble etc. • Propanone (acetone) CH3-CO-CH3 • Pentane-2-one CH3-CO-CH2-CH2-CH3 • Formed by oxidation of secondary alcohols Carboxylic acids • The functional group –COOH or • Name: stem + suffix: -anoic acid • H-bonds => higher bp’s, smaller ones are watersoluble etc. • Acidic reactions • Methanoic acid H-COOH • Ethanoic acid CH3-COOH • Formed by strong oxidation of primary alcohols Salt of Carboxylic acids • • • • • Salt form: -COO- or Name: stem + suffix: -oate ion Methanoate H-COOEthanoate CH3-COOFormed by reaction of carboxylic acid and base: NaOH + CH3COOH Na+ + CH3COO- Halogenoalkane • Functional group: -X (-F, -Cl, -Br, -I) • Name: e.g. prefix: Chloro- + alkane • Chloromethane CH3-Cl • Bromoethane CH3-CH2-Br Ester • Identify when present in structural formula • Functional group: -COOC- • Alcohol + carboxylic acid ester + water • Condensation reaction or esterification Amines • Identify when present in structural formula • Relatives to ammonia; weak base • Functional group –NH2 • H-bonds => higher bp’s, smaller ones are water-soluble etc. • Name: stem + suffix: -ylamine (or prefix amino-) • Ethylamine CH3-CH2-NH2 10.5 Halogenoalkanes Reactions Bond enthalpy kJ/mol Bond in CH3-CH2-X 484 338 276 238 C-F C-Cl C-Br C-I C—X d+ d=> Iodine compounds most reactive Nucleophiles and electrophiles- often needed in organic reactions • Nucleophile- nucleus lover • Has free electronpair and whole or part negative charge • The larger the negative charge - the better the nucleophile • Eg: C=C, H2O, -OH, -CN, NH3 • Electrophile-electron lover • Has whole or part positive charge • The larger the positive charge - the better the Electrophile • Eg: C=O, H+, C-Cl, Substitution reactions CH3-CH2-Cl (aq) + -OH (aq) CH3-CH2-OH (aq) + Cl- (aq) • The nucleophilic hydroxide ion, OH-, attacks the positively charged, electrophilic carbon • Curly arrows are used in mechanisms to show how electron pair moves • The substitution reaction can proceed by two different pathways, mechanisms SN1 and SN2 SN2 • Substitution Nucleophilic bimolecular • Bimolecular = two species in the rate determining step. Rate = k [org]*[Nu] • Favoured when primary halogenoalkanes. Less steric hindrance from neighbouring groups. • HL: If reactant is chiral (optic active) the product is also optic active. But with inversion in the structure. SN2 Mechanism Nucleophile attack Transition state Leaving group Bonds breaks and forms Enthalpy diagram for SN2 Enthalpy Reaction coordinate SN1 • Substitution Nucleophilic monomolecular • Monomolecular = one species in the rate determining step. Rate = k [org] • The rate determining step is the formation of a carbocation, an intermediate, which is only stable on a tertiary carbon • Favoured when tertiary halogenoalkanes (electrophile). The formed carbocations are stabilised by inductive effect. Stability of carbocations Mechanism for SN1-reaction Heterolytic fission Enthalpy diagram for SN1-reaction . 10.6 Reaction pathways • Deduce reaction pathway given the starting materials and the product • Conversions with more than two stages will not be assessed. Reagents, conditions and equations should be included • The compound and reaction types in this topic are summarized in the scheme on the next slide 1-3. Substitution via radical mechanism. Induced by homolytic cleavage of Cl2 by UV-light. 4. Addition reaction. Hydrogen halide, spontaneous at STP 5. Addition reaction. H2 and Ni-catalyst 6. Addition reaction. Halogene, spontaneous at STP 7. Poly-addition. Radical mechanism. Initiation, prolongation and termination 8. Substitution reaction with NaOH; SN1 or SN2 9. Oxidation of primary alcohol with acidified K2Cr2O7. Distillation to get the product 10. Oxidation of primary alcohol with acidified K2Cr2O7. Reflux to get the product 11. Oxidation of secondary alcohol with acidified K2Cr2O7