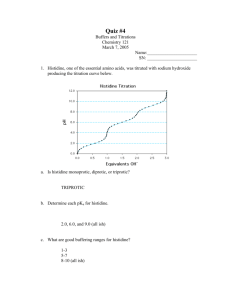
pptx
advertisement

Unless otherwise stated, all images in this file have been reproduced from: Blackman, Bottle, Schmid, Mocerino and Wille, Chemistry, 2007 (John Wiley) ISBN: 9 78047081 0866 e CHEM1002 [Part 2] A/Prof Adam Bridgeman (Series 1) Dr Feike Dijkstra (Series 2) Weeks 8 – 13 Office Hours: Room: e-mail: e-mail: Monday 2-3, Friday 1-2 543a adam.bridgeman@sydney.edu.au feike.dijkstra@sydney.edu.au Slide 2/16 e Acids and Bases Lecture 1: • Common Acids and Bases • Definitions • Equilibria • Conjugate acid-base pairs • Autoionisation of water • pH Lecture 2: • Strong Acids and Bases • Weak Acids and Bases • Polyprotic Acids Reproduced from ‘The Extraordinary Chemistry of Ordinary Things, C.H. Snyder, Wiley, 2002 (Page 245) Slide 3/16 e Strong Acids and Bases • Completely ionise in water: e.g. HCl(aq) H+(aq) + Cl–(aq) • Equilibrium lies completely to the right, Ka . Strong acids H2SO4, HCl, HBr, HI, HNO3, HClO4 Strong bases All hydroxides of Groups 1 & 2 (except Be): NaOH, Ca(OH)2, … Slide 4/16 x Examples • What is the pH of a 0.1 M HCl solution? HCl(aq H+(aq) + Cl-(aq) [H+] = 0.1 M Thus [H+] = 0.1 M and pH = – log10 [H+] = 1.0 • What is the pH of a 0.002 M NaOH solution? Completely ionised, so [OH– ] = 0.002 M, pOH = – log10 [OH– ] = – log10 (0.002) = 2.7 pH = 14 – 2.7 = 11.3 Slide 5/16 e Weak Acids • Most acids or bases are weak they do not completely ionise in water H+(aq) + A–(aq) [H ][A ] Acid dissociation constant: K a [HA] pKa = – log10Ka HA(aq) Slide 6/16 e Relationship between Ka and pKa • The larger the value of Ka, the stronger the acid and the lower the value of pKa. Acid Name (Formula) - o K a at 25 C -2 pK a Bisulfate ion (HSO4 ) 1.02 x 10 Nitrous acid (HNO2) 7.1 x 10 3.15 Acetic Acid (CH3COOH) 1.8 x 10-5 4.74 Hypobromous acid (HBrO) Phenol (C6H5OH) 2.3 x 10 -10 1.0 x 10 -4 -9 1.991 8.64 10.00 Ka = 1.02 x 10-2 then pKa = -log10(1.02 x 10-2) = 1.991 pKa = 1.991 then Ka = 10-1.991 = 1.02 x 10-2 Slide 7/16 x Example • Find the pH of 0.1 M acetic acid (CH3COOH (HAc)) DATA: pKa = 4.7, HAc(aq) Ka = 10-4.7 H+(aq) + Ac–(aq) Initial (I): 0.1 0 0 Change (C): -x +x +x x x Equilibrium (E): 0.1 - x Ka 104.7 Ka = 10-4.7 x2 0.10 x Slide 8/16 x Example (Continued) • Since the equilibrium constant is very small we assume x << 0.1, i.e. ( 0.1 – x ) 0.1 10-4.7 x2 / 0.1 x2 0.1 10-4.7 = 10-5.7 x 10-5.7 = 10-2.85 • As pH = – log10 [H+ ] pH = – log10 x = – log10 10-2.85 = 2.9 • Check: x = 10-2.85 = 1.4 10-3 << 0.1 or or (0.1 – x ) = 0.0986 M ~ 0.1 M 5 % rule Slide 9/16 e Weak Bases • Ionisation of a weak base: NH3(aq) + H2O(l) NH4+(aq) + OH–(aq) • Equilibrium constant is called base ionisation constant, Kb : Kb [NH4 ][OH ] [NH3 ] • Tactic: calculate pOH and then pH, given Kb. Slide 10/16 e Relationship Between pKa and pKb • For conjugate systems (Brønsted-Lowry acid-base pairs) Acid (HA): HA(aq) H+(aq) + A–(aq) Conjugate base (A– ): A–(aq) + H2O(l) HA(aq) + OH–(aq) [H ][A ] Ka [HA] [HA][OH ] Kb [A ] [H ][A ] [HA][OH ] Ka Kb [HA] [A ] [H ] [OH ] Kw 10 14 pKa + pKb = 14 We only need values of pKa , since pKb = 14 – pKa Slide 11/16 x Example • Find the pH of 1.0 x 10–2 M NaHCO2 (pKa of formic acid (HCO2H) is 4.1) pKb = 14 – pKa = 14 – 4.1 = 9.9 HCO2–(aq) + H2O(l) OH–(aq) + HCO2H(aq) Initial (I): 0.01 Change (C): -x +x +x Equilibrium (E): 0.01 - x x x Kb [OH ][HCO2H] [HCO2 ] large so 10 9.9 0 0 x2 10 2 x x2 102 Slide 12/16 x Example (Continued) Kb x [OH ][HCO2H] [HCO2 ] so 10 9.9 102 109.9 x2 10 2 x x2 102 1011.9 105.95 • x = [OH–] so pOH = -log10(10-5.95) = 5.95 pH = 14 – 5.95 = 8.05 = 8.1 (one significant figure) Slide 13/16 e Polyprotic Acids H3PO4(aq) H+(aq) + H2PO4–(aq) pKa1 = 2.2 H2PO4– (aq) H+(aq) + HPO42–(aq) pKa2 = 7.2 HPO42-(aq) H+(aq) + PO43–(aq) pKa3 = 12.4 • removing more protons is harder: increasing pKa = decreasing Ka : Ka1 > Ka2 > Ka3 reason: harder to remove +ve charge against increasing -ve charge • large difference in pKa values only need to consider one equilibrium at a time (simplifies maths!) Slide 14/16 x Practice Examples 1. Which one of the following is NOT a conjugate acid-base pair? (a) HCO3– and CO32– (b) H3O+ and H2O (c) OH– and O2– (d) SO3 and HSO3– (e) NH2OH2+ and NH2OH 2. What is the pH of a 0.20 M solution of boric acid? The pKa of boric acid is 9.24. (a) 0.70 (b) 2.73 (c) 4.97 (d) 5.12 (e) 5.87 3. Rank the following solutions (all 1.0 M) in DECREASING order of pH. NaCl, NaCN, KOH, HCl, CH3COOH (a) (b) (c) (d) (e) HCl > CH3COOH > NaCN > NaCl > KOH KOH > NaCl > NaCN > CH3COOH > HCl KOH > NaCN > NaCl > HCl > CH3COOH KOH > NaCl > NaCN > HCl > CH3COOH KOH > NaCN > NaCl > CH3COOH > HCl Slide 15/16 e Summary: Acids & Bases 2 Learning Outcomes - you should now be able to: • • • • • Complete the worksheet Complete acid/base calculations Use pH, pKw, pKa and pKb Define strong and weak acids & bases Answer Review Problems 11.12-11.35 in Blackman Next lecture: • Buffer systems Slide 16/16