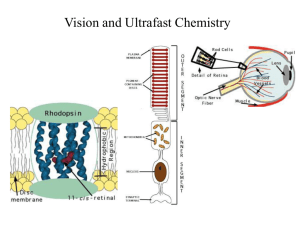
06-spectral-tuning
advertisement
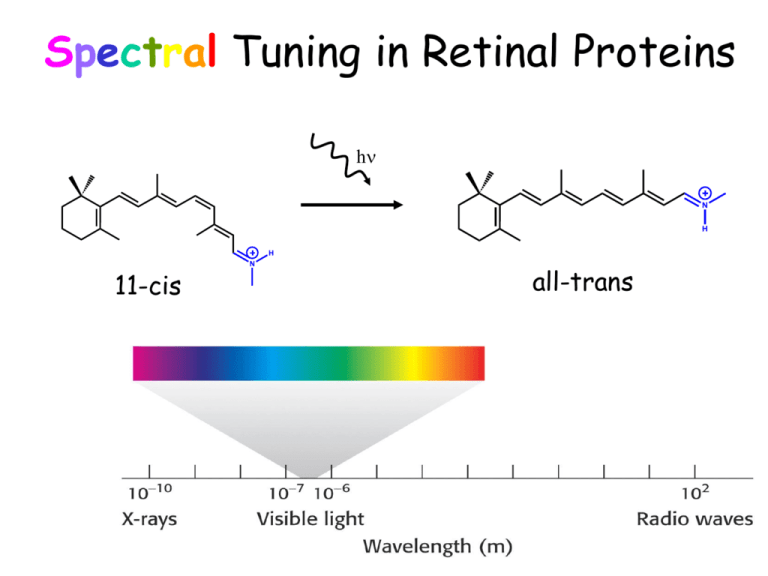
Spectral Tuning in Retinal Proteins hn N H H 11-cis N all-trans Color Vision hn N H 11-cis H N all-trans Visual Receptors Rod G-protein signaling pathway Rhodopsin Light Cone Spectral tuning in color visual receptors Color is sensed by red, green and blue rhodopsin visual receptors. 500nm 400nm absorption spectrum 11-cis H 600nm Their chromophores are exactly the same! N How does the protein tune its absorption spectrum? Spectral Tuning in bacteriorhodpsin’s photocycle Me Me Me N Me Me H How can we change the maximal absorption of retinal chromophore? Me Me Me Me N H Me Me Me Me Me Me H N Excitation energy determines the maximal absorption S1 S0 Response Me Me Me 13 7 Me Me 9 11 15 N H Electronic Absorption Me Me Me Me N H Me p-p* Absorption of light in the UV-VIS region of the spectrum is due to excitation of electrons to higher energy levels. p-p* excitation in polyenes p* p* DE photon E p Ground state (S0) p Excited state (S1) DE (excitation energy, band gap) = hn = hc/l p-p* excitation in polyenes p* p* p* p p E p blue-shift red-shift p-p* excitation in polyenes Tuning the length of the conjugated backbone b-carotene O O Vitamin A2 (retinal II) Longer wavelength Vitamin A1 (retinal I) Short wavelength Retinal I Retinal II Salmon: different retinals in different stages of life OPSIN SHIFT: how protein tunes the absorption maximum of its chromophore. Maximal absorption of protonated retinal Schiff base in: Water/methanol solution: 440 nm bR: 568 nm rod Rh: 500 nm red receptor: 560 nm green receptor: 530 nm blue receptor: 426 nm Electrostatics and opsin shift S2 positive charge Me Me + S1 Me + S2 S1 N Me Me H O S0 O C Asp (Glu) counterion S0 no protein in protein • The counterion stabilizes the positive charge of the chromophore. •The position of the counterion determines how and how much the band gap energy changes. Electrostatics and opsin shift S2 positive charge Me Me + S1 Me + S2 S1 N Me Me H O S0 O C Asp (Glu) counterion S0 no protein in protein Maximal absopriton of protonated retinal Schiff base can be changed by D85N (Purple to blue shift) Red shift from 568 to 605 nm at pH = 3 Dinosaurs had red-shifted visual receptors! Howard Hughes Medical Institute at The Rockefeller University and Yale University Microbial rhodopsins in Halobacteria purple membrane Sensory rhodopsin I (sRI): attractant (repellent) to orange (near UV) light Sensory rhodopsin II (sRII): repellent to blue-green light phototaxis (color vision of halobacteria) Bacteriorhodopsin (bR): proton pump Halorhodopsin (hR): chloride pump sRII hR bR 500nm sRI 600nm Spectral Tuning in Bacterial Rhodopsins Sensory Rhodopsin II (sRII) Phototaxis • Large blue shift of absorption maximum in sRII (70 nm) Bacteriorhodopsin (bR) Proton pump sRII 500nm bR 600nm Structures of bR and sRII X-ray crystallography shows that structures are very similar. Both include protonated alltrans retinal Schiff base orange: sRII purple: bR Binding Sites of bR and sRII sRII bR Similar structure • Aromatic residues. • Hydrogen-bond network. (counter-ion asparatates, internal water molecules) Mutagenic substitutions T204A/V108M/G130S of sRII produces only 20 nm (30%) spectral shift. What is the main determinant(s) of spectral tuning? QM/MM Calculation of spectral shift in bR and sR-II • Refinement of X-ray structures by HF (retinal, 2Asp, 3H2O) • Excitation energy calculations for retinal QM/MM helix G retinal-K205/216 D201/212 bR: purple, sRII: orange Calculated spectra DE(S1-S0) Spectral shift sRII bR DE(S1-S0) : 6.1 (exp. 7.2) kcal/mol DE(S2-S0 ): 1.7 (exp. 4.0) kcal/mol 500nm 600nm A sub-band in sRII is due to the second excited state (S2). Deprotonation of the Schiff base Me Me Me Me N H UV vision birds, honeybee Me Me Me Me Me N Me Strong blue shift p atomic orbitals Planarity is essential for maximal overlap of p orbitals in a double bond (p molecular orbital) Steric interactions and spectral shift? A highly twisted structure can decrease the overlap of p orbitals and effectively decrease the length of the conjugation, i.e., blue shift. Summary of Mechanisms of Spectral Tuning • Using a different chromophore with a longer or a shorter conjugated chain • Modifying the amino acid composition of the binding pocket (electrostatics) • Manipulating the distance and/or conformation of charged/polar groups in the vicinity of retinal • Steric interaction with the chromophore so that some of the double bonds go out of plane (a similar effect to using a shorter chromophore) • Protonation state of retinal Schiff base (Strong blue shift upon deprotonation) Me Me Me Me N H Me Me Me Me Me N H Me The chromophore retinal adopts different colors in different environments. Doesn’t it remind you of something? cameleon