Inorganic Nomenclature Caitlin, Safia, Madi, Holly
advertisement

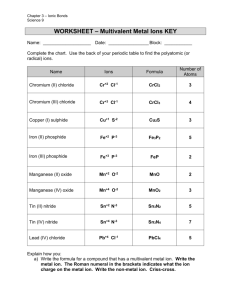
By: Madi, Caitlin, Safia, and Holly 1. First, write the formula for the positive ion and write the formula for the negative ion second. (POSITIVE goes first, NEGATIVE goes second) 2. “Criss-cross” the numbers in front of the charges on the ions. 3. Tidy up the formula in a threepart process: i) if both subscripts can be evenly divided by “2” (or “3”), do so. ii) omit the subscripted charges iii) omit any subscript which is a “1” Example: Calcium Phosphide Calcium Ion= C2+ Phosphide Ion= P 3 1)Criss cross the charges 2+ C P 2) C 3 P2 3- If the first ion in the compound only has one possible charge, simply write the names of the ions one after another. 2. If the first ion is a metal known to possess, more than one possible charge OR a metal which is not on the table, “un-criss-cross” the subscripts and use them as charges. 1. 1) Name the metal ion 2) look at the periodic table to see if the metal has more than one combining capacity 3) determine the combining capacity of the metal by looking at the formula of the co brackets write compound 4) in brackets write the metals combining capacity in roman numerals 5) name the non-metal with the ending “ide” Example: Cr2 O3 1) Cr 2+ and Cr3+ 23+ 2)Cr 2+O3+ = O and Cr from un criss crossing the combining capacity, we can see that Cr has a combining capacity of 3+ 3) chromium (III) 4) chromium (III) oxide 1) identify each ion and its charge. Ex chromium (III) nitride= Cr3+ N3 2) criss cross charges Ex Cr3+ 3) put the symbols together and get rid of the charge values ex Ca3N2 4) if the subscript ratio is not the smallest possible ratio, make them into the smallest possible ratio Ex Cr3N3= CrN Example: +1 2 1) Cu Se 2) Criss cross charges +1 Cu Se 3) Cu 2 Se 2- 1)Write the positive ion name (metal or ammonium) - if metal is a multivalent ion, you must add roman numerals after the first name in brackets. 2)Identify negative ion, name it and reverse the criss-cross of the subscripts. Example: Cr(PO4)2 The symbol “Cr” translates into: chromate Cr(PO4)2 = chromate(II) PO4 = phosphate chromate (II) phosphate 1) First identify the symbol Example: of the metal and its charge as well as the ion symbol and its charge 2) Criss-cross charges 3) Put the symbols together, using the same order as the name. 4) Reduce the charge values chromium (III) sulphate Cr 3+ SO4-2 * whole ion has subscript value =Cr2(SO4)3 *For polyatomic ions use brackets on (ion) 1) You identify the two elements in the formula. Find the symbols for each element. 2) Write out the symbols in the same order as they came in the name (make sure to leave a space between when written out) 3) Using the prefixes in front of each element find out which number corresponds to that element. 1 2 3 4 5 6 7 8 9 10 mono di tri tetra penta hexa hepta octa nona deca 4) Use the corresponding number for each element name and place it to the bottom right of the symbol from step 1). Example: NO 1 is the subscript for each (*you don’t write mono for the first element with only one as a subscript) NO= nitrogen monoxide 1) Identify the subscript value of the first element. 2) Write down the prefix corresponding to the subscript of the first element. 3) Write down the first atoms name (the prefix becomes the first syllable of the element ex. tetrachlorine) 4)Identify the subscript value of the second element 5) Write down the prefix corresponding to the fifth step. 6) Write down the seconds atoms name, ending in “ide” (the prefix becomes the first syllable of the element ex. pentaoxide) Example: N2 O4 di + nitrogen + tetra + oxide dinitrogen tetra oxide 1) First you name the Example: FeSO4 compound as you normally would depending on the type of compound. 2) After the compound name, place the corresponding prefix (# of water molecules) in front of the word hydrate. Fe= iron (II) . 5H2O SO4= sulphate 5H 2O = pentahydrate = iron (II) sulphate pentahydrate 1) You write out the Example: iron (II) symbols for the element sulphate pentahydrate the ion and the symbol for water. pentahydrate = 5H2O 2) Next place the number sulphate= SO4 correlating with the 2+ iron (II) = Fe prefix in front of H2O 3) Reverse criss-cross to find any combining capacities. 4) Write it all out.