Valence Electron Practice Worksheet Answers
advertisement
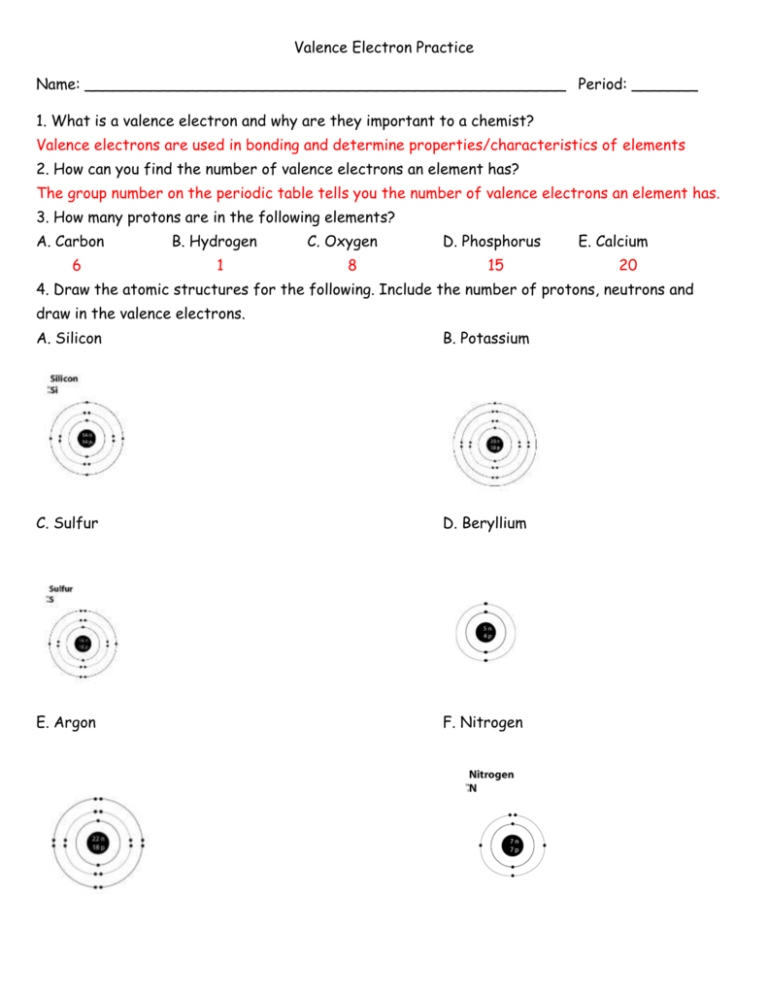
Valence Electron Practice Name: ___________________________________________________ Period: _______ 1. What is a valence electron and why are they important to a chemist? Valence electrons are used in bonding and determine properties/characteristics of elements 2. How can you find the number of valence electrons an element has? The group number on the periodic table tells you the number of valence electrons an element has. 3. How many protons are in the following elements? A. Carbon 6 B. Hydrogen 1 C. Oxygen 8 D. Phosphorus 15 E. Calcium 20 4. Draw the atomic structures for the following. Include the number of protons, neutrons and draw in the valence electrons. A. Silicon B. Potassium C. Sulfur D. Beryllium E. Argon F. Nitrogen 5. Indicate whether the following elements will GAIN or LOSE electrons to get a full outer shell, and how many electrons they will need to GAIN or LOSE. Example: Chlorine has 7 outer shell electrons, so it will GAIN 1 electron to get a full outer shell. A. Lithium C. Calcium E. Boron G. Tin Lose 1 Lose 2 Lose 3 Gain/Lose 4 B. Bromine D. Oxygen F. Argon H. Phosphorus Gain 1 Gain2 Neither Gain 3 6. What will the charge be of the following element when they GAIN or LOSE electrons to become stable? A. Aluminum C. Nitrogen E. Sulfur G. Magnesium -3 -2 +2 +3 B. Iodine D. Potassium -1 F. Neon +1 0 H. Carbon -4 or+4 7. Write the symbol and the charge for the atoms given below and identify the element as a cation/anion and metal/nonmetal. Element Symbol Charge Metal or Nonmetal Anion or Cation Calcium Ca +2 Metal Cation Bromine Br -1 Non-metal Anion Nitrogen N -3 Non-metal Anion Fluorine F -1 Non-metal Anion Aluminum Al +3 Metal Cation Phosphorus P -3 Non-metal Anion Li +1 Metal Cation Lithium




